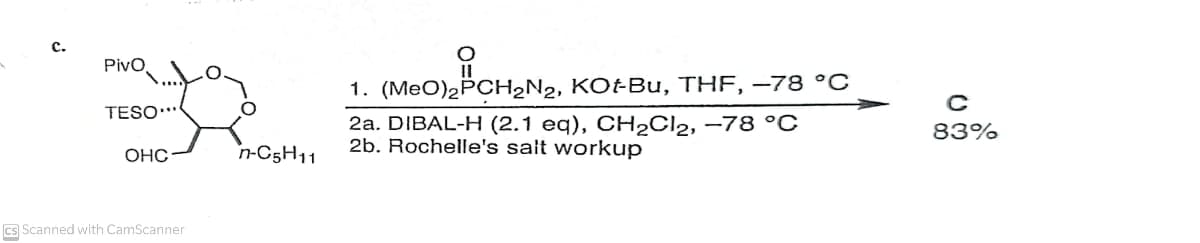
с. Pivo 1. (MeO)2PCH2N2, KO-BU, THF, –78 °C TESO** 2a. DIBAL-H (2.1 eq), CH2CI2, –78 °C 2b. Rochelle's salt workup 83% OHC n-C5H11 Cs Scanned with CamScanner
Q: Describe the preparation of a) 500 ml of 0.0550 M silver nitrate solution from solid silver nitrate…
A: A numerical problem based on concentration terms that is to be accomplished.
Q: Use thermodynamic table below to find out if the solubility product of fluorite is lårger 100°C than…
A: The value of solubility product of CaF2 is needed to be determined at 100°C and at 25°C. Given:…
Q: 2. In a laboratory experiment you react 20.0 mL of 25% by mass C2H&N2 with 8.000 NiCl2.6H2O to…
A: The balanced chemical equation can be written as follows: NiH2O6Cl2 + 3C2H8N2 → NiC2H8N23Cl2 +…
Q: A Pb-Sn alloy contains 10% primary a and 90% eutectic microconstituent immediately after the…
A: A Pb-Sn alloy contains 10% primary ἀ and 90% eutectic micro constituent immediately after the…
Q: The Ksp values of two solid carbonates, A2CO3 and BCO3, were determined by preparing 250.0-mL…
A: Sparingly soluble salts are those that do not dissolve completely in solution rather maintain…
Q: The weight of 5 ml of vinegar sample was 5.228 g, was required 30.5 ml of 0.2 M sodium hydroxide…
A: Acid reacts with base to form salt and water and this reaction is known as neutralization reaction.…
Q: Cr2072- + Fe2+ Cr3+ + Fe3- quation for alkaline solution:
A: Dear student I have given answer to your question in the image format.
Q: What weight of pyrite ore (impure FeS,) must be taken for analysis so that the BaSO, precipitate…
A: The mass of pyrite ore must be used for analysis is determined as,
Q: 4. Calculate the molar concentration of a 20.0% solution (w/w) of KCI that has a specific gravity of…
A: Number of moles is calculated by dividing the given mass with molecular mass.
Q: Six iron tablets containing FeSO4.7H2O were dissolved in 100-ml of 0.1M HNO3 with gentle heating.…
A: Given : Weight of Fe2O3.xH2O = 0.345 gm Weight loss on heating = 10.5 %
Q: Q2- A 30.0 mL H2C2O4 acidified solution, was treated with 25.0 mL of 0.102 M KMNO4 solution. The…
A:
Q: Solution: Kso=1.41-10-11 The resistance of a saturated solution of PbSO4 (very insoluble salt) at…
A:
Q: Calculate the saturation indices of the solutions describe below and classify each as…
A: The solution can be classified as under-saturated, supersaturate or in equilibrium as per the below…
Q: Examples Example (1);- The following data obtained from decomposit of H2 02 in aquees Solution:…
A: Please find the graph attached here with the solution .
Q: stion 2 24 I -----VIroxide ir - urter 2.5. 9.2 g of lithium peroxide were dissolved in 90.8 g of…
A:
Q: HWPrepare one liter of 0.IN solution of H2SO4 concentrated if density is 1.84 and percentage is…
A: Normality of the solution is the number of gram Equivalents of Solute present in one liter of the…
Q: Explain the feature of homogeneous products I need only two features !
