NISTRATOR PRESENT Justin Ruiz: Attempt 1 O a) co b) N, c) Cl, d) CO, Question 12 (Mandatory) (1 point) V Saved The chemical formula of an ionic compound shows a) how many atoms of each element a molecule contains. b) how the atoms bond. c) the lowest whole-number ratio between ions in the ionic compound. d) which molecules the ionic compound contains. Question 13 (Mandatory) (1 point) v Saved Once formed, how are coordinate covalent bonds different from other covalent
NISTRATOR PRESENT Justin Ruiz: Attempt 1 O a) co b) N, c) Cl, d) CO, Question 12 (Mandatory) (1 point) V Saved The chemical formula of an ionic compound shows a) how many atoms of each element a molecule contains. b) how the atoms bond. c) the lowest whole-number ratio between ions in the ionic compound. d) which molecules the ionic compound contains. Question 13 (Mandatory) (1 point) v Saved Once formed, how are coordinate covalent bonds different from other covalent
ChapterU1: Alchemy: Matter, Atomic Structure, And Bonding
Section: Chapter Questions
Problem 7STP
Related questions
Question
Chem help
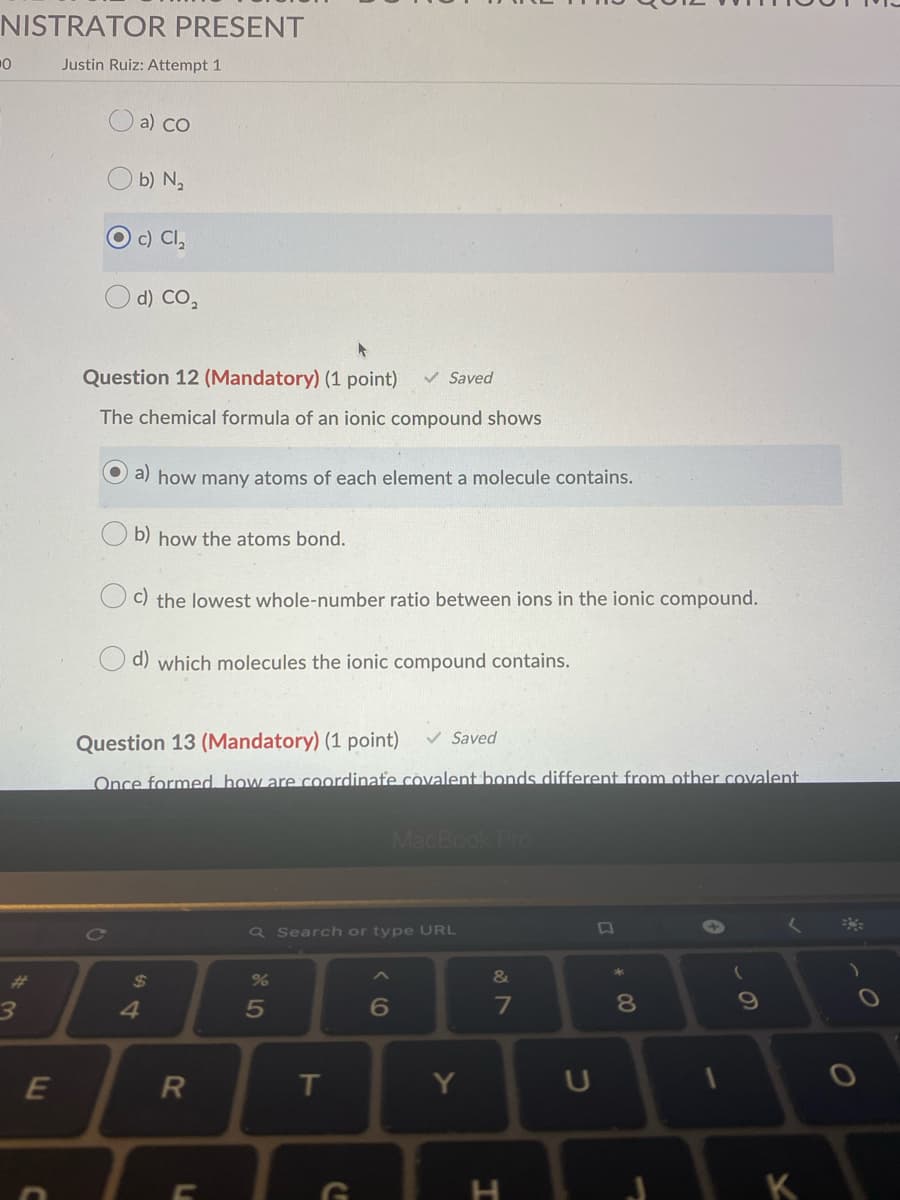
Transcribed Image Text:NISTRATOR PRESENT
Justin Ruiz: Attempt 1
a) CO
b) N,
O c) Cl,
O d) Co,
Question 12 (Mandatory) (1 point)
V Saved
The chemical formula of an ionic compound shows
O a) how many atoms of each element a molecule contains.
b) how the atoms bond.
c) the lowest whole-number ratio between ions in the ionic compound.
d) which molecules the ionic compound contains.
Question 13 (Mandatory) (1 point)
V Saved
Once formed, how are coordinafe covalent bonds different from other covalent.
MacBook Pro
C\
Q Search or type URL
%23
24
7
8.
E
T
Y
U
K
LI
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning


Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning