Q: Using Molar Mass and Avogadro Part B Caffeine is a bitter stimulant drug and is found in varying…
A: Caffeine had molecular formula C8H10N4O2 Means, 1 mole of caffeine has 8 moles of carbon, 10 moles…
Q: ▼ Part A Interactive 3D display mode H₂ H₂C Spell out the full name of the compound. Submit Part B…
A: IUPAC nomenclature of Alkenes: The longest carbon chain containing the carbon-carbon double bond is…
Q: How many mL of 0.1000 M sodium hydroxide are required to titrate 25.00 mL of 0.1502 M oxalic acid?
A:
Q: For the following "possible" reaction, which are the states of the products in order from left to…
A:
Q: COOH Ph
A: According to the IUPAC nomenclature, Carboxylic acid will get priority over ester in nomenclature.
Q: Question 2: The molecular formula of acetylsalicylic acid (aspirin), one of the most commonly used…
A: Dear student since you have posted multiple questions we will solve first question for you if you…
Q: 1. Methanol and ethanol are separated in a capillary GC column with retention times of 370 and 385…
A:
Q: (a) Explain the following observations: (1) (ii) (iii) For a given metal ion, the thermodynamic…
A:
Q: Match the correct explanations with the provided strengthening mechanisms. Strain hardening…
A: 1.Strain hardening is a condition in the stress- strain curve in the such strain is applied on a…
Q: A student measures the reaction H₂(g) + Cl₂(g) 2 HCl(g) concentrations at a few different…
A: Given that, the plot of lnK vs 1/T. From the graph, the linear fitting equation is y = 22585x -…
Q: 1. Molec Н. HO HO- H H- 0 -H -H -OH -OH CH₂OH D-mannose Draw the structures of each blomolecule…
A: Fischer , Haworth and chair conformational structure are given as:
Q: What is the mass in grams of KBr in 0.400 L of a 0.350 M solution?
A:
Q: 2.4e+05 2.2e+05 2.0E+05 1.8e+05 1.6e+05 1.4e+05- 1.2e+05 1.0e+05- 8.0E+04- 6.0E+04 4.0E+04- 2.0E+04-…
A: For 2-Methyl-2-pentanol, the number of signals expected are six. ( Refer to fig. In step 2) The OH…
Q: State the oxidation number of each atom in the following molecule
A:
Q: Calculate the molar mass for each of the following: 5,386 XX 27 Part A C₂H4O2 Express your answer…
A:
Q: (11) What is the hybridization of the central N atom in NO₂? N and O are in groups 5 and 6 and their…
A: Hybridization of central atom = electron pair ( number of orbitals which are involved in…
Q: The removal of hydrogen ion (H) from an acid will form its conjugate base, while the addition of H*…
A: In the above molecule there are four different types of protons labelled for which the acidity trend…
Q: An analytical chemist weighs out 0.085 g of an unknown monoprotic acid into a 250 mL volumetric…
A:
Q: Calculate the equilibrium concentrations of H2, I2 and HI, if the initial concentrations are 0.050 M…
A: Given, H2 + I2 ⇌ 2HI Initial concentrations are, [H2]initial = 0.050 M [I2]initial = 0.050 M…
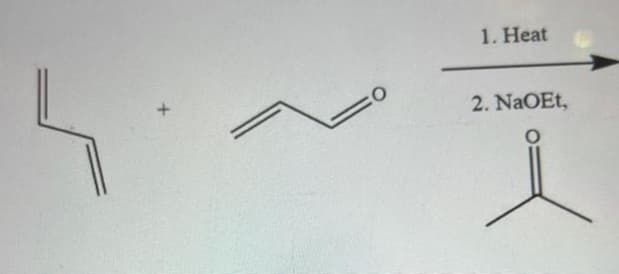
Q: Provide the major organic product of the following reaction. but-1-ene H₂O, H+ What type of…
A: Here we have to predict the type of regiochemistry observed in hydration of but-1-ene in presence of…
Q: (1) Give a reasonable Lewis structure, including formal charges, for HNC (N.B. N is the central…
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: 4.42 The reaction to form NO N₂+O₂ → 2N0 in high-temperature combustion processes is thought to…
A:
Q: Describe the arrangement of the elements in the modern periodic table.
A:
Q: OH−] = 1.5×10−2 M
A:
Q: VISUALIZATION The Photoelectric Effect Incident Light: Photon Flux ▶50% Incident Light: Frequency…
A:
Q: What will be the resulting products structure when 2-methyl-2-pentanol is treated with concentrated…
A: Given -> Reactants are -> 2-methyl-2-pentenol & HCl
Q: Predict the major organic product for each of the following reactions.
A:
Q: The principal component of natural gas is methane (CH4). How many moles of CH4 are present in 147.66…
A: Moles of methane can be calculated using the mass and molar masses of methane.
Q: Provide the correct systematic name for the compound shown here. 72°F o 2- OH CH₂-CH3 trans- cis-…
A:
Q: 40 °C O 70 °C O 50 °C 80 °C Te
A:
Q: What total mass of CO2(g) and H2O(l) is produced by the complete combustion (the reaction with…
A:
Q: 1. a. C. 2. a. C. Write a complete ground-state electron configuration for each of the following…
A: 1. Atomic number of Ar - 18 Atomic number of Rb - 37 Atomic number of S - 16…
Q: What is the correct net ionic equation when a solution of Na₂CO3 (aq) is mixed with a solution of…
A:
Q: Calculate the AHxn/mol Ca for the reaction: Ca (s) + 2 HCl(aq)-CaCl₂(aq) + H₂(g) when 0.400 g Ca…
A: Here we are required to find the Enthalpy for the reaction. Given Specific heat capacity C =4.15 J…
Q: Question 7 The rotation of an ¹H1271 molecule can be pictured as the orbital motion of an H atom at…
A:
Q: Heat of Solution (ΔHsoln) for NH4NO3 TRIAL 1 TRIAL 2 Mass of NH4NO3: (NH4NO3 = 80.05 g/mol):…
A: To determine: qcalorimeter(cal) moles NH4NO3 ΔHsoln Average ΔHsoln(NH4NO3) %error
Q: Mass of NaOH = SC Submit Part E Ascorbic acid, or vitamin C (C6H₂O6, molar mass= 176 g/mol), is a…
A: Given, E) The moles of ascorbic acid ( C6H8O6 ) = 2.81 × 10-4 mol. The mass of ascorbic acid is:…
Q: When Na3(PO4) and glucose are added to water solution(s) will conduct electricity. (K) both, both…
A: Na3( PO4) is a ionic compound and a strong electrolyte . Therefore when it is dissolved in water it…
Q: 3. Considering the techniques and instrumentation used to perform these experiments, identify 2…
A: A Calorimeter is a device that can be used to measure heat flow. it must be well insulated in order…
Q: A substance has a critical point of (456.49 K, 356 atm). It was heated to a temperature of 362.01°F…
A: •CRITICAL POINT:-Critical point is the highest value of temperature and pressure at which a…
Q: the concentration of Cl- present in a solution obtained by mixing 160.4mL of 0.25M HCl, 30.4mL of…
A:
Q: and Н CH3 ОН
A: We have to synthesized the following transformation where you utilise both reagents somewhere in the…
Q: One common way to determine Phosphorous in urine is to treat the sample after removing the protein,…
A: #(i): Calibration curve is plotted by taking concentration(ppm P) in X-axis and absorbance in…
Q: A mixture of nitrogen gas, hydrogen gas, and ammonia gas are allowed to come to equilibrium at…
A: The equilibrium partial pressures are: PN2 = 0.63 atm,PH2 = 1.5 atm, andPNH3 = 0.84 atm
Q: Which is the correct sequence, from left to right, to separate the following ions from each other,…
A:
Q: ▼ Part A Calculate the number of molecules in 6.00 moles H₂S. Express your answer numerically in…
A: The molecules of H2S in 6.00 moles is = 3.61 x 1024 H2S molecules
Q: Please help write the final product, reaction equation, table of physical properties and percent…
A: To write the final product, reaction equation, table of physical properties and percent yield for…
Q: The following catalytic cycle shows and example of what? 6 5 OC Rh Ph3P Ph P 0= MPPh 3 D H со H pph3…
A: We have to tell the given catalytic cycle to indicate what. The given options are Alkene…
Q: Provide the IUPAC name with clear explanation, as well as the retrosynthetic analysis and general…
A: Here we have to write the IUPAC name of the following given compound and it's synthesis from staring…
Q: What is the concentration of a NH₂I solution that was made by diluting 45 mL of a stock 12 molar…
A: Given data Concentration and volume of stock solutionC1=12 mol/LV1=45 mL=451000 L=0.045 L…
Give a clear handwritten answer with explanation....give the mechanism of given bleow reaction
Step by step
Solved in 2 steps with 2 images

- Mrs S has severe urticaria. The prescriber has requested 500g of 2% w/w calamine in aqueous cream. You have 5% w/w calamine in aqueous cream in stock. You decide to prepare this product by extemporaneous dilution using aqueous cream as the diluent. How many grams of 5% w/w calamine in aqueous cream is needed to make 500g of 2% w/w calamine in aqueous cream? units – g111.1Ms = ? ks show work, report sig figs and unitsA slow heat release pack is to be marketed based on mixing an anhydrous solid with just enough water to form one of its solid hydrates. Which will yield more heat, CaCl2 to CaCl2.6H2O or Na2SO4 to Na2SO4.10H2O? Assume that the same molar quantities of the anhydrous solid are used. You will have to look up the values of ∆H f,m . Sources must be cited appropriately with google
- Part: J,K.The heat capacity (Cp) 0f an hal0genated 0rganic c0p0und, chl0r0f0rm is given by the f0ll0w equati0n. Equati0n (1) Cp (J/mol K) = 19.8 +0.22 T – 0.00026 T2 (a) 0btain a new equati0n that uses T in Fahrenheit (b) Calculate the Cp 0f chl0r0f0rm at 300 K using Equation (1) (c) Calculate the Cp 0f chl0r0f0rm at 80 F using the equati0n you derived in (a)Computers are not supposed to be in very warm rooms. The highest temperature tolerated for maximum performance is 308 K. Express this temperature in C and F.
- What is the value of Kw at 25 ∘C?5. The literature value for the heat of fusion of ice is 6.02 kJ/mole. What is the percent error for the experimentally determined heat of fusion?Several ice cubes (ρi = 0.9167 g/cm3) of total volume Vi = 205 cm3 and temperature 273.15 K (0.000 °C) are put into a thermos containing Vt = 680 cm3 of tea at a temperature of 313.15 K, completely filling the thermos. The lid is then put on the thermos to close it. Assume that the density and the specific heat of the tea is the same as it is for fresh water (ρw = 1.00 g/cm3, c = 4186 J/kgK). a. Calculate the equilibrium temperature TE in K of the final mixture of tea and water. b. Calculate the magnitude of the total heat transferred QT in J from the tea to the ice cubes.
- H2SO3 = 0.2556 M, Ka1 = 1.6 x 10-2 , Ka2 = 6.4 x 10-8 NaOH = 0.3106 M please show all work all information needed is hereThe same student wanted to determine the heat of dissolution for potassium chlorate (KClO3, MW: 122.55 g/mol) in water. He measured 10 grams of KClO3 and dissolved it in a calorimeter containing 250 g of water. The temperature drop was 3.5 K. What is qH2O? What is qrxn? What is ΔHrxn?Given: F2(g) + 2 e- ⟶ 2 F-(aq) Eo = +2.87 v Hg2+(aq) + 2 e- ⟶ Hg(l) Eo = +0.87 v Calculate ΔGo for: Hg(l) + F2(g) ⟶ Hg2+(aq) + 2 F-(aq) ΔGo = -nFEo F = 96500



