Q: 15.5 (Pages 650 -656), 15.7 (Pages 661-666) Suppose that a catalyst lowers the activation barrier of...
A: Given : Temperature = 298K
Q: 69. This compound is caled, CH2 CH3 HạC- N CH -N -CH OO OCH, Resin O Chlorophyll O steroid
A: Identify the given compound by its structure and check whether that substance is correct or not acco...
Q: What is the pH created when 2.600 L of an aqueous weak monoprotic acid whose molarity is 0.1750 M an...
A:
Q: 52. In an extraction using two-solvent system, methanol can be used decrease the solubility of the n...
A: The technique in reducing of organic solutes by saturating mixture is called Salting out process
Q: Predict the MAJOR product(s) of each reaction or sequence of reactions. Show stereochemistry where a...
A: Nucleophilic substitution reactions (NSR) : These are two types 1. SN1 : Unimolecular nucleophili...
Q: Goal #1: See page 3 of the procedure for equations. Calculate the mass of CaCl2•2H>O@ required to ma...
A: The molecular weight of CaCl2.2H2O = 147 g/mol The molecular weight of KIO3 =214g/mol The molecula...
Q: Calculate the osmolarity of a 1.15% (m/v) KBr solution. concentration: osmol/L
A:
Q: If you start with 150 g of Fe2O3 as the limiting reagent, what is the theoretical yield of Fe? (b) I...
A: Carbon monoxide (CO) reduces ferric oxide to iron. The calculation shown below.
Q: Predict the shape of SiF4,SF4 and XeF4 molecules. Explain why the shapes differ.
A: Shape of SiF4, SF4 and XeF4 = ? Explain why shape is different.
Q: ОН НО HO- ОН HO НО- OH OH ОН ОН HO What are the monosachharides that make up this trisaccharide? O a...
A:
Q: Part A What is the order of this reaction?
A: Let's say, we have the following reaction: A→P Where, A is the reactant and P is the product. Now, i...
Q: 3. Solve the following. Express the answers in appropriate number of significant figures. Show your ...
A: Significant digit is defined as any of the digits of a number beginning with the digit that is not z...
Q: Consider the reaction 2 H, + O, → 2 H,0 AHxn = -484 kJ Which answer best describes the transfer of h...
A:
Q: 3A+4B→2C is found to be second order with respect to A and the rate constant for the reaction is k ...
A:
Q: Draw the structural condensed formula of 2,2-dimethylhexan-3-one.
A:
Q: What is the formula of molality? a. moles solute / L of solution O b. moles of solute / kg of solven...
A:
Q: Determine what could be the best solvent to differentiate anthracene and benzylamine based on their ...
A:
Q: How many significant figures are in each measurement? 1) 3.005 g 2) 820 m 3) 0.000 670 km 4) 0.405 ...
A: Given question is : How many significant figures are in each measurement = ?1) 3.005 g2) 820 m3) 0.0...
Q: b) Show the mechanism for the radical chlorination of neopentane. Illustrate homolytic bond cleavage...
A:
Q: 2. The cell potential of this electrochemical cell depends on the pH of the solution in the anode ha...
A:
Q: The radioactive nuclide ⁶⁰₂₇ Co has a half-life of 5.27 years. Calculate the mass of ⁶⁰₂₇Co that rem...
A: Given,
Q: Which of the following statements about buffers is TRUE? Select one: A. When pH = pKa, the weak acid...
A: An acid is substance which can release hydrogen ions and base is substance which can release hydroxy...
Q: the molarity of cl- in an aquoues solution which was %w/v 5.85% naCl , 11.1% cacl2 and 5.35% nh4cl w...
A: Given, %w/v of NaCl = 5.85% %w/v of CaCl2 = 11.1% %w/v of NH4Cl = 5.35% What is the molarity of Cl...
Q: 10x TE is 0.1 mol/L Tris-Cl and 0.01 mol/L EDTA. It is commonly made from 1.0 mol/L Tris-Cl and 0.5 ...
A: TE buffer is called Tris-EDTA buffer.
Q: Kp
A:
Q: Consider the reaction. 2 Fe,0, 4 Fe +302 AHixn +824.2 kJ The formation of 58.0 g of Fe results in
A: As per the balanced reaction- Generation of 4 mol Fe requires 824.2 kJ energy. So, Energy absorbed =...
Q: Linear polymers are more water-soluble than branched ones. True False
A:
Q: 1. When temperature of a gas increases under isobaric conditions, volume will [ Select ] This relati...
A: In isobaric process , pressure = constant
Q: The ionic strength of a solution containing 0.00100 F KCl, 0.0048 F K3PO4, and 0.0070 F Pb(NO3)2 is ...
A:
Q: 4. Assume that a student places 1.0 mol of solid PbBr2 in 1.0 L of 0.0025 M Pb2+. Part of the lead b...
A: Recall the given equation of dissociation that will occur here PbBr2s→Pb2+aq + 2Br-a...
Q: use a walsh diagram to explain which is more stable BrF3 (Linear) or BrF3 (bent) . please explain...
A: Interpretation- To explain walsh diagram and and tell which is more stable BrF3 (Linear) or BrF3(be...
Q: gal(a1-6)gal(a1-6)glc(a1-28)fru. Which of the complete IUPAC of this tetrasaccharide? O a-D-galactop...
A: Here we have to write IUPAC name of the following tetra saccharide
Q: oxidation number
A:
Q: _is used since the polarity of an organic molecule is related 19. to the presence of polar bonds tha...
A: Solubility of organic molecules: The solubility of organic compounds is controlled by intermolecula...
Q: 2. When temperature of a gas decreases under isometric conditions, pressure will [ Select ] This is ...
A:
Q: 63. The figure below is the structural formula of O cyclohexane O cyclohexene O cyclopentane
A:
Q: How do you calculate molarity of vinegar when vinegar is 5% by mass acetic acid and it's density is ...
A:
Q: H OH H но. NHCH, CH,NHCH, HO, CH HO (-)-ephedrine (-)-epinephrine (adrenalin)
A:
Q: Can you fill the rest of the chart please using the definitions from the top
A: This chart is self explanatory for the given chemicals. No need to fill out these if there is blank ...
Q: 56. The picture of the organic compound below is called Napthalene O Antracene O Phenol
A: I will give you the answer for this question. As you have given the options will select the best amo...
Q: Provide the correct IUPAC name for the compound shown here.
A: In IUPAC naming select the longest carbon chain which is has more number of substituents. If the sub...
Q: Molecule ЕPG Hyb (circle if 2D Lewis Structure 3D Lewis Structure MG polar) BA
A: We have to tell about the molecule IBr5. Thus the hybridization of the molecule is = (No. valence el...
Q: SOME MOLECULAR FORMULAS: Practice line drawings using your SPONCH rules 1. Hydrogen sulfide H2S 2. C...
A: We have to draw the structure of the compound given above. Draw a nucleotide with the base represen...
Q: Match the left phrase with the right phrase. Choose the best match. Non-crystalline Cubic structure ...
A:
Q: How much heat is required to convert 40.0 g of ice at -10.0 oC to 100.00 oC? The specific heat of ic...
A:
Q: What is the empirical formula of X
A: The empirical formula of the compound can be calculated with the help of percentage composition of e...
Q: A sample of ideal gas at room temperature occupies a volume of 15.0 L at a pressure of 752 torr. If ...
A: Given, Note: 760 torr = 1 atm Initially: Pressure of a gas (P1) = 752 torr = 0.989 atm Volume of a g...
Q: Calculate the mass percent composition of (NH4)2SO4 (Show work please)
A: The solution is as follows:
Q: the mass percent of magnesium in MgSO4.
A:
Q: What color will be produced if cyclohexene and toluene are reacted with sulfuric acid?
A: Alkane doesn't react with sulphuric acid or inert to concentrated sulphuric acid. If I take into con...
What are the reagents required for this reaction?
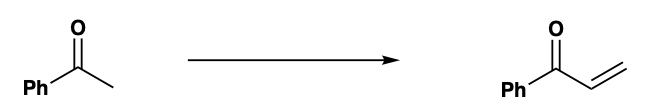
Step by step
Solved in 2 steps with 1 images


