Q: Niergen H3C HB ? not H. &+ Carhon HB? not H Hystrogen H-C=C-H HB P not :O: ... Thymine H. HB ? not…
A: This is the concept of Hydrogen bonding and basicity
Q: Compound mL MW d moles bp °C 2-methylcyclohexanol 8.5 85% phosphoric acid 2 product mixture…
A: Compound ml Mw d g moles bp degreeC 2-methyleyclohexanol 8.5 114g/mol 0.93 g/mL d=m/v m = 7.9…
Q: Zoom Day 9 lonic (). Molecular (M) or Acid (A) Formula ulfate Asz05 Hzsoz NHyoz (NHy), O entoxide M…
A: Molecular bond is formed by sharing electrons between two atoms Ionic bond is formed by the…
Q: 7. wnał iN the maximum number of itereoirome powribie for COch compound ? Q. CHg CH (애 )CH(OH)H』애9…
A:
Q: when 10.5 of HgOcss CMV 216.3 9g Imol is decomposedto Form Hg a) and o 2zcg) e Zcg) 8 the heat or…
A: Provided amount of HgO=10.5g Molar mass of HgO=216.39g/mol The reaction is 2HgO(s) → 2Hg(l) +O2(g)…
Q: Place these in order of most likely to proceed to completion (products), where 1 is least likely and…
A: For proceed the reaction in for direction. The equilibrium constant should be high. Higher the value…
Q: Identify A–J:
A: Benzene is the starting material in the reaction. Benzene undergoes electrophilic substitution…
Q: Calculate the ∆H of the following reaction. Show the complete and step by step solution. 1. 2C(s) +…
A:
Q: Draw the structures of compounds A–H in the following synthesis scheme.
A:
Q: Nibromethane undergoes combustion CH3 NO₂(1) + according to - - O2(g) A -CO₂(g) + H₂O(g) + - N₂cg- a…
A:
Q: What is the ΔH°rxn of this given reaction? Round your answers in 2 decimal places KOH + HNO3 →…
A: KOH+ HNO3 ->>KNO3+H2O
Q: i. H2N CH3CO2H, reflux j. 1) Sn, HCI 2) NaNO2, HCI O2N 3) k. 1) LIAIH, CN 0- 2) HO* - I. Br2, FeBr3
A: Major products are given below
Q: HO Cro3 Cro3 Ca) Product (d) Product OH H30 H30 HO РСС Cl (e) PCC b) Product Product OH
A: The structure of correspond to omitted structure for each of the given synthetic sequence can be…
Q: (a) HgCl2 • 2 MeMgCI (b) GaMes Me,NH heating (c) BH3 сно BrPh,P base (d)
A: Here the reactions are reaction of Grignard reagent with metal chloride Forming an adduct of ammonia…
Q: 2Hed znclzt H2 -우 3.6 molo 아 zimt Jicac+, tow many gramg of hydrochloric वतर्व वढर needcd o
A:
Q: Sucent a beaiser containine SS-0g has 100-0°C.How much heeit is neecled to convent t water t0.…
A: Given : Mass of water = = 55.0 g Temperature of water = 100 oC And ΔHvap i.e heat of vaporisation…
Q: Calculate the DH°rxn for the following reaction. SiO2(s) + 4HCl(g) ® SiCl4(g) + 2H2O(g) Given:…
A:
Q: Using the provided data table, calculate ΔrxnH for the following reactions: a) 2O3(g) ⇌ 3O2(g) b)…
A: The ΔHrxn for the given reaction can be calculated as follows: ∆Hrxn=∆Hf (products)-∆Hf (products)
Q: Use the following reagent table for the reaction of: cyclohexanol + H2 SO4 → cyclohexene Chemical MW…
A: Limiting reagent is that react completely in the reaction. In this reaction, 1 mol of cyclohexane…
Q: Griven domats of cyclohexame is o-7785glm and AHamb=-46.6KI|| Hole Many Jaurneddo vreleade ag mmuch…
A: Heat of combustion is the heat energy released when 1 mol of a substance is burnt.
Q: -Er a) H b) MgBr 1. 2. H3O+ ai (second step is just a workup) c) OH D3C- F3C CF3 • of you wholes CD3…
A: a. Aldehyde + alcohol in the presence of acid gives ketal. b. Grignard addition to keto FG. c.…
Q: CH3 ((i CH3-C=CH2CI Gilman rcagent ? BF, 1) LDA →? 2) CH31 -78 °C 1) LIAIH4 → ? 2) H30® Cliv)…
A:
Q: & 0 e) (1) excess PhMgBr (2) H₂O* f) -OCH3 CH₂OH CH, 1 g) PhCH₂-CH-CH₂-C-NH₂ 1. excess LIAID 4 2.…
A:
Q: а) CI Ph3Pl - PPH3 Rh PPH3 + H2 Ph3P ČO b) Mn2(CO)10 + HCl – > ? c) CH;I CO Co
A:
Q: OH H2CrO4 PHCHO E F heating NaOH MeSH H2CrO4 H HO excess H*/MEOH MeONa J
A: The details solution for this reaction is provided below in attach image.
Q: How do I calculate moles used in this lab from the information in the table?
A: The mass of nitric acid used is calculated below with the help of density equation in which m is the…
Q: EtO NaOEt, E1OH H NaOEt, E1OH EtO H NaOEt, E1OH N2OET, E1OH Eto I OEt
A: All reactions are condensation reactions. All involves common steps i) Generation of Carbanion ii)…
Q: 81) IL [0] CH3CH > A) OH CH3CHCH3 82) B) OCH3 Ni CH3CCH3 + H2 CH3 CH2CCH3 OH 83) Ni CH3CH H2 → C)…
A: Aldehydes undergoes oxidation to produce carboxylic acids. RCHO + [O] ------> RCOOH…
Q: ОН MnO2 HCI (h) NaCN A C H20 В HCI (Give all products (2 pts each) 1. 2 eq CH3M9BR (e) CI 2. H*
A: When alcohol is oxidized with mild oxidizing agent then it will form either aldehyde or ketone…
Q: Hg(OAc)2 NABH4, HO" TsCl, pyridine KCN CN (b) (a) of OTS (b) (a) 17C Hae AU
A: We have given the organic reaction and we have to find the major product of the reaction.
Q: COOME (1) MeOK 70. 0+ CH, ,HO00 (2) H,O®, a →Product is: COOME (a) HOOC- ОН (b) НООС- HO НОС (d)…
A:
Q: What are the products of the following reactions?
A: a)
Q: a) Water with 0=SeCl2 to form O=Se(OH)2 b) Water with O=SeCl2 to form SeO2 c) Methanol with POC13 to…
A: As you not specified so I am giving answer of first 3 subparts. First nucleophile attack on…
Q: OH Ph Ph NH2 TFA (+)-13 (10 mole%). NaBH4, CICO,CHз Меон, -50 to 0 "C CH;CN-H20 0 °C, 24 h quant.…
A:
Q: CH3CH2CH2CH2CI (1 mole) ? OMe AIClg, heat O Meo Meo Meo OMe O Meo OMe O There is no reaction under…
A: The reaction is the aromatic substitution reaction . The position of the entering group is directed…
Q: What is AH° for the following reaction? 2C2H2(g) + 502(g)4CO2(g) + 2H2O(1); C2H2(g) AH°F (kJ/mol)…
A:
Q: EXP 20 Report Sheet Heats of Formation Name Wet Lab: a.Is the reaction in the flask warm or cold? Is…
A: Soln:- Given reaction NH4Cl(s)---------> NH4+(aq) + Cl-(aq) Since NH4Cl is dissociating into ions…
Q: HOET Br
A: See product in red box.
Q: s. oluboM Oic pent 9. 5 ОН 10.
A: The naming is done on the basis of IUPAC naming rules.
Q: HO Name of this compoinat न+we १ळ्चंऋयु कन्ब कच्य चक is peimouy, seconday amal yeotiary
A:
Q: CI „NO2 1) 1 equiv NaOH 2) H3O* CI
A: Note: Since you have posted multiple independent questions in the same request, we will solve the…
Q: Find ΔHrxnΔHrxn for the following reaction: N2O(g)+NO2(g)→3NO(g)N2O(g)+NO2(g)→3NO(g) Use the…
A: If A+B →C+ D ∆H=xthen C+ D →A+ B ∆H=-xand2( A+B →C+D ) ∆H=2x
Q: K
A:
Q: с. Pivo 1. (MeO)2PCH2N2, KO-BU, THF, –78 °C TESO** 2a. DIBAL-H (2.1 eq), CH2CI2, –78 °C 2b.…
A: Mechanism of the organic reaction is given below
Q: Would this be a correct synthesis?
A: The given conversion can be done as follows :Firstly, the reaction of the reactant with Br2 in the…
Q: i) H. .CO2H 150°C ? + H20 90% H `CO,H Maleic acid ii) (CH;CO),O + (CH3)½C-OH ÇOOCH3 i11) + OH NAOH ?…
A:
Q: 12.2 g of 2-methylbutan-2-ol (MW 88.15 g/mol) reacts with excess H2SO4 to form 7.98 g of…
A: Given data,Mass of 2-methylbutan-2-ol=12.2 gMolecular weight of 2-methylbutan-2-ol=88.15 g/molMass…
Q: Draw the organic products formed in each reaction.
A: As per the Bartleby guidelines, I am answering the first three sub-parts only. Please resubmit the…
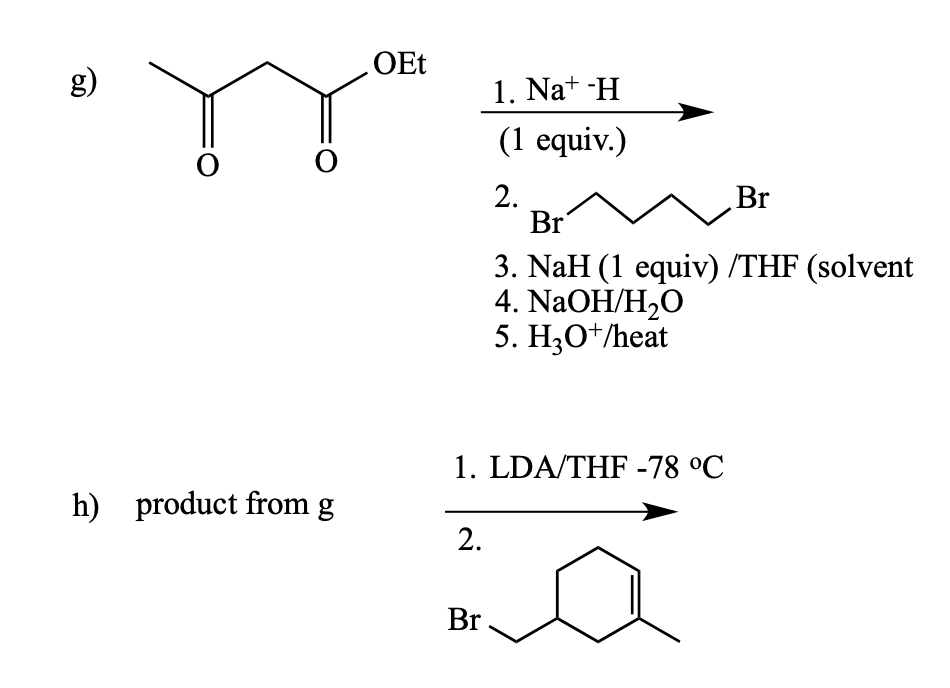
Give product
Step by step
Solved in 2 steps with 1 images

- Give two reasons and explanations why cyclohexane, m.p=25 c and b.p= 161 c would not be a suitable solvent for recrystallization.What are the volume (uL), moles, and/or weight (mg) of each reagent and solvent in:50 mg of the “Bromo” starting material (1 equivalent), acid catalyst (if any, 0.1 eq) and in 0.25 M of solvent?why is numbering of reagents important?
- What is the limiting reagent, and their ratios to each other? ( example: 2:3:1) Bromobenzene: 4.5mL Magnesium:1.0g Methyl benzoate:2.5mL Product: 1.82gConvert 0.3 mM p-nitrophenol to µmol for the following cuvette: Cuvette 1 2 0.3 mM p-nitrophenol (ml) 0.05 0.10In the beginning of this video https://www.youtube.com/watch?v=9Ng6Zv9oLzk, what reason is given on why the dienophile likes the diene?
- mmol of 1,3-dibenzoylpropane used to 4 sig. fig Used: 405 mg molecular weight: 252.31 g/molAssuming a 56.8% yield, how kany ml of alkene is required to produce 22.5 mL of 2,3-dimethyl-2-butanol? MW of alkene: 84.16, d: 0.653 MW of 2,3-dimethy-2-butanol: 102.17, d:0.823Reagent 5 options; H2O H+CrO3 H2SO4 H2O CH3CH2Br NaBH4
- For 20.7 the solutions for part c and d dont seem right. Shouldn't reacting with 1 eq of H2 just take away the C=C bond and not the C=O bond. (H2 reduces C=C selectively to form a ketone according to page 738. Also the both the C=C and C=O bond should be reduced if excess H2 is used6. Identify the best reagent(s) for this reaction. (see attached screenshot). a. H2SO4, HgSO4, H2O b. 1. Disiamylborane, 2. HO–, H2O2 c. K2Cr2O7, H+ d. NaOCl e. H2, Pd1. Alcohols X and Y formed layers with Lucas reagent. Compound X formed brown precipitate with KMnO4 while compound Y did not. Which are the possible identities of compounds X and Y? a.tert-butanol, propanol b.isopropanol, isopentyl alcoholc. methanol, ethanold.tert-butanol, isopropanol2. Alcohols that cannot be distinguished using the Lucas test? a.isopropanol and methanol b.t-butyl alcohol and ethanol c.t-butyl alcohol and 2-methylbenzyl alcohol d.ethanol and n-propanol

