Q: a marble is placed on a scale and is found to has a mass of 22.2g.if the radius of the marble ball…
A: The mass per unit volume of a substance is known as the density of the substance. The density of a…
Q: 2- The chemical shift value of o-nitro toluene is higher than that of m-nitro toluene.
A: 2) In 1HNMR spectroscopy , the substituents, such as -NO2 can act as an electron withdrawing group…
Q: Convert the following into line-bond formula, indicating all lone pairs. н нн H-C-C H I H-C-C H H H…
A: Given compound: We have to convert this into the line-bond formula.
Q: In the specific heat equation the delta T symbol (∆T) stand for?
A: The specific heat capacity equation is used to find the heat change in a material due to a change in…
Q: If I have a mass of 78.8 kg and a volume of 79.3 L, how dense am I in grams per microliter?
A:
Q: This assignment will require some internet research. Choose a chemical reaction that is used in…
A: The reactions provided in question can be placed under the following categories as follows: a.…
Q: READ THIS: When you hear that NASA's space probes are looking for "evidence of life" on other…
A:
Q: 0.010 J. -C). :).0- mmol. C , mol "C
A:
Q: Which of the following species is/are not a resonance form(s) of the anionic species in the box? S S…
A:
Q: Prelab Questions: These questions must be completed before starting the lab activity. 1) For the…
A: Increment : Increment is defined as smallest possible measurement in scale between two graduations…
Q: During the compression stroke of a certain gasoline engine, the pressure increases from 1.00 atm to…
A:
Q: If I have a mass of 78.8 kg and a volume of 79.3 L, how dense am I in grams per microliter?
A:
Q: TRY IT OUT 1. LICIO3 2. Sr(CIO3)2
A:
Q: Circle every active position and indicate at each whether it is electrophilic or nucleophilic.
A: An electrophile is an atom or group of atoms that is deficient of electrons and hence attracted…
Q: The specific heat capacity of a pure substance can be found by dividing the heat needed to change…
A:
Q: is my answer to Q10 (a,b) correct, is there anything else to include also,for this…
A: Your answer to part-(a) is almost correct. Your answer to part-(b) is almost incorrect because you…
Q: How many milliliters of a solution that is 10.57% (w/w) HNO3 with a density of 1.2306 g/mL should be…
A:
Q: Calculate the original concentration of a solution if 50 mL of the original solution was diluted to…
A: Here we are required to find the concentration of original solution when original solution is…
Q: 6. (a) In structure 3 of Model 2, the double bond electrons move to form a lone pair on oxygen. The…
A:
Q: Classify each of these solids as ionic, molecular, metallic, or covalent (also known as…
A:
Q: Area or Field Medicine ommunication Consumer Product Some Chemical Components (Chemical Formula) Use…
A: Consumer Product Consumer products are those that are purchased by the typical customer for…
Q: Components (distilled water + ammonium chloride) (distilled water + naphthalene) (distilled water +…
A: Polar molecules: The molecules which have an electronegativity difference between the bonded…
Q: [Review Topics) [References) The chemical environment of protons can be deduced from their chemical…
A: The most shielded protons are those that are surrounded by the highest electron densities. Since…
Q: Chemistry A KCl solution (Mm = 74.55 g/mol) was prepared by diluting 0.3473 ± 0.0002 g in 250.0 ±…
A: #A: Given the mass of KCl taken = (0.3473 ± 0.0002) g Molar mass of KCl = 74.55 g/mol Required…
Q: Calculate the enthalpy change using the net ionic equation for the given neutralization reaction at…
A: Answer: Chemical reaction mentioned in the problem statement is a neutralization reaction between…
Q: Convert 8.85 × 10-⁹ S to microseconds. Use only the metric system.
A:
Q: 6-6-6-6 CH3 I CH₂ CH₂ CH₂-C-CH₂-C- CH₂ CH3 I CH3 I CH3 CH-CH-CH₂-CH₂-CH₂ I I CH3 CH₂ - CH₂ CH₂ - CH₂…
A: We follow certain rules assigned by IUPAC to name organic molecules. Some of these rules are given…
Q: Briefly discuss how you would experimentally confirm the curcumin molecule is coordinated to the…
A: Curcumin forms stable complex with many metal ions and it is a didentate ligand. It binds with metal…
Q: Q_ The product of poly ethylene contains twelve molecules, four of the molecules have Dp-220. three…
A:
Q: 5. Using what you've learned in class about the buffering system of the blood, derive an equation…
A: #5: The equilibrium reaction for the formation of carbonic acid, H2CO3(l) from the dissolved CO2,…
Q: 2+ A current of 3.40 A is passed through a solution containing Cu²+ (aq) for 30.0 minutes, with…
A:
Q: TRY IT OUT 1. LiOH 2. Sr(OH)2
A:
Q: You are the public health officer in the water treatment facility for a city of 50,000 people. A…
A:
Q: 3 Questions 1. Draw the molecular structures of the two isomers A and B of [Mo(CO)4(PPH3)2] that you…
A: According to the bartleby guidelines for the multiple questions, I can solve only first one. So I'm…
Q: 2) If a brick has a length of 13.77 cm, a width of 8.50 cm, and a height of 5.12 cm: a) What is the…
A:
Q: 1. The antibacterial activity of flavonoids was tested against the bacterial strain, Escherichia…
A: A question based on molecules that is to be accomplished.
Q: methanol is less polar than water, but it is more polar than the C-18 molecule, a completely…
A: Solution: We know that polarity of any compound depends upon the resultant dipole moment. However…
Q: 4. Complete the following reactions by adding the missing starting material, reagents and/or produ…
A:
Q: Identify the hybridization (sp, sp2, sp³) of each carbon atom in the following molecule.
A:
Q: WRITE THE STRUCTURAL FORMULA OF THE FOLLOWING: (NOT SKELETAL FORMULA) 1. 2 Ethyl, 5 Methylnonane…
A:
Q: If the fatty acid 12:0 is catabolized completely, to carbon dioxide and water, the net yield of ATP…
A: Given that the fatty acid is catabolised completely. We can calculate the net yield of ATP per…
Q: Calculate the theoretical yield of CO₂ in grams when 3.5 g of C₂H4 is reacted with 30.0 g of O₂…
A:
Q: Chemistry Organize the following three molecules in order of increasing stability in water (least >…
A: We have to organize the three molecules acid chloride, ester and an amide in order of increasing…
Q: Starting with the following equation, Fe₃O₄(s) + CO(g) → Fe(l) + CO₂(g) calculate the mass in grams…
A:
Q: The compound cyclopentanone has the molecular formula C,H,O. The 13C-NMR spectrum has three distinct…
A:
Q: Pentane (C5H12) and hexane (C6H14) combine to form an ideal solution. At 25°C the vapor pressures of…
A: Introduction to Raoult's law Raoult's law is a physical chemistry relationship that has…
Q: An unknown substance shows a molecular ion peak at m/z = 170 with a relative intensity of 100. The M…
A: Dear student since you have asked multiple questions but according to guidelines we will solve 1st…
Q: Model 3: Stability of Resonance Forms Not all resonance forms have the same energy; some forms may…
A: #8: We are given the resonance structures for the carboxylic acid, RCOOH and we have to answer the 5…
Q: I am asked to obtain the Fischer projection of compound A. I am given two possible answers (B and…
A:
Q: Identify the element of arts and discuss each in your own words (150 word)
A: Element of Art Through an endless vocabulary of eight terms, all art communicates in the same…
Please answer parts i and j
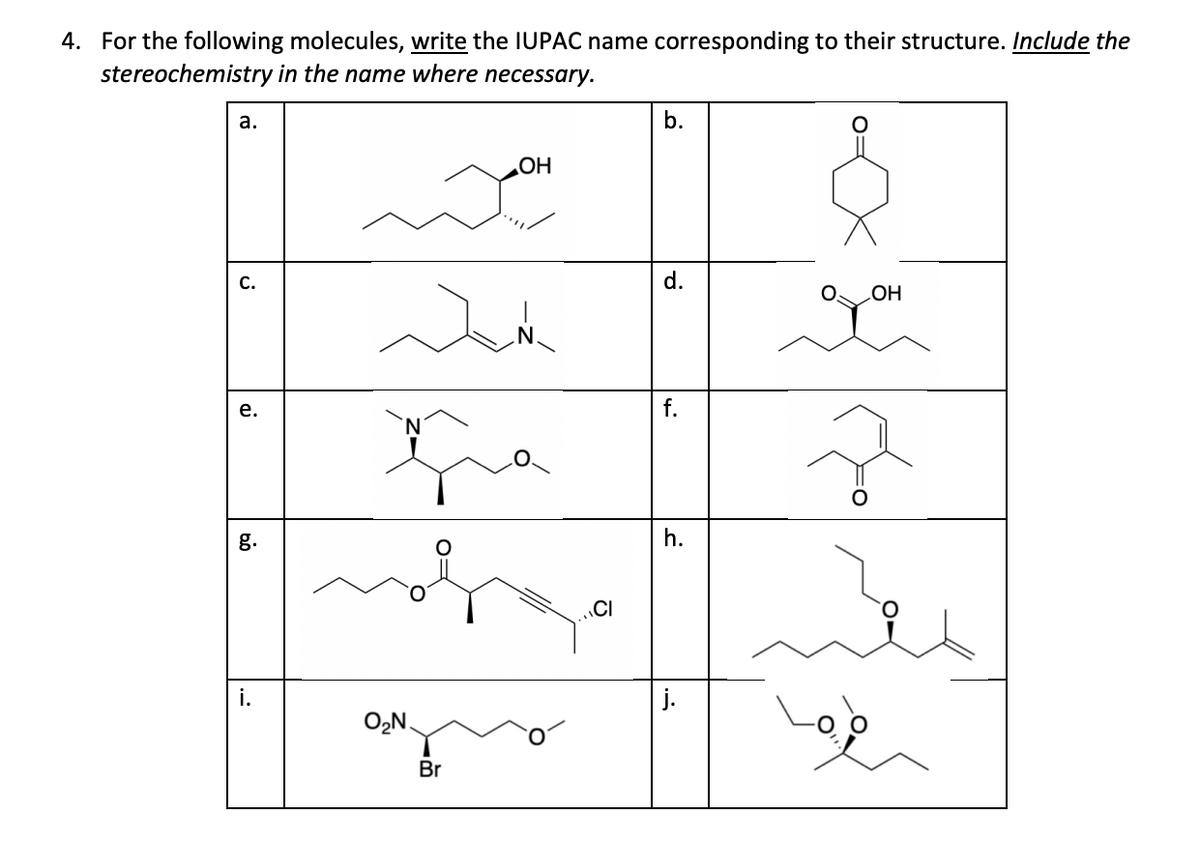
Step by step
Solved in 2 steps with 1 images

- help me please Please fill in the reagents,intermediates, and/or products of the following reactions. all the reactions shall be covered in Chapter 12-15 from Organic Chemistry, 6th Edition by Marc Loudon and Jim Parise.a) Write a description of the compounds involved in the synthesis (functional groups, specific features). b) Describe the steps involved in the synthesis and the role of each step. Each step should clearly indicate the reagent used, the stereochemistry involved in the reaction and its importance or relevance to the multistep synthesis.H8. Organic chemistry Text answers and (descriptive structures and / or reaction mechanisms). Explain the chemistry of the carbonyl group (direct addition of nucleophiles and addition with elimination)
