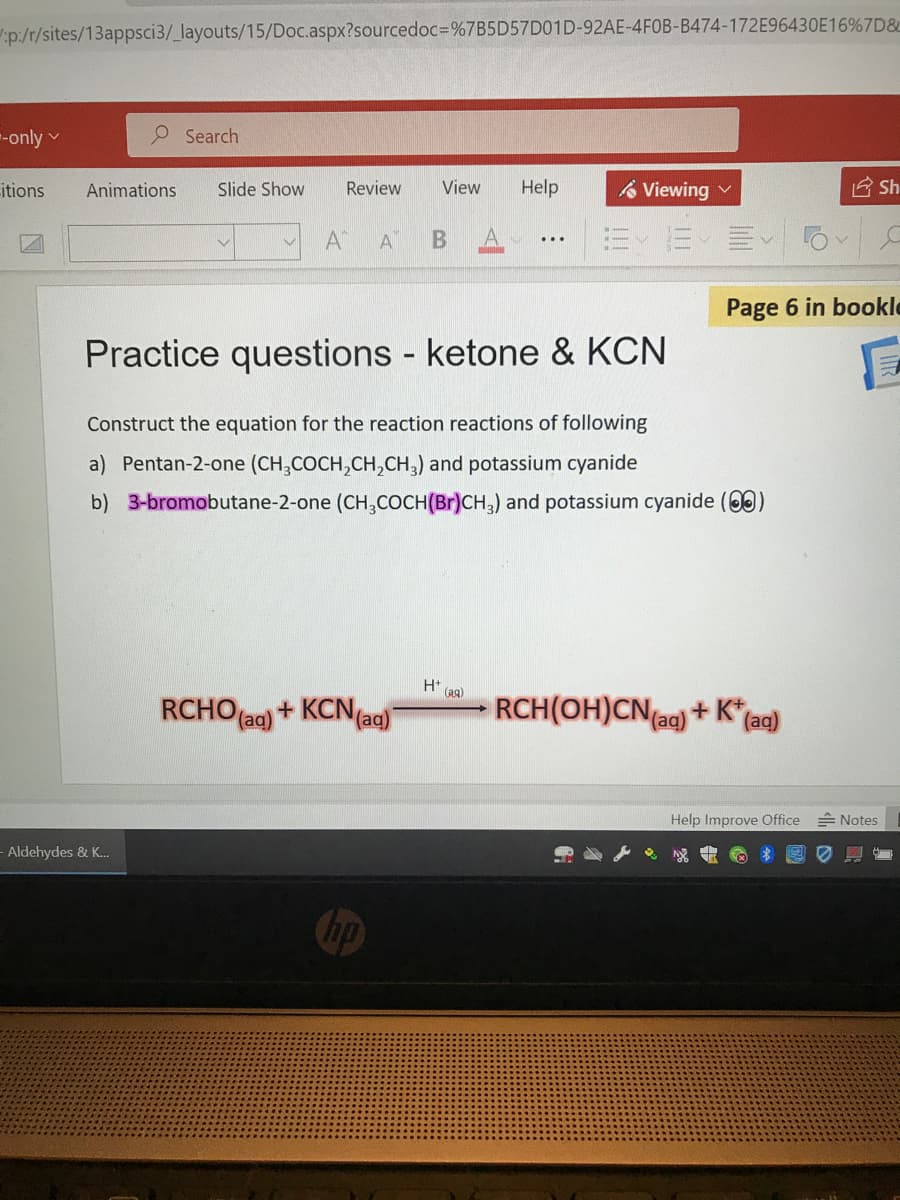
Page Practice questions ketone & KCN Construct the equation for the reaction reactions of following a) Pentan-2-one (CH,COCH,CH,CH,) and potassium cyanide b) 3-bromobutane-2-one (CH,COCH(Br)CH,) and potassium cyanide (0
Page Practice questions ketone & KCN Construct the equation for the reaction reactions of following a) Pentan-2-one (CH,COCH,CH,CH,) and potassium cyanide b) 3-bromobutane-2-one (CH,COCH(Br)CH,) and potassium cyanide (0
Chapter7: Statistical Data Treatment And Evaluation
Section: Chapter Questions
Problem 7.10QAP
Related questions
Question
Transcribed Image Text::p:/r/sites/13appsci3/_layouts/15/Doc.aspx?sourcedoc%3%7B5D57D01D-92AE-4FOB-B474-172E96430E16%7D&
-only v
Search
itions
Animations
Review
View
Help
A Viewing v
台 Sh
Slide Show
A
AT
Page 6 in bookle
Practice questions ketone & KCN
Construct the equation for the reaction reactions of following
a) Pentan-2-one (CH,COCH,CH,CH,) and potassium cyanide
b) 3-bromobutane-2-one (CH,COCH(Br)CH,) and potassium cyanide (00)
H (aa)
RCHO ag) + KCN(aq)
RCH(OH)CN(a) + K" (ac)
(aq)
Help Improve Office Notes
Aldehydes & K.
Chp
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

