@ Prepare a "pH curve" as follows: a) Find the pH of the acid solution just before the end point when 0.1 ml of base less than the end point voiume has been added. potnt, therefore at the end of the titration ph = 7. We are assuming that end point equals equivalence b) Repeat calculation 'a' for a volume 1 ml less and 5 ml less then end point and before any base is added.(TThat is Oml base added. I Before end potnt, we assume that all the base is used up by the acid. c) Assuming you had continued the titration past the end point, calculate the pH when 0.1 ml of base beyond end point had been added. d) Repeat calculation 'c' for 1 ml, S ml and 10 ml of base beyond end point. Show all calculations and record your results in a table similar to the one shown below: excess H+ excess OH END 0 -5 -1 +5 +10 -.1 PT +.1 +1 (ML) BASE VOLUME O 20 24 24-9 25 25.26 30| 35 _pH
@ Prepare a "pH curve" as follows: a) Find the pH of the acid solution just before the end point when 0.1 ml of base less than the end point voiume has been added. potnt, therefore at the end of the titration ph = 7. We are assuming that end point equals equivalence b) Repeat calculation 'a' for a volume 1 ml less and 5 ml less then end point and before any base is added.(TThat is Oml base added. I Before end potnt, we assume that all the base is used up by the acid. c) Assuming you had continued the titration past the end point, calculate the pH when 0.1 ml of base beyond end point had been added. d) Repeat calculation 'c' for 1 ml, S ml and 10 ml of base beyond end point. Show all calculations and record your results in a table similar to the one shown below: excess H+ excess OH END 0 -5 -1 +5 +10 -.1 PT +.1 +1 (ML) BASE VOLUME O 20 24 24-9 25 25.26 30| 35 _pH
Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter15: Acid-base Equilibria
Section: Chapter Questions
Problem 132IP: A 10.00-g sample of the ionic compound NaA, where A is the anion of a weak acid, was dissolved in...
Related questions
Question
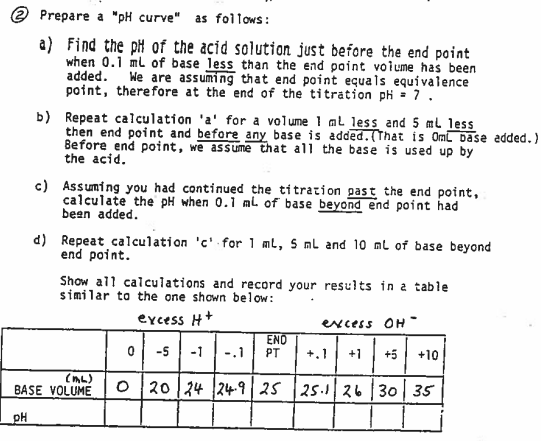
Transcribed Image Text:@ Prepare a "pH curve" as follows:
a) Find the pH of the acid solution just before the end point
when 0.1 ml of base less than the end point voiume has been
added.
potnt, therefore at the end of the titration ph - 7.
We are assuming that end point equals equivalence
b) Repeat calculation 'a' for a volume 1 ml less and 5 ml less
then end point and before any base is added. That is Oml base added.)
Before end point, we assume that all the base is used up by
the acid.
c) Assuming you had continued the titration past the end point,
calculate the pH when 0.1 ml of base beyond end point had
been added.
d) Repeat calculation 'c' for 1 ml, S ml and 10 ml of base beyond
end point.
Show all calculations and record your results in a table
similar ta the one shown below:
excess H+
excess OH
0 -5 -1
END
PT
-.1
+.1
+1
+5
+10
(ML)
BASE VOLUME
20 24 24-9 25 25.1|26 30 35
pH
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps with 5 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning



Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning
