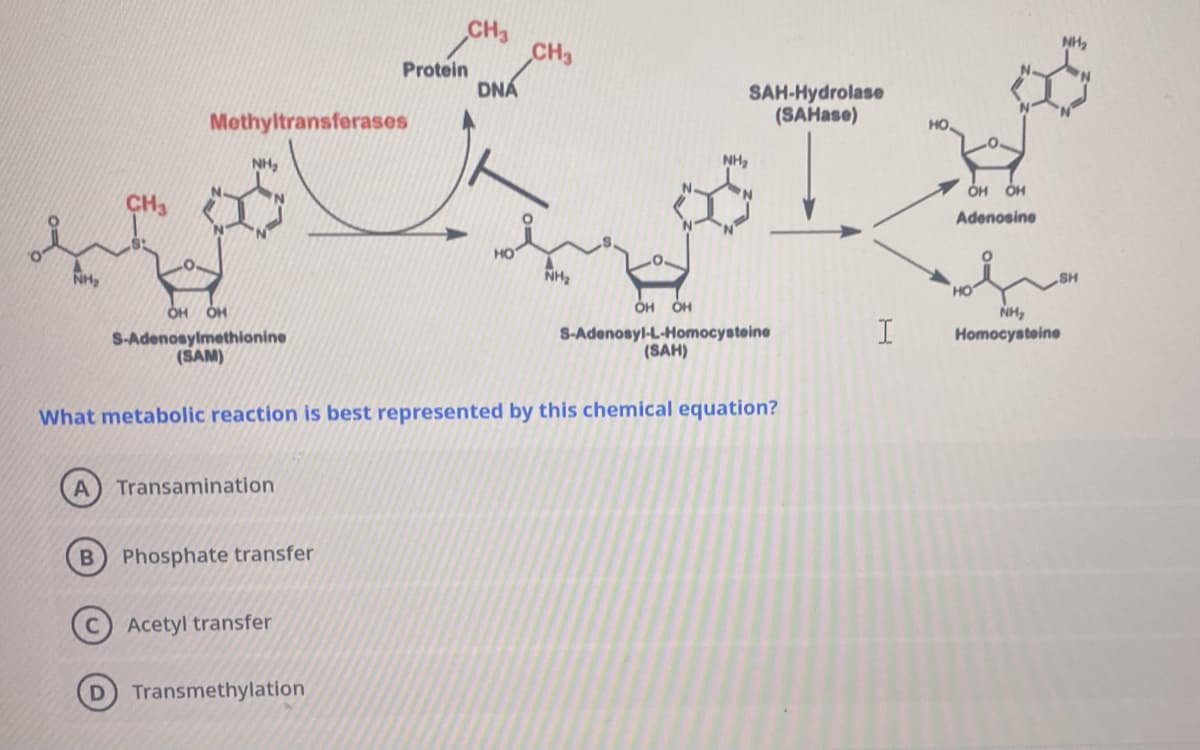
NH₂ Methyltransferases NH₂ OH OH S-Adenosylmethionine (SAM) Transamination B Phosphate transfer Protein Acetyl transfer Transmethylation CH3 DNÁ HO CH3 NH₂ What metabolic reaction is best represented by this chemical equation? SAH-Hydrolase (SAHase) NH₂ OH OH S-Adenosyl-L-Homocysteine (SAH) I OH OH Adenosine NH₂ SH NH₂ Homocysteine
Q: How can a noncompetitive inhibitor affect enzyme activity without binding to the active site? a. It…
A: The substance that decreases the activity of enzyme is called enzyme inhibitor. There are two types:…
Q: Which of the following statements concerning SDS in SDS-PAGE is INCORRECT? a. SDS is a negatively…
A: SDS-PAGE is an electrophoresis technique used to separate protein molecules. Protein molecules can…
Q: Question 23 of 25 Among the given statements, which ones describe the lysosomes? Select the correct…
A:
Q: Please help with 2a) 2a) There are two different DNA polymerase enzymes, DNA Polymerase I and DNA…
A: Replication is the process of duplication of two strands of a double stranded DNA. In bacteria, the…
Q: a) What is the Kd you measure for this interaction?
A: INTRODUCTION : Actin - Actin is a family of globular multi-functional proteins which form…
Q: For the tetrapeptide YIRG: a. Draw its complete protonic equilibria. Indicate the net charge of each…
A: Proteins are composed of amino acids, which are bound together by peptide linkage. Amino acids…
Q: Which one of the following statements about PEP (phosphoenolpyruvate) synthesis is correct? Pyruvate…
A: PEP, also known as 2-phosphoenolpyruvate, is a crucial biochemical intermediary. It participates in…
Q: Detergents disrupt hydrophobic interactions by coating hydrophobic molecules with molecules that…
A: Hemoglobin is an oligomeric protein with four polypeptides. The individual polypeptides are joined…
Q: A generalized enzyme active site is shaped like a hemisphere with a radius of 45Å. The active site…
A: Given that the active site is made up of HAVARILKHAVARILKHAVARILK amino acids at pH 7.38. The net…
Q: Describe how a protein’s structure determines its function. What determines a protein’s structure?
A: Proteins have four levels of conformations known as primary, secondary, tertiary, and quaternary. In…
Q: Given the following reaction below, what amino acid is involved and what is the specific reaction…
A: Phenylalanine, tyrosine, and tryptophan are aromatic amino acids. The side chain of the…
Q: Describe the p-nitrophenol assay and how a standard curve can be used to calculate the concentration…
A: An assay is a biochemical lab procedure to determine the quantity of molecules of interest in a…
Q: 3. Briefly describe and explain the shape of the curve in Q2.
A: In an enzyme-catalysed reaction, the substrate binds reversibly to the enzyme's active site to form…
Q: Calculate ΔGinward. Is energy required for transport to happen? The internal pH of a lysosome= 4.0…
A: The amount of change in free energy i.e. change in Gibbs free energy for the transport of an ion…
Q: Which statements best describe serotonin? Select the correct response(s): It is essential for…
A: Serotonin is a neuro transmitter that transmit the messages from one neuron to other neuron. It is…
Q: is device used to measure temperature.
A: The device used to measure temperature is .........
Q: Which of the amino acids does NOT have an aliphatic side chain? a. Lysine b. Alanine…
A: R groups in aliphatics are nonpolar and hydrophobic. The hydrocarbon chain's hydrophobicity rises as…
Q: A Biuret test is a chemical test used to determine the presence of a peptide bond in a substance.…
A: The biuret test detects the presence of proteins in a sample solution. The copper ions in the biuret…
Q: Question - is if the cells in our bodies were to convert the required energy into our food substance…
A: Energy must be supplied continuously for living things to exist. This energy is employed in part to…
Q: Which is the strongest non-covalent interaction that occurs between triglycerides: hydrogen bonds,…
A: - A non-covalent bond is one in which there is no sharing of electron pairs. It mainly occurs…
Q: Which test can you use to distinguish a monosaccharide from a disaccharide? hexoses from pentoses?…
A: Any of the fundamental substances that form the basis of carbohydrates are referred to as simple…
Q: Give 2 structural features in each of the given proteins below that make them perform their…
A: Hemoglobin is an oligomeric, conjugated protein with four polypeptides. The function of the…
Q: 8. Why is it important to maintain blood glucose during exercise? How is this achieved?
A: During exercise, it is important to maintain glucose homeostasis. There is a tight regulatory…
Q: referred to feedback inhibition as “Goldilocks regulation”. Describe how feedback inhibition keeps…
A: Biochemical pathways and reactions are occurring constantly in cells. These pathways convert the…
Q: Calculate ΔGinward. Is energy required for transport to happen? The internal pH of a lysosome= 4.0…
A: The amount of change in free energy i.e. change in Gibbs free energy for the transport of an ion…
Q: The active site _______________. a. is the compound that an enzyme reacts with during the chemical…
A: The active site is that region of an enzyme where substrate molecules bind. The binding of substrate…
Q: Which of the following experiments can be used to determine the localization of a protein in living…
A: Confocal microscopy is an optical imaging technique used for increasing optical resolution and…
Q: The starting substrate and active site of a Type I topoisomerase is shown below. During this…
A: Topoisomerases change the topology of DNA by breaking either one or two of the DNA strands and allow…
Q: A dialysis tube that contains 10 mL of a 5% sucrose solution and that is permeable to water but not…
A: Osmosis is a process by which a water moves from higher water concentration to lower water…
Q: What is the net ionic charge for the peptide at pH 5 and pH 11? The peptide is…
A: A peptide is a short chain of amino acid residues linked via a peptide bond. As a general rule of…
Q: What is one technique or property of a protein that you could use to monitor the fractions so you…
A: In column chromatography, there is a stationary phase and the mobile phase. The stationary phase…
Q: discuss the biochemistry behind disorders related to aromatic amino acids with the aid of an…
A: Metabolic disorders are caused due to abnormalities in one or more processes in a metabolic pathway.…
Q: The mitochondrial matrix is home for the following: I. ribosomes II. circular DNA III. Kreb's…
A: Mitochondria is a membrane bound organelle which is also known as power house of the cell as it is…
Q: Which of the following statements is CORRECT? A) Hexokinase IV is allosterically inhibited by…
A: Enzyme plays an important role in all the metabolic activities in our body. They themselves remain…
Q: Choose the best answer The active site of an enzyme is the place where the following happens:…
A: A catalyst is a component that speeds up a chemical reaction without really being a reactant.…
Q: Which of the following concerning hemoglobin is true? a) BPG decreases at high altitudes and causes…
A: 1 Hemoglobin is a globular protein, ie it is roughly spherical. It is an oxygen-binding protein that…
Q: How many cycles of the synthesis pathway are needed to produce lauric acid, C₁1H23COOH? ||…
A: The production of triglycerides from acetyl-coenzyme A (acetyl-CoA) subunits is known as…
Q: 5. State whether the following statements are True or False, If False, explain why 5.1 Oxaloacetate…
A: 1. Oxaloacetate is formed by the oxidation of L-malate - Oxaloacetate is formed via oxidation of…
Q: Nomenclature of vitamins (empirical, biochemical, medical).
A: Vitamins are organic nutrients which can not be synthesized in the body, hence, need to be supplied…
Q: The brown discoloration of apples and potatoes is due to an enzyme catalyzed chemical reaction.…
A: Cut apples and potatoes turn brown or discolored on exposure to air. This is due to conversion of a…
Q: Based on what you know about what powers ATP synthesis and how NADH and FADH2 interact with various…
A: Introduction Cellular respiration is a process by which glucose molecule breaks and produce carbon…
Q: quantitative testing methods. Discuss two of them. (macromolecules lab questions) ( tests for…
A: Qualitative tests tell us whether or not the biomolecule of interest is present in a sample.…
Q: 1a Briefly describe or explain what the term "supercoiling" means in the context of DNA structure.…
A: Supercoiling means the coiling of the coil. Cellular DNA is extremely compacted and implies a high…
Q: ) What would be the predominant ionization states of this amino acid at physiological pH (7.4) and…
A: Amino acids contain an alpha-amino group, an alpha-carboxylic group, and a side chain. The side…
Q: Draw the structure(s) of the following amino acids (Asp, Lys, Phe, and Val) at pH 3.5, pH 6.5, and…
A: Amino acids are biomolecules that have an ionisable amino group and an ionisable carboxyl group…
Q: Phosphatidic acid phosphatase (PAP), also called lipin, converts phosphatidate to diacylglycerol…
A: Phosphatidic acid phosphatase (PAP) or lipin converts phosphatidate to diacylglycerol (DAG), while…
Q: For the questions below be sure to show your work as well as the answer to the question. In this…
A: Hi! Thank you for the question. We are authorized to answer five subparts at a time, since you have…
