ou may want to reference Pages 306 - 308) Section 9.5 while completing this roblem. Part A etermine the volume, in milliliters, required to repare each of the following diluted solutions. 23.0 mL of a 0.250 M KNO3 solution from a 6.50 M KNO3 solution Express your answer with the appropriate units. µA ? Value Units Submit Request Answer Part B 28.0 mL of a 2.50 M H2SO4 solution from a(n) 12.4 M H2SO4 solution Express your answer with the appropriate units. HA ? Value Units
ou may want to reference Pages 306 - 308) Section 9.5 while completing this roblem. Part A etermine the volume, in milliliters, required to repare each of the following diluted solutions. 23.0 mL of a 0.250 M KNO3 solution from a 6.50 M KNO3 solution Express your answer with the appropriate units. µA ? Value Units Submit Request Answer Part B 28.0 mL of a 2.50 M H2SO4 solution from a(n) 12.4 M H2SO4 solution Express your answer with the appropriate units. HA ? Value Units
Chemistry: Matter and Change
1st Edition
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Chapter14: Mixtures And Solutions
Section: Chapter Questions
Problem 75A: Stock solutions of HCl with various molarities are frequentlyprepared. Complete Table 14.7 by...
Related questions
Question
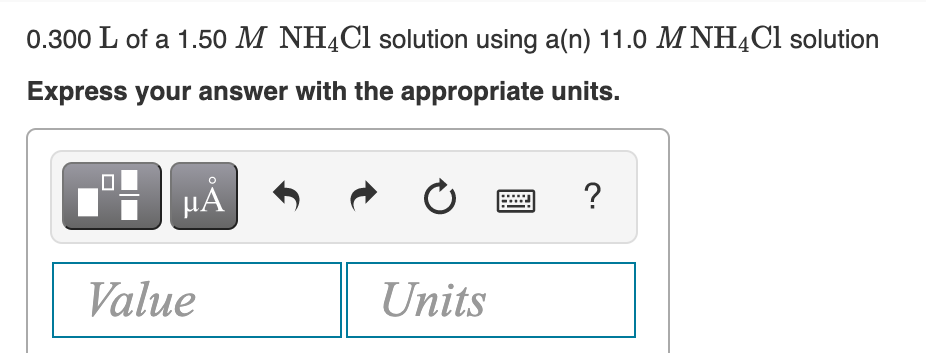
Transcribed Image Text:0.300 L of a 1.50 M NH¼C1 solution using a(n) 11.0 M NH4CI solution
Express your answer with the appropriate units.
HẢ
?
Value
Units
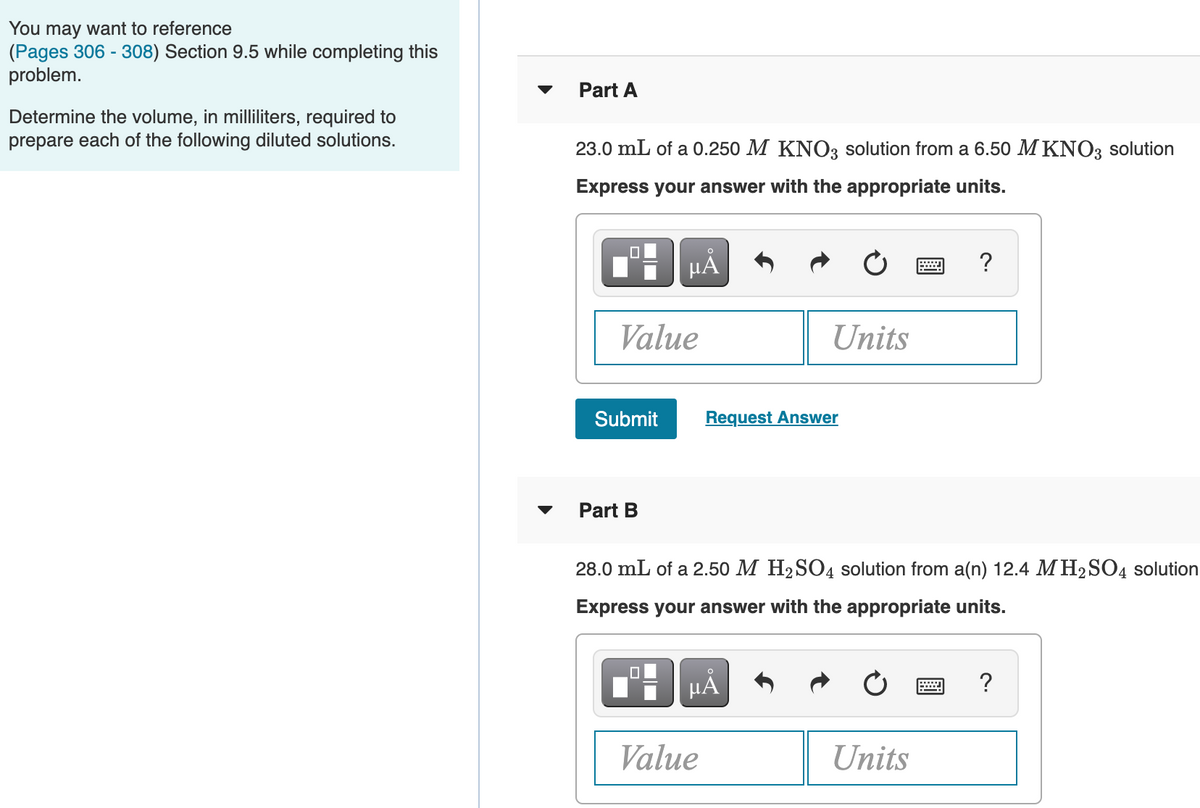
Transcribed Image Text:You may want to reference
(Pages 306 - 308) Section 9.5 while completing this
problem.
Part A
Determine the volume, in milliliters, required to
prepare each of the following diluted solutions.
23.0 mL of a 0.250 M KNO3 solution from a 6.50 M KNO3 solution
Express your answer with the appropriate units.
HÁ
Value
Units
Submit
Request Answer
Part B
28.0 mL of a 2.50 M H2SO4 solution from a(n) 12.4 M H2SO4 solution
Express your answer with the appropriate units.
HA
?
Value
Units
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning



Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning