Four amino acid samples were subjected to a reverse-phase chromatography set up and yielded the following Rf values. Which among the samples is the most polar? Sample Rf values 0.49 0.53 0.21 0.55
Four amino acid samples were subjected to a reverse-phase chromatography set up and yielded the following Rf values. Which among the samples is the most polar? Sample Rf values 0.49 0.53 0.21 0.55
Chapter1: Relative Value, Addition, And Subtraction Of Decimals
Section: Chapter Questions
Problem 39SST
Related questions
Question
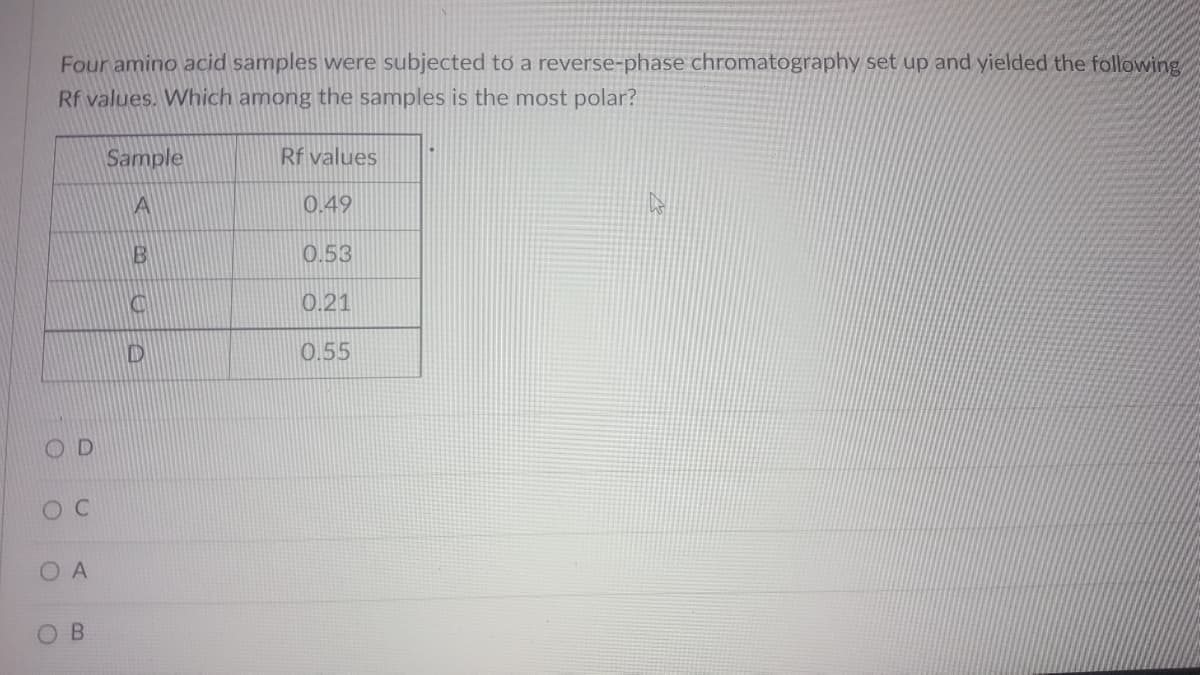
Transcribed Image Text:Four amino acid samples were subjected to a reverse-phase chromatography set up and yielded the following,
Rf values. Which among the samples is the most polar?
Sample
Rf values
A
0.49
0.53
0.21
0.55
O A
B.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps

Recommended textbooks for you



Principles Of Radiographic Imaging: An Art And A …
Health & Nutrition
ISBN:
9781337711067
Author:
Richard R. Carlton, Arlene M. Adler, Vesna Balac
Publisher:
Cengage Learning



Principles Of Radiographic Imaging: An Art And A …
Health & Nutrition
ISBN:
9781337711067
Author:
Richard R. Carlton, Arlene M. Adler, Vesna Balac
Publisher:
Cengage Learning


Human Biology (MindTap Course List)
Biology
ISBN:
9781305112100
Author:
Cecie Starr, Beverly McMillan
Publisher:
Cengage Learning
