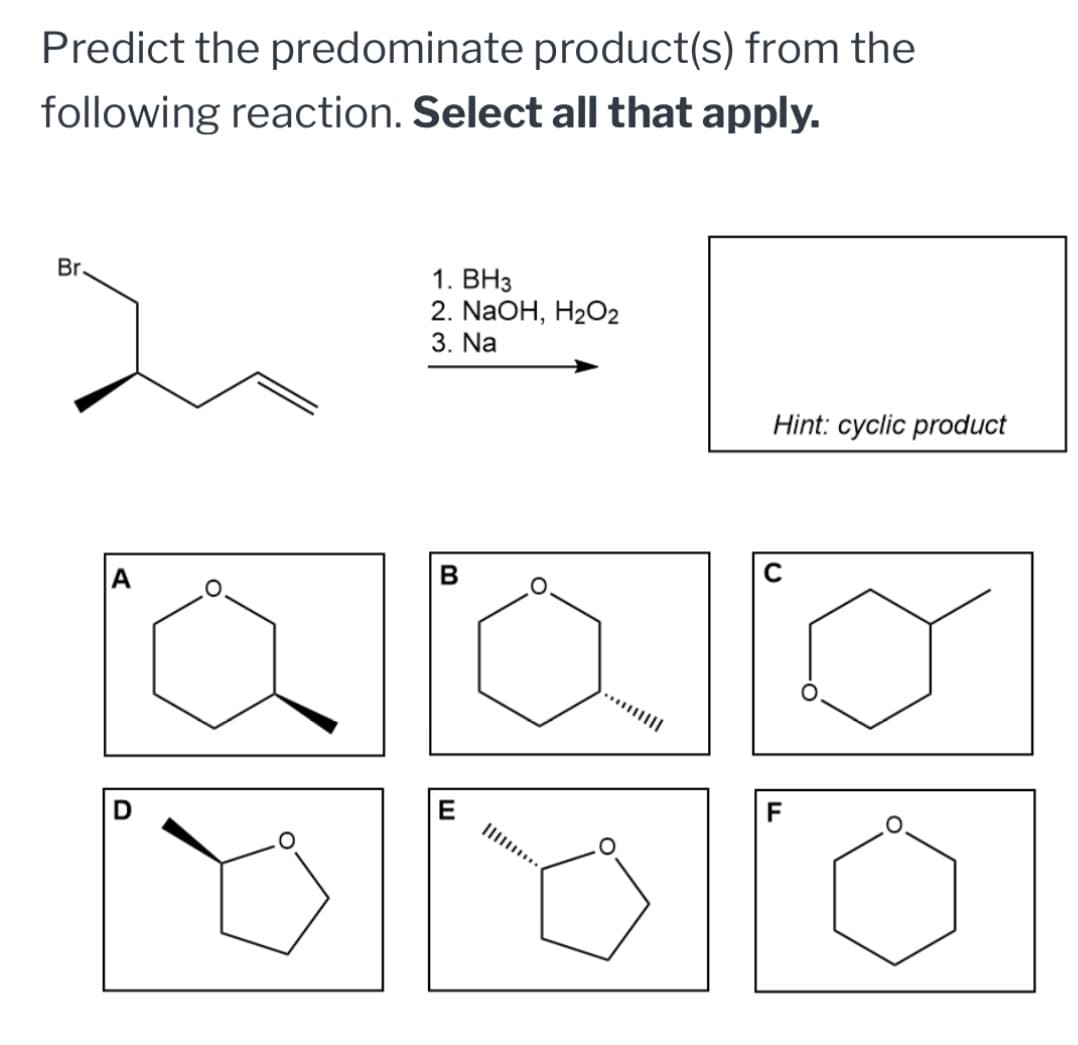
Predict the predominate product(s) from the following reaction. Select all that apply. Br. 1. BH3 2. NaOH, H2O2 3. Na A B Hint: cyclic product D E F
Q: A sample of ore containing radioactive strontium 38 Sr 90 has an activity of 6.5 × 105 Bq. The…
A: The objective of this question is to calculate the amount of strontium in grams in a sample of ore,…
Q: What is the expected product for the reaction? 1. B2H6 CH3 2. H2O2, NaOH CH3 O The following…
A: This is hydroboration oxidation reaction. This is an organic chemical reaction which is employed for…
Q: The general solution of Schrodinger Equation is a wave, Ψ=eikx=Cos kx + isinkx, where k= (2m/ħ2)1/2.…
A: Step 1:Step 2:Step 3:
Q: Curved arrows are used to illustrate the flow of electrons. Use the reaction conditions provided and…
A: Step 1: Reaction of 2-iodopent-1-ene on heating with methyl amide ion gives E2 elimination…
Q: A concentration cell is constructed of silver electrodes at 25∘C, and the half cells contain…
A: A concentration cell is a type of electrochemical cell that generates electrical energy from the…
Q: Don't provide hand writing solution
A: The objective of the question is to find the light intensity at a certain point on the screen when…
Q: Consider the following reaction at 298K. 2 Cu²+ (aq) + Ni (s) — 2 Cu* (aq) + Ni²+ (aq) Which of the…
A: Given: T=298K2Cu(aq)2++Ni(s)→2Cu(aq)++Ni(aq)2+Step 1: Write the…
Q: Chemistry
A: To convert Toluene to 3,5-dibromo-4-iodo benzoic acid First step involves Toluene undergoes…
Q: Draw the two possible products of this oxidative cleavage of an alkyne. Ignore any inorganic…
A: The reaction is an ozonolysis in an oxidative work-up of an internal alkyne which produces two…
Q: Draw the peptide Glycine-Alanine-Serine-Cysteine-Isoleucine-Glutamic acid Tryptophan-Valine. Circle…
A: Approach to finding a solution to the problem:To begin, you will need to draw the peptide sequence.…
Q: Please don't provide handwriting solutions..
A: The objective of this question is to calculate the pH of a solution during a titration process. The…
