Q: Which of the following sugars is least stable? А. B. OH HO Е. сновн он CH SH OH с. нон с НО. НОН.С с…
A: We have been given 5 structures of carbohydrates and we are asked to predict which of these 5…
Q: 35. Write the complete ionic and net ionic equations: NaOH + Al(NO)3 -> Na(NO3) + Al(OH)3
A: Given,The reaction :NaOH + Al(NO3)3 → Na(NO3) + Al(OH)3 Note: There is a subscript 3 in Al(NO3)3…
Q: Use this relation to calculate Keq for the following redox reaction that occurs in an…
A: Given reaction Cu2+(aq)+Zn(s)---> Cu(s)+Zn2+(aq) ECu2+/Cu = 0.337 V…
Q: Identify the optimum C-C-C bond angle for each of the indicated carbon atoms in the molecule. CH3…
A: we have to determine the bond angle between the C atoms for the given structure
Q: Magnesium oxide can be made by heating magnesium metal in the presence of the oxygen. The balanced…
A:
Q: A sample of ore containing manganese weighed 1.2354 grams. This sample was heated in a solution of…
A:
Q: ron(II) sulfide reacts with hydrochloric acid according to the reaction…
A: Limiting reactant :- A reactant which is present in lesser number of moles than required as per…
Q: Incorrect Separation Scheme بلند سلوک aq tie OH OH 6M HCl, vac. filtration aq NaCl ether, 10% NaHCO3…
A: In the given scheme there are two organic compounds of which one is acid and the other is neutral.…
Q: 13. Fill in the necessary data in the following table: Indicator Methyl Orange Bromothymol Blue…
A: Here's the completed table with the requested information: Indicator | pH Range | Color Change…
Q: In Chemical Kinetics, Why is the reaction rate independent of the amount of Zn used in the…
A: In the given question we have been provided with the information that Rate of a particular…
Q: oman's Interacti X 68U9zhHgbWASuJSIN7n6mmMh30WJVXTzxEOQhl/edit tensions Help S 100 120 Spanish 24…
A: 4 questions based on nuclear reactions. Each of them are to be suitably answered.
Q: 2- Equivalent conductance of 1 M propanoic acid CH3CH2COOH is 10 ohm¹ cm² eq' and that at infinite…
A: The degree of dissociation formula in terms of equivalent conductance is : α= ∧eq∧eq∞ where α is…
Q: Chemical species by reducing power
A: The reducing and oxidizing tendency of elements can be determined using…
Q: Nitrogen has 5 valence electrons. What types of bonds would it form?
A: We have to find out the type of bonds formed by Nitrogen which has 5 five valence electrons.
Q: a) Derive R35 b) Derive 300
A:
Q: How does trebling the concentration of HCl affect the reaction rate in chemical kinetics?
A: In chemical kinetics, the reaction rate is influenced by the concentration of the reactants.…
Q: 20) How many a-cleavages are possible for this molecule? 'N
A: We have find out the answer
Q: he structure and formula for these 1-(1,3 propane diammine)Cd(CN)2) 2-(iso-Propane)2diamine…
A: 1) 1) ,1,3-Diaminopropane is a three carbon diamine, which has a great many modern applications…
Q: 200-10. Balance the following oxidation-reduction S8 + NO3- equation. The reaction occurs in a basic…
A: To balance the oxidation-reduction equation in a basic solution, we need to follow these steps:Split…
Q: 1. Predict all of the products and mechanisms for the following reactions. a) CH3SH
A: The substitution reaction is the reaction where one functional group is replaced by another…
Q: proposed Lewis structure N-0- : 0: 0=0= CI: If two or more : 0: [H=C=C:]¯ atome WIS structures are…
A: • Lewis structure are those diagrams which represents the available valance electrons of atom in…
Q: 67. Which isoelectronic ion is the smallest in size? (A) A1³+ (B) Na+ (C) F- (D) 68. Which of these…
A: Isoelectronic species are those which contain same number of electrons. Cations are formed by the…
Q: Which mechanism do the following nucleophiles/bases favor? хон OH SH Drag answer here Drag ans ver…
A: Tertiary alcohols being bulky enough favour both E1 & SN1 by virtue of their stable tertiary…
Q: Balance the following chemical equation. Na2S4O6 + H₂O2 → Na2SO4 + H₂O + H₂SO4
A: Balanced chemical equation can be define as a reaction in which number of atoms of all the elements…
Q: Identify the class type of compound shown: HCIO3 O Weak Base Strong Base O Weak Acid Strong Acid
A: The basic definition of an acid is it is a species that can easily donate H+ . And a base is a…
Q: Compare the structures of the fatty acids A and B below. Based on your conceptual understanding of…
A: We have find out the answer.
Q: For the following reaction, 25.4 grams of diphosphorus pentoxide are allowed to react with 13.6…
A: diphosphorus pentoxide s + water l → phosphoric acid aq Mass of diphosphorus pentoxide P2O5 = 25.4 g…
Q: Give only typing answer with explanation and conclusion 1) A buffer solution contains 0.308 M acetic…
A:
Q: For the molecule shown below, provide the names for all of the indicated functional groups: 1.…
A: Here some…
Q: % Transmittance (a.u) 100 80 60 40 20 0 4000 r Pure anhydrous CAF 3500 3000 2500 2000 Wavenumber…
A: The question is based on the concept of IR spectroscopy. we need to assign characteristic bands for…
Q: r the following reaction, 37.3 grams of sulfuric acid are allowed to react with 42.5 grams of zinc…
A: Zn(OH)2 + H2SO4→ ZnSO4 + H2OZinc hydroxide…
Q: Draw a diagram showing all possible hydrogen bonding interactions between molecules of H2O and…
A: A hydrogen bond is a kind of intermolecular force of attraction between the partial positive charge…
Q: In the gravimetric analysis of iron, hydroxide may be added to a solution containing Fe3+ to…
A:
Q: QUESTION 9 The solubility of potassium chloride is 36 g/100 g water at 25 °C. Suppose that a…
A: Saturated solution has the right amount of solute dissolved in it such that no more solute can be…
Q: 29. What happens when gasoline is burned in an automobile engine? A. potential energy is transformed…
A: According to the first law of thermodynamics, the energy of the universe remains constant. For a…
Q: If cell potential of the cell Mg/ Mg2+ (0.01 M) // HCl(XM) / H2(g. 1.0 atm) / Pt, 2.09 v at 25 °C,…
A:
Q: Part C In the PhET simulation, make sure that the checkbox Stable/Unstable in the bottom right is…
A: We have find out the answer.
Q: I need help with two compounds that are some kind of isomers, the ones i picked are the optical…
A: tris-ethylenediaminecobalt(III) chlorideDeriving the structure:It has three ethylenediamine groups,…
Q: rank the following 0.1 M solutions in order of increasing pH. a. HI, HF, NaF, NaI b. NH4Br, HBr,…
A: The strength of the acids and bases present must be considered when comparing the pH of the given…
