Chapter2: Basic Statistical Analysis With Excel
Section: Chapter Questions
Problem 12P
Related questions
Question
Q1
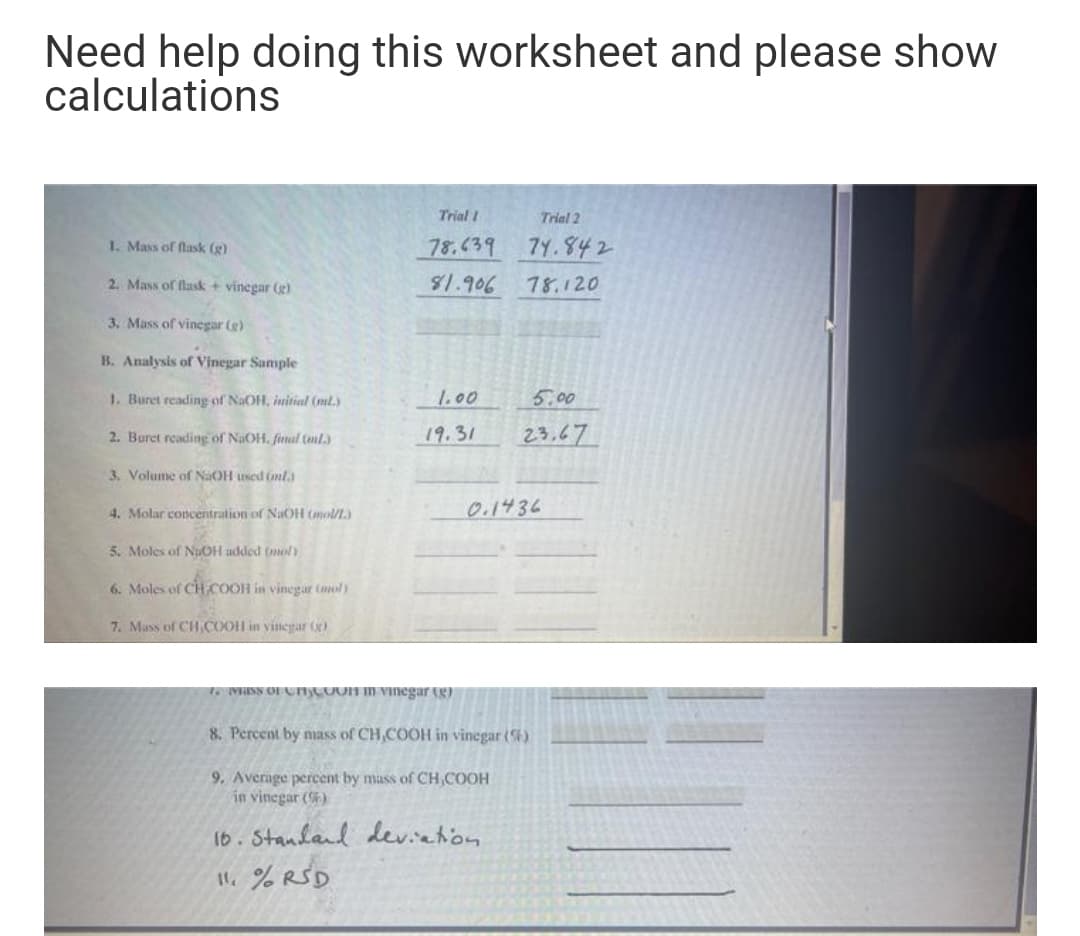
Transcribed Image Text:Need help doing this worksheet and please show
calculations
Trial I
Trial 2
1. Mass of flask (g)
78.639
7Y.842
2. Mass of flask + vinegar (g)
81.906
78.120
3. Mass of vinegar (e)
B. Analysis of Vinegar Sample
1. Buret reading of NaOH, initial (ml.)
1.00
5.00
2. Burct reading of NaOH. final (ml.)
19.31
23,67
3. Volume of NaOH used (nl.
4. Molar concentration of NaOH (mol/.)
0.1436
5. Moles of NuOH added (nol)
6. Moles of CHCOOH in vinegar (amoly
7. Mass of CH,COOH in vinegar ()
16 MHRNS OFCTCOON IN Vinegar g)
8. Percent by nass of CH,COOH in vinegar (4)
9. Average percent by mass of CH,COOH
in vinegar ()
16. Standand deviation
11.% RSD
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you



