Part A One of the following compounds is responsible for the IR spectrum shown. Choose the structure of the responsible compound. 25 26 27 28 29 3.5 Wavelength (um) 4.5 5.5 10 12 13 14 15 2600 2400 2200 2000 1600 1400 1200 600 1000 4000 330 3600 3400 3200 3000 Wavenumber (cm)
Part A One of the following compounds is responsible for the IR spectrum shown. Choose the structure of the responsible compound. 25 26 27 28 29 3.5 Wavelength (um) 4.5 5.5 10 12 13 14 15 2600 2400 2200 2000 1600 1400 1200 600 1000 4000 330 3600 3400 3200 3000 Wavenumber (cm)
Chapter12: Structure Determination: Mass Spectrometry And Infrared Spectroscopy
Section12.SE: Something Extra
Problem 48AP: The infrared spectrum of the compound with the mass spectrum shown below lacks any significant...
Related questions
Concept explainers
Question
100%
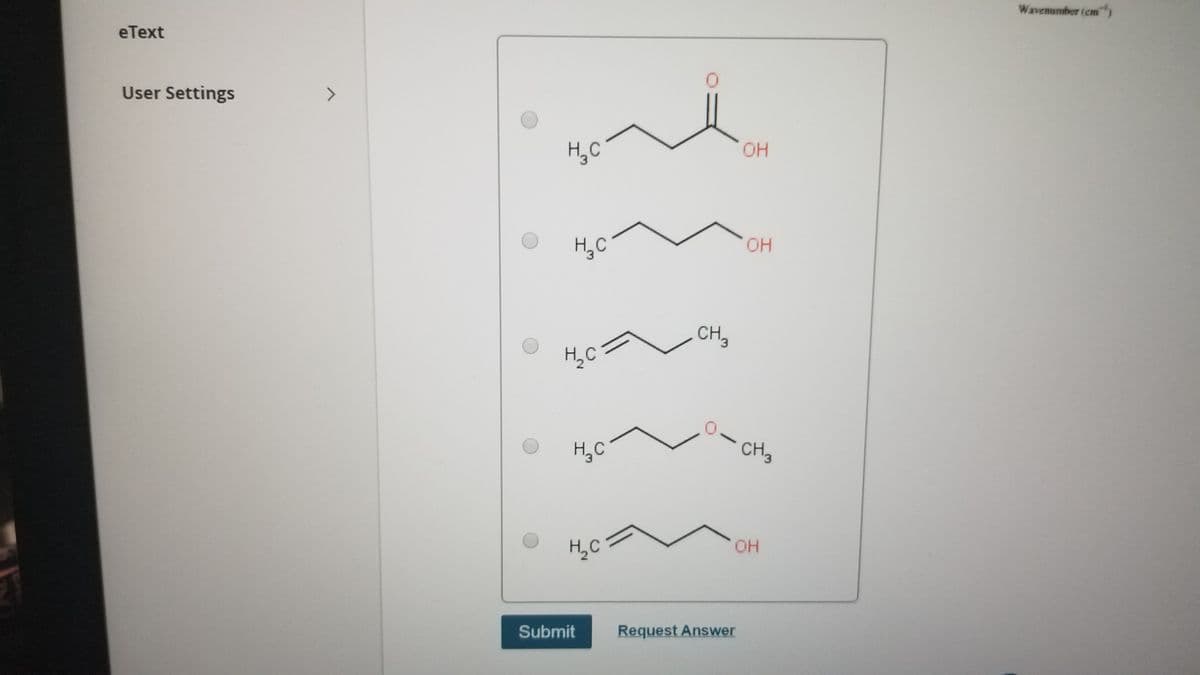
Pick correct choice
Transcribed Image Text:Wavenumber (cm)
eТext
User Settings
H,C
OH
H,C
OH
CH,
H,C
H,C
CH2
H,C
OH
Submit
Request Answer
Transcribed Image Text:I Review | Constants
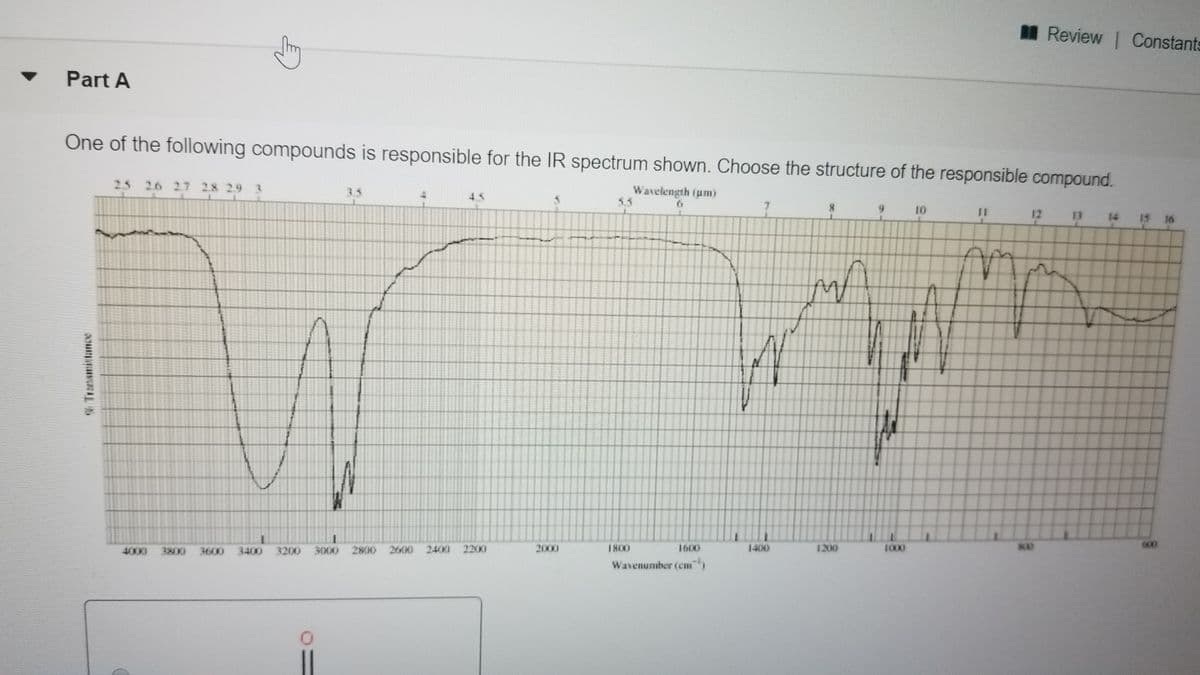
Part A
One of the following compounds is responsible for the IR spectrum shown. Choose the structure of the responsible compound.
2.5 26 27 28 2.9
Wavelength ium)
10
12
13
14
15 16
4000 3800
3600
3400 3200 3000
2MH) 2600 2400 2200I
2000
1800
1600
1400
1200
1000
600
Wavenumber (em)
Tranamictano
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

