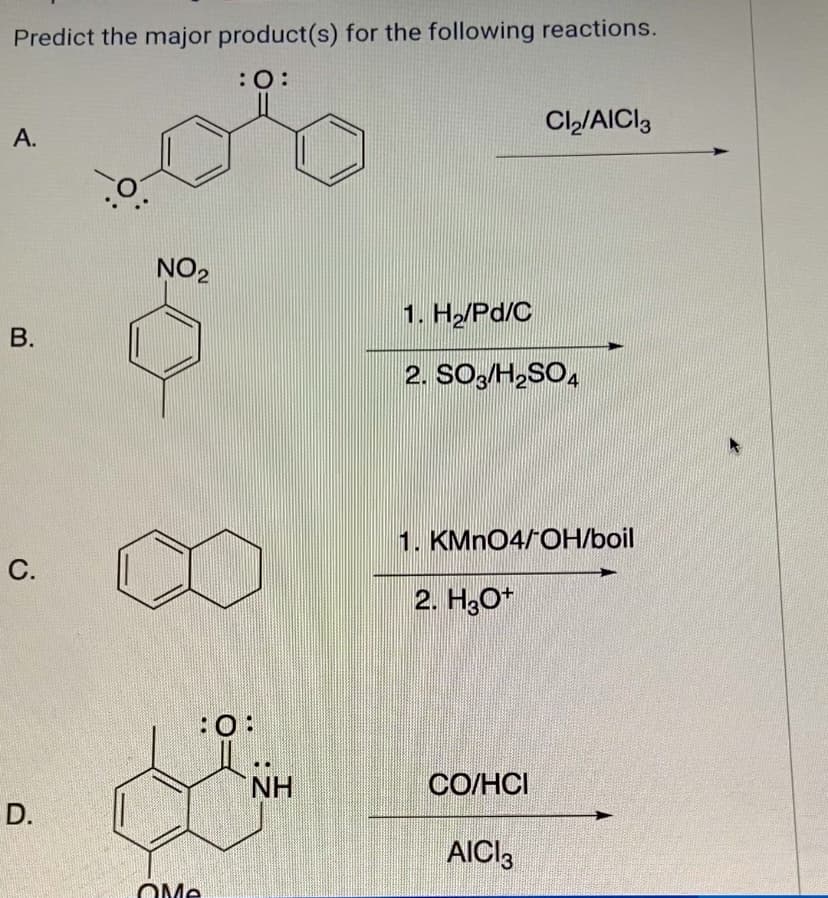
Predict the major product(s) for the following reactions. :0: A. Cl₂/AICI 3 B. NO2 1. H₂/Pd/C 2. SO3/H2SO4 C. D. QMe :0: 1. KMnO4/OH/boil 2. H3O+ NH CO/HCI AICI 3
Q: Please correct answer and don't use hend raiting
A: This reaction is a Wittig reaction. The Wittig reaction is a chemical reaction used to synthesize…
Q: A solution contains 8.30×10-2 M ammonium bromide and 0.455 M ammonia.The pH of this solution is
A: The objective of this question is to calculate the pH of a solution containing ammonium bromide and…
Q: Question 20 Choose the correct chemical structure for every single acronym used below: Py ? A B с D…
A:
Q: It says that my answers are wrong and I can’t figure out what went wrong please help!