Q: 4 GIBBS ENERGY The following reaction happens in red blood cells CO2(g) + H2O (1) HCO3(aq) Using the…
A:
Q: OH This is another synthesis problem. Show reagents and intermediates synthesized along the way that…
A: The strongly activating hydroxyl group (-OH) on a benzene ring increases its electron density and…
Q: -1 A solution is prepared by dissolving A certain liquid Xhas a normal boiling point of 100.30 °C…
A:
Q: Draw one of the two enantiomers of the product of this hydrogenation reaction. Use a dash or wedge…
A:
Q: None
A: The general equation for an acid dissociation reaction is, HA(aq)⇌H(aq)++A(aq)− A stronger acid…
Q: || wwwwww wwwwwwww Benzophenone dissolved in ethanol (EtOH) reacts with 2, 4-dinitrophenylhydrazine…
A:
Q: What is the major product of this reaction?
A:
Q: Draw the product of the reaction shown below. Use a dash or wedge bond to indicate the…
A:
Q: Label the major peaks, especially those those related to fumaric acid
A: Acid are known to nake Hydrogen bond and this the reason we get broader peak left side of image…
Q: None
A: FOR ANY QUERIES, PING ME HAPPY LEARNING
Q: 11:00 PM Fri Apr 26 NÅ 88 0 Chapter-17+Homework-1 Chapter-11+Homework (1) Short answer questions 1)…
A: The objective of the question is to identify the peptide bond in the given compound, characterize…
Q: Imagine an electrochemical cell that is composed of an Ag/Ag+ half-cell and a Fe2+/Fe3+ half-cell.…
A: The objective of this question is to calculate the electrochemical potential of a cell composed of…
Q: What is the expected product of the reaction shown? Omry Br 1 ٥١ HBr OH none of these O IV HO II Br…
A: Thank you.
Q: A patient ingests 1.42 μg�g of 131I (iodine-131), a beta emitter with a half-life of 8.0 days.…
A: Convert the initial activity from micrograms (μg) to disintegrations per second (dps):We know that 1…
Q: Predict the structure of the product of each of these reactions. If no reaction occurs write NR…
A:
Q: I need help with questions please show on charts, please information Functional group- primary…
A: Step 1: Molecular formula of the compound : C3H9N.Degree of unsaturation = [C+1]−2[H+X−N]Here C =…
Q: please answer 7 ,thanks!!
