O 65 degrees Fahrenhelt O None of the above QUESTION 29 In the "DNA Extraction from Fruit" laboratory, what solution disrupted the cell and nuclear membranes? O Methyl orange O Hydrogen peroxide O Tap water Detergent and a salt-based lysis solution Ice-cold ethanol
O 65 degrees Fahrenhelt O None of the above QUESTION 29 In the "DNA Extraction from Fruit" laboratory, what solution disrupted the cell and nuclear membranes? O Methyl orange O Hydrogen peroxide O Tap water Detergent and a salt-based lysis solution Ice-cold ethanol
Biology: The Unity and Diversity of Life (MindTap Course List)
15th Edition
ISBN:9781337408332
Author:Cecie Starr, Ralph Taggart, Christine Evers, Lisa Starr
Publisher:Cecie Starr, Ralph Taggart, Christine Evers, Lisa Starr
Chapter6: Where It Starts-photosynthesis
Section: Chapter Questions
Problem 12SQ: Which of the following substances does not participate in the CalvinBenson cycle? a. ATP b. NADPH c....
Related questions
Question
Please help me
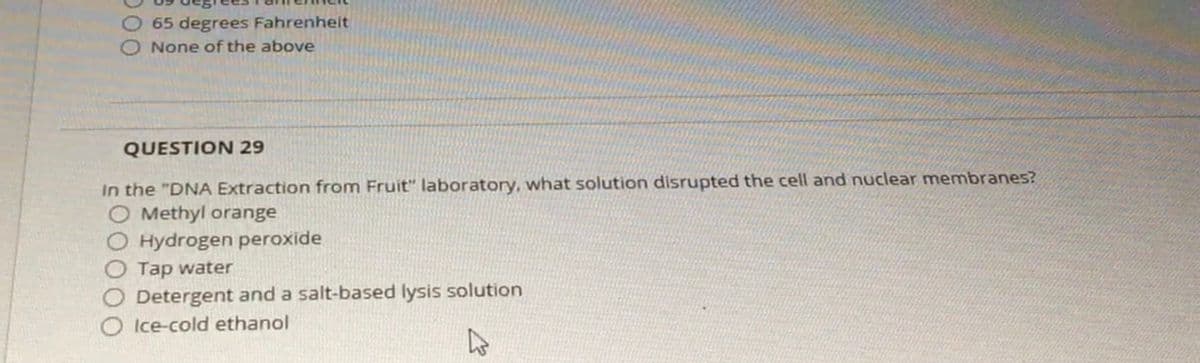
Transcribed Image Text:65 degrees Fahrenheit
O None of the above
QUESTION 29
In the "DNA Extraction from Fruit" laboratory, what solution disrupted the cell and nuclear membranes?
O Methyl orange
Hydrogen peroxide
Tap water
Detergent and a salt-based lysis solution
O Ice-cold ethanol
DOO
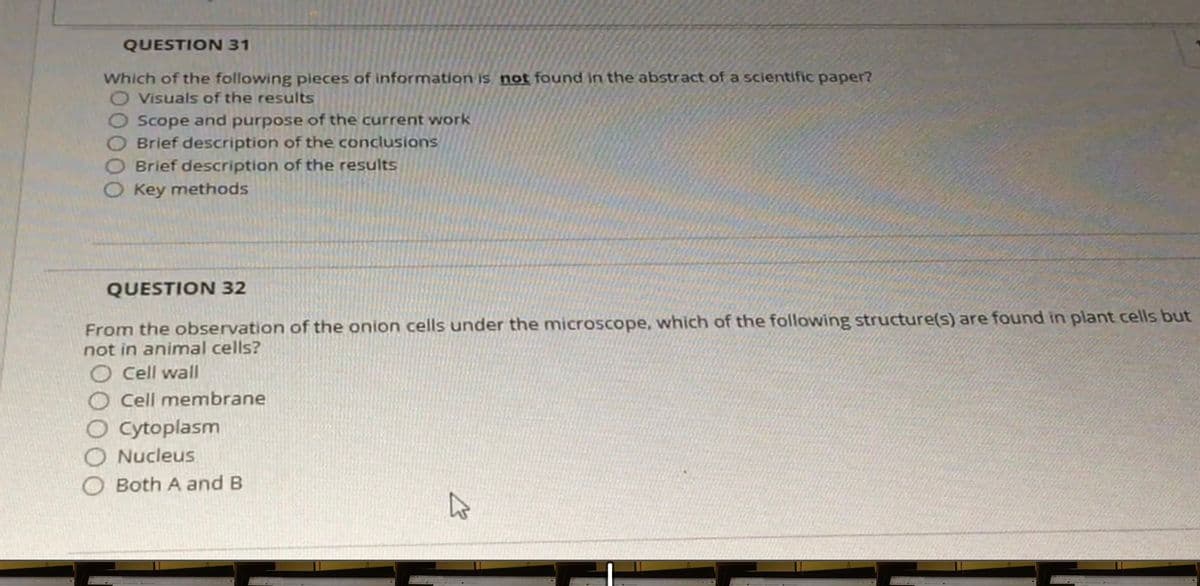
Transcribed Image Text:QUESTION 31
Which of the following pieces of information is not found in the abstract of a scientific paper?
O Visuals of the results
Scope and purpose of the current work
OBrief description of the conclusions
Brief description of the results
Key methods
QUESTION 32
From the observation of the onion cells under the microscope, which of the following structure(s) are found in plant cells but
not in animal cells?
O cell wall
Cell membrane
Cytoplasm
Nucleus
Both A andB
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Recommended textbooks for you

Biology: The Unity and Diversity of Life (MindTap…
Biology
ISBN:
9781337408332
Author:
Cecie Starr, Ralph Taggart, Christine Evers, Lisa Starr
Publisher:
Cengage Learning

Biology 2e
Biology
ISBN:
9781947172517
Author:
Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:
OpenStax

Anatomy & Physiology
Biology
ISBN:
9781938168130
Author:
Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Publisher:
OpenStax College

Biology: The Unity and Diversity of Life (MindTap…
Biology
ISBN:
9781337408332
Author:
Cecie Starr, Ralph Taggart, Christine Evers, Lisa Starr
Publisher:
Cengage Learning

Biology 2e
Biology
ISBN:
9781947172517
Author:
Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:
OpenStax

Anatomy & Physiology
Biology
ISBN:
9781938168130
Author:
Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Publisher:
OpenStax College