A:
Q: Describe how you could achieve a higher degree of separation for the bands of food dyes on a TLC…
A: Step 1:Optimize the Mobile Phase: The choice of mobile phase, which is the solvent that moves up the…
Q: Which of the statements about benzoin synthesis is wrong? A. The synthesis carried out is an…
A: Benzoin can be prepared by the action of an alkali cyanide on an alcoholic solution of benzaldehyde…
Q: d Use curved arrows to show the mechanism of the step below. Make the ends of your arrows specify…
A:
Q: When the Ag* concentration is 1.21 M, the observed cell potential at 298K for an electrochemical…
A: The objective of the question is to find the concentration of Cr3+ in an electrochemical cell…
Q: A prepared buffer solution contains a mixture of H2PO4 and HPO ions. Which species will react with…
A: The objective of the question is to identify which species in a buffer solution will react with any…
Q: Question 15 Choose the correct chemical structure for every single acronym used below: n-BuLi ? A B…
A: Step 1:The acronym "n-BuLi" stands for "normal butyllithium." "n-" Prefix: In this context, "n-"…
Q: Question 10 Please predict the products for each of the following reactions: 1. Na 2. PrBr เวรเ 10…
A: Step 1:At first presence of Na metal ,it released one electron , which abstract alcoholic proton and…
Q: Using reaction free energy to predict equilibrium composition elld Consider the following…
A: Given only N2 and NH3 in a vessel and without H2, the only direction that the reaction will go to is…
Q: The following data was obtained by measuring the volume of a trapped sample of gas at different…
A: a,b AnsC)d) ANY DOUBT PLEASE ASKThank you.
Q: Home - Profession... A mixture of nitric and sulfuric acids generates the nitroniium ion, *NO2…
A:
Q: 3. Beginning with the indicated starting materials (plus another starting material of your choice…
A: Step 1: Step 2: Step 3: Step 4:
Q: ← 68 Problem 15 of 27 Submit Draw the major product of this reaction. Ignore inorganic byproducts.…
A: Step 1:Step 2:
Q: O Chemical Reactions Determining the volume of base needed to titrate a given mass of acid 3/5 Bis A…
A:
Q: What are some characteristics of proteins? Question 27 options: Average…
A: The objective of the question is to identify the correct characteristics of proteins from the given…
Q: Question 23 Predict the FINAL (?) product for each of the following reaction or synthetic chain: QA…
A: Step 1:
Q: For each organic compound in the table below, name the highlighted side chain. Note for advanced…
A: Thank you.
Q: All are true of the Embden-Myerhof pathway EXCEPT Question 88 options:…
A: The objective of the question is to identify the statement that is not true about the Embden-Myerhof…
Q: Q/ How many grams of Anilin Co Hts NHz should be dissolved in water to prepare a solution of 1 liter…
A: The concentration of hydroxide ions in the solution = Equilibrium constant = Desired volume =
Q: Draw the product of the following reaction. 1. 2. но Li
A:
Q: show the mechanism please
A: The reaction is the Friedel-Craft acylation reaction. First, acyl chloride reacts with AlCl3 and…
Q: Identify whether each of the following molecules can exist as optical isomers: NH₂ Br-C-COOH H…
A: The objective of the question is to determine whether the given molecules can exist as optical…
Q: Differential Thermal Analysis (DTA) and Differential Scanning Calorimetry (DSC) are used to support…
A: Thermal analysis methods that are often used to analyze polymers and other materials include…
Q: Give the IUPAC name of the molecule.
A: Step 1:name of the molecule is "3-tert-butylcyclopentane-1-carboxylic acid"Explanation 1. Select…
Q: Using thermodynamic data to calculate K Using any data you can find in the ALEKS Data resource,…
A: STEP 1: to compute for the equilibrium constant of the reaction we need to use this equation:ΔG° =…
Q: Using chemical equations, show how the triprotic acid HPO ionizes in water. Phases are 4 optional.…
A: The objective of the question is to illustrate the ionization process of the triprotic acid H3PO4 in…
Q: Which statement is true about acids and bases? Brønsted-Lowry acids are proton acceptors, and…
A: The objective of the question is to identify the correct statement about acids and bases according…
Q: Identify the Brønsted-Lowry acid and base in each of the reactions. HF + H₂O → H3O+ + F¯ H₂O + H2CO3…
A: Step 1: Step 2:Acids: According to the Brønsted-Lowry definition, an acid is a substance that…
Q: Draw all the products of this reduction reaction: H₂N NIR H3C CH3
A: Step 1: Determine the functional group: Determine the amine group (NH2) in the reactant molecule.…
Q: Draw the bridged bromonium ion that is formed as an intermediate during the bromination of this…
A: Step 1: Step 2: Step 3: Step 4:
Q: 1. Provide an acceptable name for the following molecule. Only specify R and S where shown as 3D., H…
A: Identify the longest continuous carbon with the highest priority functional group.Replace the end of…
Q: help question 10
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: None
A:
Q: 8. Identify the most practical route to synthesize the following compound among the various…
A: To effectively answer your query on synthesizing the compound with a phenyl group (Ph) and a…
Q: Why are oxygen and nitrogen important gases in our atmosphere?