- I need specific description of the compounds involved in the synthesis (functional groups, specific features). and also describe the steps involved in the synthesis and the role of each step. And most important!!devote one to each step of the synthesis. Each step should clearly indicate the reagent used, the stereochemistry involved in the reaction and its importance or relevance to the multistep synthesisOrganotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides.One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(C2H5)3). 3SnCl4 + 4Al(C2H5)3 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.230 L of SnCl4 (d = 2.226 g/mL) was treated with 0.396 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). If 0.335 L of tetraethylstannane (d = 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?Hello. Kindly help me understand if this is a possible schematic of the reaction. The specific question is in Organic Chemistry Principles and Mechanisms by Joel Karty 2nd Edition, Problem 12.11 on page 614. I believe the predicted major product should come as well as with its enantiomer hence 4 products predicted for this reaction as there is no easy rotation about the cyclic structure
- II. Alcohols in Synthesis B.) How are alcohols used to make ethers in the Williamson ether synthesis? How do you design the synthesis of ethers using both acidic and basic Williamson ether synthesis strategies?Topic: Alkyl Halides Instructions: Fill in the empty boxes below with the proper reagent(s). Struggling with this. I would appreciate a walk through.1. Using Br2 in C2H4Br2 will result in HBr and ______. a. C2H3Cl3 b. C2H4Cl3 c. C2H2Cl3 d. none of the above 2. How many halogenation are posible in propane? a. 3 b. 8 c. 6 d. 10 3.Sulfonation of pentane will result in ________ and water. a. C5H11SO3H b. C5H12SO3H c. C5H14SO3H d. none of the above 4.Nitration of hexane will result in ________ and water. a. C6H13SO3H b. C6H15NO2 c. C6H13NO2 d. C6H14NO2 5.How many moles of O2 in heating a C12H26 (dodecane) a. 27 b. 37 c. 24 d. none of the above
- Draw the major organic product you would expect to isolate from the reaction of p-bromoethoxybenzene with a misture of HNO3HNO3 and H2SO4H2SO4.Give the major products or reagents represented by letters E and F:The standard procedure for synthesizing a compound is the stepwise progress toward a target molecule by forming individual bonds through single reactions. Typically, the product of each reaction is isolated and purified before the next reaction in the sequence is carried out. One of the ways nature avoids this tedious practice of isolation and purification is by the use of a domino sequence in which each new product is built on a preexisting one in stepwise fashion. A great example of a laboratory domino reaction is William S. Johnsons elegant synthesis of the female hormone progesterone. Johnson first constructed the polyunsaturated monocyclic 3 alcohol (A) and then, in an acid-induced domino reaction, formed compound B, which he then converted to progesterone. A remarkable feature of this synthesis is that compound A, which has only one stereo-center, gives compound B, which has five stereocenters, each with the same configuration as those in progesterone. We will return to the chemistry of Step 2 in Section 16.7 and to the chemistry of Steps 3 and 4 in Chapter 19. In this problem, we focus on Step 1. (a) Assume that the domino reaction in Step 1 is initiated by protonation of the 3 alcohol in compound A followed by loss of H2O to give a 3 carbocation. Show how the series of reactions initiated by the formation of this cation gives compound B. (b) If you have access to a large enough set of molecular models or to a computer modeling program, build a model of progesterone and describe the conformation of each ring. There are two methyl groups and three hydrogen atoms at the set of ring junctions in progesterone. Which of these five groups occupies an equatorial position? Which occupies an axial position?