A: Step 1:7 a) To calculate the percent error for the accepted value of the acid dissociated constant…
Q: 33. Platinum crystallizes with the face-centered cubic unit cell. The radius of a platinum atom is…
A: The relation between the radius of an FCC crystal unit cell to its edge length is 4R=2a where R is…
Q: Calculate the standard entropy change for the following process at 298 K:H2(g)+F2(g)⟶2HF(g) The…
A:
Q: How many mL of 1.3 % mL m (7) AICI, solution would contain 7.1 g of aluminum chloride? Round your…
A: Approach to solving the question:Please see attached photos for detailed solutions. Thank you.…
Q: A novel new method of peptide synthesis involves formation of an amide bond by reaction of an a-keto…
A: Step 1: Step 2: Step 3: Step 4:
Q: 16
A: I tried my best to make you understand but still if any further doubt regarding the mechanism of the…
Q: For the following electron-transfer reaction: Sn2+(aq) + Cd(s) Sn(s) + Cd2+(aq) The oxidation…
A: 1.Step 1: Bear in mind that electron gain occurs during reduction and electron loss occurs during…
Q: help please?
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: acetyl COA, H₂O, NAD+ HSCOA, CO2, NADH/H+ H CO₂ CO₂ a-ketoisovalerate a-ketoisocaproate Leucine is…
A: Step 1: Step 2: Step 3: Step 4:
Q: Please don't provide handwritten solution ...
A: Step 1: Step 2: Step 3: Step 4:
Q: A researcher is preparing a reaction mixture to test the activity of a protein. They combine the…
A: To determine the pH of a solution, we use the formula:pH = -log[H+]Given the hydrogen ion…
Q: provided starting structure, draw the curved electron-pushing arrows for the following reaction or…
A: Step 1: Step 2:
Q: Normalize the following wavefun (a) Y=sin(nx/L) 0≤x≤L
A:
Q: (k) 25°C MeOH Br Draw the major product(s) of the reaction. एल Draw Your Solution Draw the minor…
A:
Q: Please provide the appropriate products for the attached halogenation reactions. Thank you.
A:
Q: Can you provide a mechanism for this reaction? I’d also like maybe a sentence or two explaining each…
A:
Q: Salt bridge A concentration cell similar to the one shown is composed of two Ag electrodes and…
A: Step 1:
Q: What is the final product of the reaction scheme? The following compound: The following compound:…
A: Final answer is written inside Green box!
Q: 10) The dimerization of butadiene was studied at 500. K: 2C4H6 (g) →C8H12 (g) The following rate…
A: The objective of this question is to determine the forms of the differential and integrated rate…
Q: Draw the correct product for the reaction. (If there is no reaction, draw the starting material.)…
A:
Q: mead II. Short Synthesis. Beware of potential complications and be sure to avoid them. (10 pts each,…
A: Step 1: Step 2: Step 3: Step 4:
Q: Day 1 + ether
A:
Q: Please don't provide handwritten solution ....
A: The objective of this question is to determine the amount of sodium acetate (NaCH3CO2) that needs to…
Step by step
Solved in 2 steps with 2 images

- Chemistry please provide the flow of electrons aswell!! thank you! using the starting material (on the left) to determine the sythetic route which will be the most reaosnable and effective to theres none, you have to start witth the begin products to get to finish needed by using the minmium reagents and reactions needed to get to the final productCarefully solve the major product of each reaction and show the steps of the process.(a) Analyze SN1 or SN2 is preferred in the reaction below. Explain your choice and express themechanisms.
- ( give mechanism of 3 and 4using as much chemical and reactions as you need write in a reaction and show the mechanisms to convert starting material to product.No carcon allowed accept from starting material.Please help with this calculation of this, don't know to get the n(product) Page2. please help with it too Thanks in advance
- Determine reagents C and D from the reaction scheme below.Product the product from the reaction. Then provide an electron push mechanism for the reaction. Solve it asapGiven this organic synthesis, is there any limiting and excess reagents? also describe the procedure is run, how is the reaction monitored?Is the order fo addition important? N-ETHYLALLENIMINE[Aziridine, 1-ethyl-2-methylene-] Submitted by Albert T. Bottini and Robert E. Olsen1.Checked by Thomas H. Lowry and E. J. Corey. 1. ProcedureCaution! This preparation should be carried out in a good hood to avoid exposure to ammonia. The operator should wear rubber gloves and protective goggles because 2-haloallylamines and ethylenimines can cause severe skin and eye irritation. A 2-l. three-necked flask is fitted with a sealed mechanical stirrer, a gas-inlet tube, and a dry ice condenser protected from the air by a soda-lime drying tube (Note 1). The system is flushed thoroughly with dry ammonia, and 32.8 g. (0.84 mole) of sodium amide (Note 2) is added to the flask. The system is again flushed with ammonia, the condenser is provided with dry ice covered by acetone, and 1.2 l. of liquid…