A: The importance of oxygen and nitrogen in our atmosphere can be understood by looking at their roles…
Q: 10. Provide a Fischer projection for the sugar shown below. At which position(s) is the…
A:
Q: The scientific report about Nitration of methyl benzoate experiment
A: ### Introduction to Electrophilic Aromatic SubstitutionElectrophilic aromatic substitution (EAS) is…
Q: What is the pH of the solution that results when the following solutions are combined? 0.020 L of…
A: Thank you,Please rate my response.
Q: Name the following molecule using the IUPAC system Br
A: The following steps are taken in consideration while naming Alkylbromides:Step 1:Identify the…
Q: Calculate the cell potential of a cell composed of a cathode consisting of a Cd wire immersed in a…
A: This is question is from physics.This is one step solution.We can find cell potential by using…
Q: None
A: Here is the drawing of the catalytic cycle for the Grubbs generation 2 catalyzed olefin metathesis…
Q: None
A: Step 1:Step 2: Step 3: Step 4:
Q: dont provide handwriting solution ...
A: Note: HCOO (formic acid) is a Bronsted-Lowry acid and not a Bronsted-Lowry base. It can donate a…
Q: Super hero expert Hand written solution is not allowed
A:
Q: The first enzyme to rise in acute pancreatitis is ________________, and the enzyme which stays…
A: The objective of the question is to identify the enzymes that are affected during acute…
Q: 4. Which acid and alcohol must be reacted to generate the indicated esters? (a) (b) (c) ов ↑ (d) OH
A: Esters are formed when acid chlorides are reacted with alcohol nucleophiles. This is the more…
Q: a. Composition of the eutectic mixture b. Consider a mixture with overall B composition of 0.57.…
A: The main aim of this question is to show that the Txy diagram is a graphical representation of the…
Step by step
Solved in 2 steps with 2 images

- Describe how the product is purified. 4,4'-DIBROMOBIPHENYL [Biphenyl, 4,4'-dibromo-] Submitted by Robert E. Buckles and Norris G. Wheeler1. Checked by R. S. Schreiber, Wm. Bradley Reid, Jr., and Robert W. Jackson. 1. Procedure In a 15-cm. evaporating dish is placed 15.4 g. (0.10 mole) of finely powdered biphenyl (Note 1). The dish is set on a porcelain rack in a 30-cm. desiccator with a 10-cm. evaporating dish under the rack containing 39 g. (12 ml., 0.24 mole) of bromine. The desiccator is closed, but a very small opening is provided for the escape of hydrogen bromide (Note 2). The biphenyl is left in contact with the bromine vapor for 8 hours (or overnight). The orange solid is then removed from the desiccator and allowed to stand in the air under a hood for at least 4 hours (Note 3). At this point, the product weighs about 30 g. and has a melting point in the neighborhood of 152°. The crude 4,4'-dibromobiphenyl is dissolved in 75 ml. of benzene, filtered, and cooled to 15°. The…Taking Ka for acidic acid as 1.8 x 10-5, calculate H3O+ , using the known concentraions of acidic acid and soudium acecate contained in reaction mixtures 1 and 4. Reaction mixture time (s) Acidic acid concentration (mol/L) sodium acetate concentration (mol/L) calculated H3O+ concentration 1 237 0 1 x 10 -5 4 236 1 x 10 -4 1 x 10 -5The methoxy group is reported as having σpara and σmeta values that differ in sign (σmeta =+0.12; σpara = –0.27). What specific experiments are performed to determine these s values?Explain why the σpara and σmeta values are of different sign and what those signs indicate.