Q: If there is ample oxygen available, how many ATP can be produced from complete oxidation of glucose…
A: The aerobic breakdown of nutrients into carbon dioxide, water, and energy occurs during aerobic…
Q: Why can plant material be substituted? Plants contain high amounts of saturated fats. Certain plants…
A: Introduction : Linoleate - A linoleate is a salt or ester of linoleic acid(which is an organic…
Q: When one molecule of glucose is processed through the glycolysis pathway, there will be: Net gain of…
A: All living cells use the catalytic process of glycolysis. The Embden-Meyerhof-Parnas (EMP) pathway…
Step by step
Solved in 2 steps

- Using the ActiveModel for aldose reductase, describe the structure of the TIM barrel motif and the structure and location of the active site.Given each set of information which may include common name(s) and the reaction catalyzed, you are required to identify the main class of the specific enzyme described. _____________________1. Name: alkaline phosphataseReaction: a phosphate monoester + H2O = an alcohol + phosphate_____________________2. Reaction: L-threonine = D-threonine.Other information: Inverts both chiral centers, a racemase. _____________________3. Name: glycine-N-acylaseReaction: acyl-COA + glycine = CoA + N-acylglycine_____________________ 4. Name: lysine decarboxylaseReaction: L-lysine = cadaverine + CO2 _____________________5. Name: methanol dehydrogenaseReaction: methanol + NAD+ = formaldehyde + NADH + H+ _____________________6. Name: citryl-CoA synthetaseReaction: ATP + citrate + CoA = ADP + phosphate + (3S)-citryl-CoA_____________________7. Name: D-xylulose reductaseReaction: xylitol + NAD+ = D-xylulose + NADH + H+ _____________________8. Name: cellobiose phosphorylaseReaction: cellobiose phosphate =…Galactose 1-phosphate: UDP-glucose uridyltransferase (GALT) is an enzyme important for the catabolism of galactose. Patients with GALT deficiency present within days of birth with feeding difficulty, fatigue, jaundice and liver problems, and failure to thrive. In addition, a buildup of galactose and its metabolites are present in the bloodstream. GALT catalyzes the following reaction in galactose catabolism. NOTE that GALT is reversible!! Classify this enzyme and explain why you classified it in this way.
- Assume that all carbon atoms and nitrogen atoms in both alanine and aspartate are labelled with radioisotopes. Then, the radioisotope-labelled alanine and aspartate molecules areallowed to undergo metabolism in the liver. All carbon atoms and nitrogen atoms in both ureaand glutamine are found to be labelled with radioisotopes. Please write down the metabolic reactions that lead to the incorporation of radioisotope-labelled carbon and nitrogen atoms into urea and glutamine.Dicyclohexylcarbodiimide (DCCD) is a reagent that reacts with Asp or Glu residues. Explain why the reaction of DCCD with the c subunits of F1F0-ATP ase blocks its ATP-synthesizing activity.Using the principles described in the text regarding pyridoxal phosphate mechanisms, propose a mechanism for the reaction catalyzed by serine hydroxymethyltransferase.
- Draw a schematic illustration of the hydrolysis of N-acetylphenylalaninamide catalyzed byalpha-chymotrypsin in which you indicate the important catalytic residues in the active site and how thesubstrate undergoes transformation to products through two different tetrahedral intermediates.Enolase catalyzes the reversible reaction shown below. Draw the detailed mechanism for the reaction that occurs in gluconeogenesis, namely the reaction of phosphoenolpyruvate to form 2-phosphoglycerate (the reverse direction in the reaction below).The high phosphoryl transfer potential of 1,3-bisphosphoglycerate is explained by the resonance stability of the product, 3-phosphoglycerate. The same explanation applies to the reaction that generates resonance stabilized pyruvate from phosphoenolpyruvate. Show the resonance structures for both products, 3-phosphoglycerate and pyruvate, and state why the reactant in both reactions is not resonance stabilized.
- The interconversion of DHAP and GAP greatly favors the formation of DHAP at equilibrium. Yet the conversion of DHAP by triose phosphate isomerase proceeds readily. WhyAssume that all carbon atoms and nitrogen atoms in both alanine and aspartate are labelledwith radioisotopes. Then, the radioisotope-labelled alanine and aspartate molecules areallowed to undergo metabolism in the liver. All carbon atoms and nitrogen atoms in both ureaand glutamine are found to be labelled with radioisotopes. Please write down the metabolicreactions that lead to the incorporation of radioisotope-labelled carbon and nitrogen atoms intourea and glutamine.In the hexokinase reaction (in glycolysis) Keq caclualtion, what is the OBVIOUS problem with getting a Keq of 678? what should keq be normally? how can you EXPLAIN a Keq of 678?

