Chapter7: Statistical Data Treatment And Evaluation
Section: Chapter Questions
Problem 7.19QAP
Related questions
Question
Transcribed Image Text:O My Apps
6 Schoology x
9 Vaping hez x
SIS Logout
E Microsoft x
E Draw Conc x
O World War x
W Ame-
Ô https://epsnj.schoology.com/common-assessment-delivery/start/5614731235?action=Donresume&submis
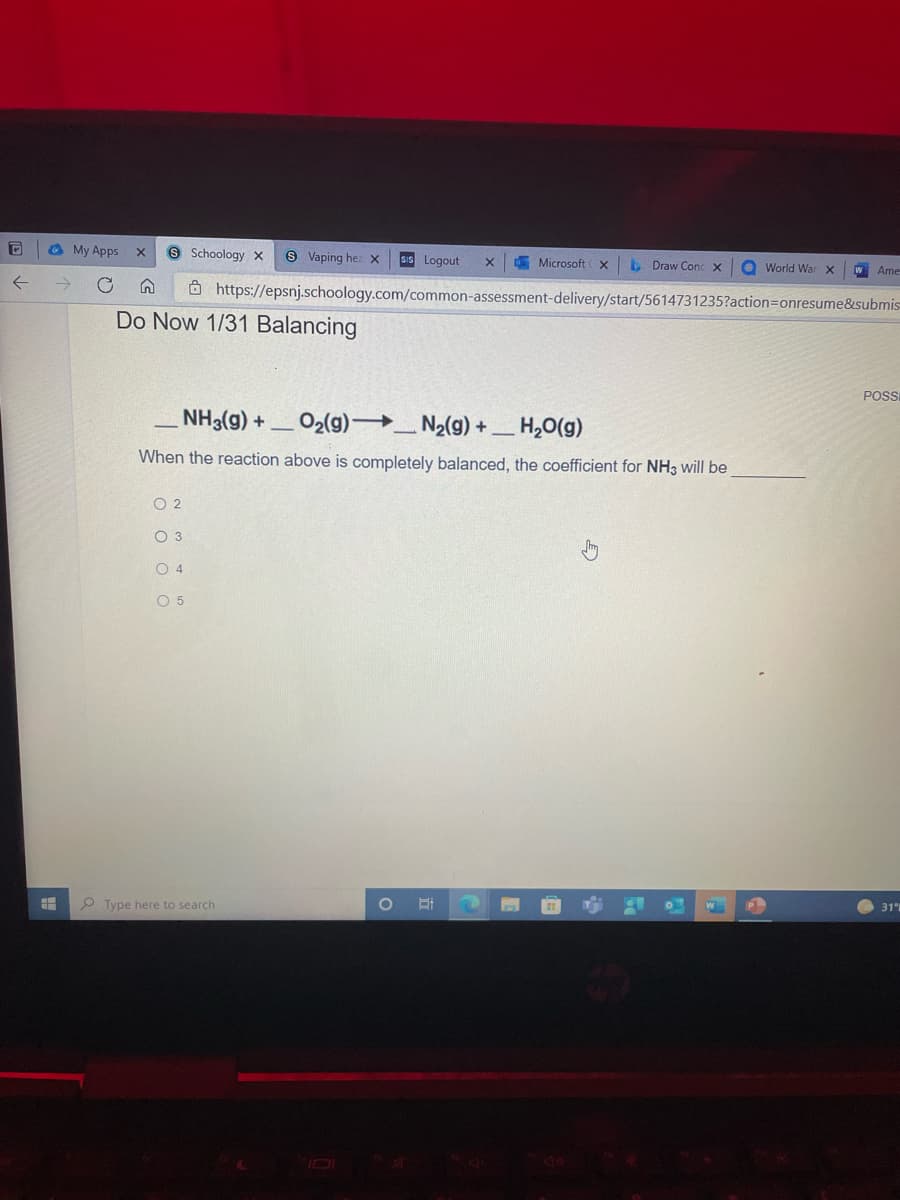
Do Now 1/31 Balancing
POS
NH3(g) +
O2(g) _ N2(g) + _ H,0(g)
When the reaction above is completely balanced, the coefficient for NH3 will be
O 2
O 3
O 4
O 5
31
P Type here to search
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning


General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning