Q: Drag and drop the product of the following fatty acid reaction. + CH;NH/H
A:
Q: Calculate the energy required to heat 557.0 g of ethanol from 8.0 °C to 18.8 °C. Assume the specific...
A: Given mass of ethanol = 557 gram
Q: Which of the following best describes a gas? * It assumes the volume and shape of the container, and...
A: Best defining feature of gas is...
Q: Cyclopropane, C3H6, is converted to its isomer propylene, CH2=CHCH3, when heated. The rate law is fi...
A:
Q: For the reaction : H2 (g) + CO2 (g) ⇆ H2O (g) + CO (g), KC is 0.771 at 750 0C, if 0.0100 mol of H2 ...
A:
Q: Assuming that the analyte in A and B are the same organic molecule, select all possible correct reas...
A: The correct option is concentration differences.
Q: Rank order the following in terms of relative reactivity (most reactive on the left, least reactive ...
A: Since for this problem we have to determine the order or reactivity of different carbonyl. We are sh...
Q: CH,CH, 4. Which of the following pure compounds can exhibit hydrogen bonding between molecules? CH;C...
A: We know the hydrogen bond exist between the Hydrogen and the electronegative atoms ( F, N, O). The a...
Q: or each solute, identify the better solvent: water or carbon tetrachloride. Water, H,O Carbon tetrac...
A: ANSWER : Water is a polar solvent. Carbon tetrachloride is a non-polar solvent. So, The suitable sol...
Q: Question 6 Which of the following chemicals is flammable? A) iron(III) chloride solution В acetone C...
A: A solution is made up of salute and solvent.Solute is present in small amount and solvent is present...
Q: [H+]
A:
Q: Are the following halogenated or non- halogented? (a) Cyclohexyl hydrogen sulfate (b) p-Toluenesulfo...
A: Halogenated compounds are those compounds that contain halogen atom and non halogenated compounds do...
Q: What is salt
A: Salt is an ionic compound which is formed by combining of metal and non- metallic elements.
Q: If 1 CN- ion kills 10 cancer cells, how many cells would be killed by drinking 25 ml of a saturated ...
A: Cu(CN)2 <--------> Cu+2 + 2CN-
Q: 67- g/ Give a reaction mechanism OH EtO,CN=NCO,Et ʼNO2 CH2NO2 PPH3 92%
A: This reaction is Mistonbou inversion reaction where inversion of a nucleophilic centre takes place ...
Q: Sulfuryl chloride, SO2Cl2, decomposes when heated. SO2Cl2(g) → SO2(g) + Cl2(g) In an experiment...
A: Given reaction is followed first order kinetics
Q: How many secondary hydrogens are there in the following compound? > View Available Hint(s) O 4 Submi...
A:
Q: Bry - NaOH
A: The 3- hexanone react with NaoH and NaoH act base which abstract the proton which form enolate an...
Q: how you wo accomplish the ing syntheSIS. Indicate all necessary reagents, and show any intermediates...
A: Organic reaction mechanisms:
Q: g age Rest reduction potentials.
A:
Q: What are the
A: Solute can be defined as a substance that get dissolved into solvent. For example-, sugar solute ge...
Q: Cyclobutane, C4H8, consisting of molecules in which four carbon atoms for a ring, decomposes when he...
A: Given reaction is followed first order kinetics
Q: Unit cell) calculate atomic radius in picometers; not sure how to get volume or side length if I eve...
A:
Q: 8. From the given starting material provide a reasonable synthetic route to the following molecule. ...
A: Organic reaction mechanisms:
Q: Sodium hydrogen carbonate (NaHCO,), also known as sodium bicarbonate or "baking soda", can be used t...
A:
Q: rate law
A:
Q: Which route would be better: one that produces a small amount of hazardous waste or one that produce...
A: The correct answer is given below
Q: atoms
A:
Q: A 0.478 g sample of an unknown strong base is dissolved in 50.0 mL of water and titrated with a 0.13...
A: When acid and base combine with each other then they form salt and water. The reaction is called Neu...
Q: Question 10 Consider placing a piece of iron, Fes, into a 1 M solution of cadmium ion, Cd2*(ag). Sel...
A: The true statements are given below
Q: K does depend on the concentrations or partial pressures of reaction components.
A: Given statement :- K does depend on the concentrations or partial pressures of reaction components ...
Q: If the concentration of [OH-] in a solution is 10-7 M, the pH of the solution is ________, and there...
A:
Q: A compound contained 4.6g Na, 2.8g N, 9.6g O. Find the empirical formula. (RAM data Na = 22.99, N = ...
A: In empirical formula problem first we determine mole ratio of given atom
Q: A 12.345-g sample of a solid mixture, containing both sodium hydroxide (NaOH) and sodium hydrogen ca...
A: For the produced CO2 gas, Volume, V = 1.171 L Temperature, T = 23.3 °C = (...
Q: Which of the following are typical impurities in active pharmaceutical ingredients? I. intermediate...
A: An impurity in pharmaceuticals is classified as any component that is not the entity defined as the ...
Q: В. 0.22
A:
Q: 4. The table below shows the standard enthalpy of formation of carbon dioxide, water and nitromethan...
A:
Q: hát was the funclion of the anhydrous calcium chloride? Illustrate this function by completing this ...
A: Extraction is the method here to get your compound that is in aqueous medium with the help of an org...
Q: What is/are the productis) of the following acid-base mechanism? (CHa),NH OCH, + (CH,NH Li OH (CH3)N...
A: In an organic reaction mechanism, a curved arrow represents the transfer of electrons. In an organic...
Q: Consider the solubilities of a particular solute at two different temperatures. Temperature (C) Solu...
A:
Q: 1. An ethanol solution is prepared by dissolving 10 ml of ethanol (C,H;OH) (density: 0.800 g/ml) in ...
A: calculation of concentration
Q: Chromium is electroplated industrially by the electrolysis of solutions of K2Cr2O7. How much time wo...
A: Time taken to deposit 1 Kg of Cr is 55677.36 sec or 15.4659 hr.
Q: The following potential diagram summarizes the results of electrochemical studies of the aqueous sol...
A: The reduction potential of ClO4- to Cl2 will be the change of total reduction potential of every st...
Q: I need help solving for a reactant in solution.
A:
Q: How will you know which ions to include in the net ionic equation of NaCl(ag)+ AgN03(aq) -> AgCI(s) ...
A:
Q: at percentage of the c
A:
Q: Enter a molecular equation for the precipitation reaction that occurs (if any) when each pair of aqu...
A:
Q: Calculate the pH of a solution that is 1.8*10^-3 M morphine if the pKa of its conjugate acid is 8.21
A: We have to predict the pH of the morphine solution.
Q: How many grams of CO are produced when 2.50 grams of heptane is reacted with excess oxygen? Equation...
A:
Q: e of Kp for t
A:
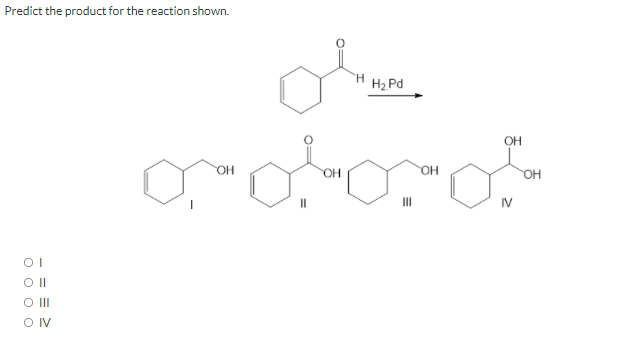
Step by step
Solved in 2 steps with 2 images

- The Br produced by the reaction in Figure 9.2 could combine with the carbocation you drew to give a dibromide product (a product with two Br atoms). Use a curved arrow to show the Br making a bond to the carbocation, and draw the resulting dibromide product.Which product is the final product in the reaction below, shown as numbered?Predict the main product of the reaction in Figure 10. * A B C D The main product of the reaction cannot be predicted.