- Provide details about the reaction workup. 4,4'-DIBROMOBIPHENYL [Biphenyl, 4,4'-dibromo-] Submitted by Robert E. Buckles and Norris G. Wheeler1. Checked by R. S. Schreiber, Wm. Bradley Reid, Jr., and Robert W. Jackson. 1. Procedure In a 15-cm. evaporating dish is placed 15.4 g. (0.10 mole) of finely powdered biphenyl (Note 1). The dish is set on a porcelain rack in a 30-cm. desiccator with a 10-cm. evaporating dish under the rack containing 39 g. (12 ml., 0.24 mole) of bromine. The desiccator is closed, but a very small opening is provided for the escape of hydrogen bromide (Note 2). The biphenyl is left in contact with the bromine vapor for 8 hours (or overnight). The orange solid is then removed from the desiccator and allowed to stand in the air under a hood for at least 4 hours (Note 3). At this point, the product weighs about 30 g. and has a melting point in the neighborhood of 152°. The crude 4,4'-dibromobiphenyl is dissolved in 75 ml. of benzene, filtered, and cooled to 15°.…I’m currently trying to write a lab report for the synthesis of dimolybdenum tetraacetate [Mo2(O2CCH3)4] from the reaction of molybdenum hexacarbonyl, Mo(CO)6 in glacial acetic acid and acetic anhydride under a nitrogen atmosphere, involving the difficult formation of a quadruple bond and requires high heat and long reaction time (approximately 20 hours). But these are the question I’m stumped on: 1. Why need the reaction be done under nitrogen? We also added dichlorobenzene and hexanes during the reaction. 2. Explain the purpose of dichlorobenzene and hexanes. 3. Why does the reaction take 20 hrs?In light of your answer to Problem 30-40, explain why a mixture of products occurs in the following reaction:
- What is the percent dissociation of 0.150 M triethylamine? (The Kb value for triethylamine is 5.25 × 10-4.) a. 5.92 %b. 5.75 %c. 39.4 %d. 5.75 x 10-30 %Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides. One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(CzHs)3). 3SnCl4 + 4Al(C2H5)3 → 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.160 L of SnCl4 (d= 2.226 g/mL) was treated with 0.346 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). What is the theoretical yield in this experiment (mass of tetraethylstannane, Sn(C2H5)4)? If 0.257L of tetraethylstannane (d= 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?Describe every aspect of the procedure clearly and explicitly. Include temperatures, appearance of the reaction (include pictures!). How is the reaction monitored? Is the order of addition important? 4,4'-DIBROMOBIPHENYL [Biphenyl, 4,4'-dibromo-] Submitted by Robert E. Buckles and Norris G. Wheeler1. Checked by R. S. Schreiber, Wm. Bradley Reid, Jr., and Robert W. Jackson. 1. Procedure In a 15-cm. evaporating dish is placed 15.4 g. (0.10 mole) of finely powdered biphenyl (Note 1). The dish is set on a porcelain rack in a 30-cm. desiccator with a 10-cm. evaporating dish under the rack containing 39 g. (12 ml., 0.24 mole) of bromine. The desiccator is closed, but a very small opening is provided for the escape of hydrogen bromide (Note 2). The biphenyl is left in contact with the bromine vapor for 8 hours (or overnight). The orange solid is then removed from the desiccator and allowed to stand in the air under a hood for at least 4 hours (Note 3). At this point, the product weighs…
- SHOW COMPLETE SOLUTIONS Q3. Which isolate has A260/280 ratio of above 2.0?Predict the product of the following reaction and classify the reaction. Pb(NO3)2+FeSO4--->PbSO4+________20- In the lab, you prepared phenylmagnesium bromide reagent using 0.81 g magnesium turnings (MW- 24.31 g/mol) and 3.6 mL bromobenzene (MW- 157.01 g/mol, density- 1.50 g/mL) in 15 mL diethyl ether (MW-74.12 g/mol, density - 0.713 g/mol). You then added 2.04 g benzophenone (182.217 g/mol) which was previously dissolved in 6.0 mL of diethyl ether solution to the phenylmagnesium bromide solution.What is the theoretical yield for triphenylcarbinol (MW- 260.33 g/mol) in grams? with the appropriate number of significant figures.