A: Homogeneous products are the products which consists of particles having same size, quality, color,…
Q: D. None of the above 11. A 315mL of water was added to 2100 mL of 19M NaCI solution. What is the new…
A:
Q: 0.105g of the K3[Fe(C2O4)3] (MW= 437.2 g/mol) were dissolved in sulfuric acid and got rated with 9…
A: Balanced Reaction 5 C2O42- + 2 MnO4-+16 H+ →2Mn2+ +10CO2 + 8 H2O So, from this we can determine the.…
Q: Previous research on a soil like yours measured an isotherm for the sorption of P by the soil. The…
A: Adsorption refers to the higher concentration of a molecular species at the surface of a solid or…
Q: 1. Calculate the Ksp of the following aqueous solutions a) 3.60x10-5M Ag2CO3 b) 4.30x10-3M of Ag+…
A: We have to calculate the Ksp.
Q: The weight of 0.1012 g of magnesium sulphate consumed 14.5 mL of EDTA solution (0.05M). What is the…
A: Given data,Mass of Magnesium sulphate=0.1012gMolarity of EDTA=0.05MVolume of EDTA=14.5mL
Q: QI / Prepare one liter of 0.1N solution of H,SO, concentrated if density is 1.84 and percentage is…
A: Normality of Preparation of H2SO4.
Q: CuCl2 + AgNO3 → + No new data to save. Last checked at 4:58pm. Submi
A:
Q: A mixt. of 46.32 of pure NaOH ir dinsolved in water and diluted to 1000mL. what vol- of 1-022 N HCA…
A: SOLUTION: Step 1: Neutralization is the process where H+ and OH- react to form water therefore…
Q: If a pharmacist fortified 10 g of by adding 12.5 g of an ointment containing 0.03% w/w of the same…
A: using the formula to calculate
Q: 2. The solubility of Fe(OH)2is 3.00 x 10-3g in 2.00 liters at 18ºC. What is its Ksp at 18°C? (MW of…
A:
Q: Solution for 0.105g of the K3[Fe(C2O4)3] (MW= 437.2 g/ mol) were dissolved in sulfuric acid and got…
A: In this question determine amount of oxalate by makin equivalents of oxalate and KMnO4 equal.
Q: An exhausted zeolite softener was regenerated by passing 100 litres, of NaCl. Solution containing…
A:
Q: Lactated Ringer’s/5% Dextrose solution contains: 6 g/L of Sodium Chloride (NaCl MW 58.5) 3.1 g/L of…
A: Each KCl forma 1 potassium ion, K+. Hence mEq/mL of potassium ion is the same as mEq/mL of KCl…
Q: How much Fe3+ from ferrihydrite would dissolve at pH 2 and 7? How much Fe3+ from goethite would…
A:
Q: Chemistry Calculate Mn Mw Mz n1 100 80 70 50 30 M1 50 40 30 20 10 Example 2. Calculate Mn, Mw for…
A: The number average molecular weight can expressed as: Mn=∑niMi∑ni where, n is the number of…
Q: If to a solution of NaOH, in water and ethanol (20 ° C) 3 mmol of A (106.11 g / mol) and 2 mmol of B…
A:
Q: Extraction of DNA involves the use of a 1 M phosphate buffer (1-liter volume) having the hydrogen…
A: Given: Hydrogen ion concentration of buffer = 6.165950019 x 10-9 M Molarity of buffer used = 1 M…
Q: The solubility of PbF2 at 25°C is 0.64 dm³. Calculate Ksp of PbF2. (Moza mass of PbF₂ =245.2gmol¹)…
A: Given: Solubility of PbF2 = 0.64 gdm-3 Molar mass of PbF2 = 245.2 gmol-1
Q: Calculate the molar concentration of H3PO4 commercial reagent that is 86% (w/w) and has a specific…
A:
Q: f to a solution of NaOH, in water and ethanol (20 ° C) 3 mmol of A (106.11 g / mol) and 2 mmol of B…
A: Given reaction is 2A + B ⟶ C It is given that: Amount of A = 3 mmol Amount of B = 2 mmol Amount of…
Q: 2. A 0.05 M solution of sucrose (C12H22011) is isotonic to the saturated solution of PbCl2 at 30°C.…
A: Given: Concentration of sucrose solution = 0.05 M Temperature = 30°C
Q: To prepare orange chrome dissolve 2g of lead acetate in 25cc of Warm distilled water, add 4cc oF 2…
A: Chrome orange is a mixed oxide with the chemical formula, Pb2CrO5.
Q: How many milliliters of 1.5% alcoholic dimethylglyoxime should be used to provide a 36.0% excess…
A:
Q: synthesis of Tris(ethylenediamine)nickel(1I) chloride dihydrate The equations of the preparation of…
A:
Q: The conductivity of a saturated solution of SRSO4 at 298 K is found to be 1.482 x 104 S cm', while…
A: Given data : Conductivity of SrSO4(κ)=1.482×10-4Scm-1λ∞m(Sr2+)=118.92 Scm2mol-1λ∞m(SO42-)=159.6…
Q: The researcher extracts a sample of well water and prepares it for TXRF analysis with a 10.00 mg/L V…
A: As per the given data, TXRF = 10.00 mg/L Calibration coefficient = 69297.81
Q: A sample of feldspar weighing 1.500g is decomposed, and eventually there is obtained a mixture of…
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- Example: Calculation the weight of barium iodate Ba(IO3)2 if it dissolved in 500mL of distilled water (D.W) at 25 °C if M.wt of ppt = 487 g/mole and %3D Ksp= 1.57x10.To verify Beer’s Law for solution of KMnO4 or K2Cr2O7 using colorimeter.2. Calculate the molar concentration of the nitric acid if you know that specific gravity is 1.42 and the percentage is 70% to prepare. Solution has concentrated of 0.4 M in 100ml of distilled water
- The Henry’s law constant for CO2 in water at 25 °C is3.1x 10-2 M atm-1. (a) What is the solubility of CO2 inwater at this temperature if the solution is in contact withair at normal atmospheric pressure? (b) Assume that all ofthis CO2 is in the form of H2CO3 produced by the reactionbetween CO2 and H2O:CO2(aq) + H2O(l)------>H2CO3(aq)What is the pH of this solution?* A 1.20 g sample of unknown compound is disolved in 50 g of CHe. The solution freezes at 4.92 C. Calculate the MW. (f.p of CH6= 5.48 C, K; of CHo=5.12 C/m)Desalination is one possible process of extracting water from salty water ( from sea ) for industrial & domestic use. Briefly discuss and illustrate how to do it psychrometrically. attach the illustration pls
- Adrenaline is the hormone that triggers the release of extraglucose molecules in times of stress or emergency. A solutionof 0.64 g of adrenaline in 36.0 g of CCl4 elevates theboiling point by 0.49 °C. Calculate the approximate molarmass of adrenaline from this data.Calculate the osmolarity in mOsm/L of 500 mL of a solution containing 5% w/v dextrose, 0.78% w/v sodium chloride, and 27mEq of potassium acetate. dextrose: MW 180, # particles on dissociation 1 sodium chloride: MW 58.5, # particles on dissociation 2, valence 1 potassium acetate: MW 98, # particles on dissociation 2, valence 1What are the respective concentrations (M) of Cu2+ and Cl- afforded by dissolving 0.637mol0.637mol CuCl2 in water and diluting to 289mL?289mL? What are the respective concentrations (M) of Cu2+ and Cl- afforded by dissolving CuCl2 in water and diluting to 0.454 and 0.092 2.20 and 2.20 2.20 and 4.41 0.00220 and 0.00220 0.00220 and 2.20
- You are handed a 15.85% by mass aqueous KCl (m.m. = 74.5513 g mol–1) solution (at 25 °C). Assume the density of the solution is that of pure water (dsolution = 1.000 g mL–1). The ebullioscopic constant (Kb) for water is 0.513 °C m–1. The cryoscopic constant (Kf) for water is 1.86 °C m–1. The vapor pressure of pure water is 0.0313 atm. Determine the following: Vapor pressure of the solution in atm279.28 mL of 1.22 M organic salt was mixed with 168.14 mL of Ethanol (d= 0.789 g/mL, MW 46.07g/mole). Pure ethanol has a vapor pressure of 0.059 atm what will be the vapor pressure of the solution?For the following, remember that the boiling point elevation constant for water is Kb = 0.512 C/kg, and thefreezing point depression constant for water is Kf = 1.86 C/kg 50.0 g of table salt (Sodium Chloride) is dissolved into 500.0 mL of water. At what temperature will thesolution boil?