Q: (a) Using Lewis symbols, make a sketch of the reaction between potassium and bromine atoms to give…
A: Lewis dot structures are the structures of atoms or molecules that have valence electrons of all…
Q: The mixture consists of 14.50 g ethane and 310.0 g of propane. What is the total vapor pressure at…
A: Any volatile liquid always is in equilibrium with its vapour phase. At a particular temperature the…
Q: i. Which reaction conditions will best give the following transformation? OH H₂O, H* b) 1) OsO4 2)…
A: Alkenes on acidic hydration form alcohol through Carbocation intermediate. Alkenes on reaction with…
Q: 1. a. Provide the molecular formula for the following ester. a b b. On the structure, clearly…
A: A question based on introduction to organic chemistry. 6 subdivisions are given that are to be…
Q: In the UV/Vis spectroscopy the most appropriate wavelength for analysis is that where the is…
A: UV/Vis spectroscopy, the absorption of light by a sample is measured as a function of wavelength.…
Q: Write the chemical equation for the formation of acrolein from glycerol and potassium bisulfate.
A: Glycerol is dehydrated under acidic conditions in the chemical equation for the synthesis of…
Q: In the equilibrium-constant expression, product and reactant concentrations are raised to an…
A:
Q: Describe the differences between the infrared spectra of butanoic acid and 2-butanone
A: Butanoic acid and 2-butanone are two different compounds with distinct molecular structures, and as…
Q: Which of the following structures corresponds to the mass spectrum shown below? 100- Relative…
A: The mass spectra is used to determine the structure of a compound based on m/z ratio and peak arises…
Q: Balance the following chemical equation. Na2S4O6 + H₂O2 → Na2SO4 + H₂O + H₂SO4
A: A balanced equation is an equation for a chemical reaction in which the…
Q: Calculate the ratio of moles of H2O to moles of anhydrous KAl(SO4)2. Show all work including units.…
A: By determining the moles of ratio of two substance in any unknown compound, we can determine the…
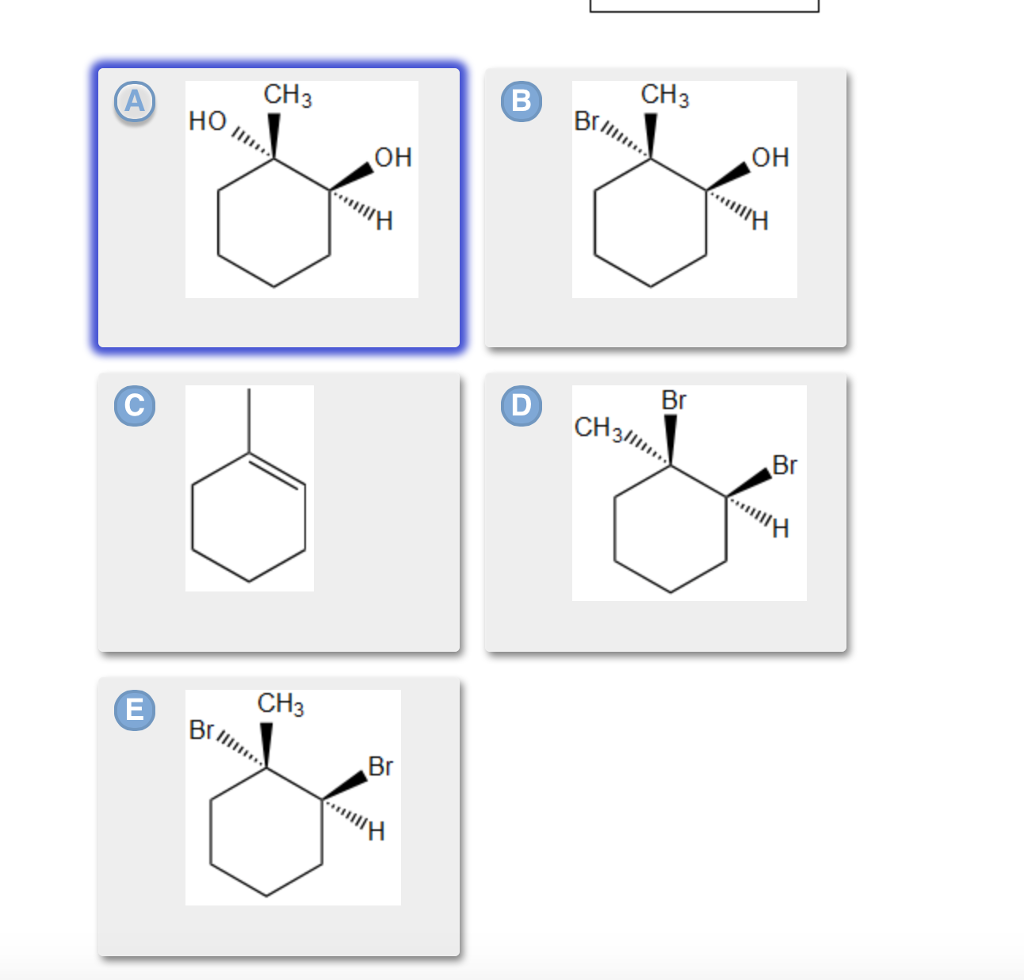
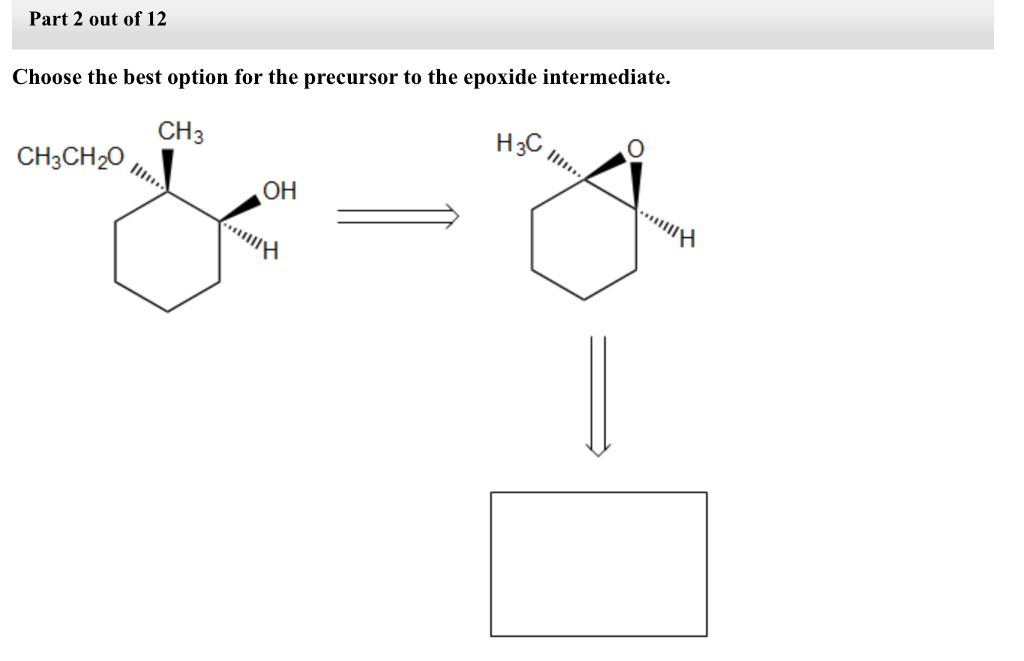
Step by step
Solved in 3 steps with 2 images


