Q: Indicate the reagent(s) needed to carry out the following transformations. a) он OTs b) но
A: NOTE : In the 2nd reaction, the H atom is present in the product attached to the carbon making…
Q: Msing protecting group Arategy. propose Compound shown below You should wse stra a synthes is for…
A: Given starting material: 3- Bromo-cyclohexanol This is basically done upon the retrosynthesis…
Q: For the following Schemes, provide the missing compounds, reagents, and conditions to complete cach…
A: In this reaction, first, ketone should be reduced to alcohol using a suitable reducing agent. Then,…
Q: which reagents uould gide rise to the following trens formatians? (HO (HO
A:
Q: Give the alkyl halide and appopriatle recgents thatwould lead to This product, using an E2 reacton.…
A: E2 elimination is a single step elimination reaction, in which base abstracts acidic hydrogen and…
Q: Prepare the following showing the stepwise mechanism: .COOET NH, N. H.
A: The Amine Shown here is an active nucleophile which attacks on carbonyl carbon and then Gradually…
Q: Draw the major product(s) when the followiịng molecule is reacted with excess NaOEt in EtOH,…
A:
Q: Но OH NH2 H2N 210 °C
A:
Q: Synthesize B from A keeping stereochemistry in mind: N 111 A у ome HC
A: In this question, we will synthesized B from starting material A and also keeping stereochemistry in…
Q: When reacted with methanol and an organic acid, dihydopyran yields one major product. Draw a…
A: Since the alkene is nuclophilic. Hence it takes the proton from the solution and form carbocation.…
Q: (e) (p) ()) "HIVIT MgBr (q) (e) 9SH nucleophiles in a-e. Problem 17.25: Draw the major product for…
A: a) CH3SNa Methanethiol , generate a soft nucleophile ,center at sulfur atom. Soft nucleophile will…
Q: Rank the following alkyl halides in order of increasing SyI reactivity. Br Br B: Br C: А: A B C…
A: In SN1 reaction formation of carbonation takes place so the reactivity of that alkyl or Aryl halide…
Q: Which isomer of 1-bromo-2-tert-butylcyclohexane reacts faster when refluxed with potassium…
A:
Q: Select the expected product of reacton shown. NaCN HeN
A:
Q: the and retrosynthesis corresponding forward reaction for the following compounds : OH 3. (a)…
A: Retrosynthesis: It is the phenomenon of breaking down the specific molecule into immediately present…
Q: identity the reagents necessary to achienve the trandormation below. You will need to une at ntane…
A:
Q: What is(are) the most appropriate reagent(s) for the following reaction: NH2 NO2 NO2
A:
Q: HBr ? peroxides
A: The addition of HBr in presence of peroxides takes place according to anti Markovnikov's rule. It…
Q: OB O C O A A. OD Which of the following is the most likely structure of the following cation after…
A: The stability of carbocation is : 3° > 2° > 1° 3° carbocation means the the carbon atom at…
Q: 2. Which of the following would give a meta product when submitted to EAS reaction conditions? OA OB…
A: The solution is given below -
Q: 18.65 Propose a plausible synthesis for the following trans- formation. CH3 CH3 CH3
A:
Q: (b) How can be prepared from OEt CO,Et and at EtO OEt EtO2C 95 °C ? Provide the mechanism.
A:
Q: Attached compounds undergoes E2 elimination with strongbase? For compounds that undergo elimination,…
A:
Q: Some of the following oolemles Can be made using 2 Cansecustire electrophilic aromatic…
A: The solution is given below -
Q: Br. O,N
A: Alcl3 is used for generation of electrophile.
Q: (e) Provide a reasonable mechanism of the following reaction .CI OCH3 CH3ONA Ether (5) (c) Show how…
A:
Q: Choose which compound is more reactive when reacted with NaOH in water solvent and ne reason…
A: The compounds given are carboxylic acids. And the reaction is taking place with base NaOH.
Q: Draw the structure of compounds A–E in the following reaction scheme.
A: The given reaction is shown below. In order to obtain the structure of all the compounds, the…
Q: In the reaction between 1-methylcyclohexanol with hydrogen bromide to form 1-bromo-…
A: SN1 reaction :- Unimolecular nucleophilic substitution (SN1) reaction are go via carbocation…
Q: Provide the reagents necessary to carry out the following conversiom CH,NH2
A: This reaction is example of multistep synthesis. It involve Friedle Craft acylation, Friedle Craft…
Q: Synthesize the following compound from the given starting materials. OEt
A:
Q: B) Provide the minimum reagents required for the following syntheses below. Me. Me. OMe HO,
A: In the given reaction 1, ester undergoes hydrolysis to produce carboxylic acid in the presence of…
Q: 11. OMe Me
A:
Q: Select the expected Major product of the reaction Shawn Jelect the expected product of the tollowing…
A: 1) First carbonyl group protonated by the gain of H+ and then nucleophile amino group attack on…
Q: Og Meas
A:
Q: Resveratrol is an antioxidant found in the skin of red grapes. Itsanticancer, anti-inflammatory, and…
A:
Q: Wnte reaction schemes (no mechanism required) to show how the followin transformations can be…
A: In order to solve this question, we will use reactions like aldol condensation, reduction using…
Q: Drawthe MAJOR product(s) of the followingreaction. 1) O3 2) DMS 3) excess LAH 4) H2O 1. 1) Hg(OAc)2.…
A:
Q: alkene that, upon ozonolysis reaction, gives only H. H' Select one:
A: In this question we will Identify the starting material Alkene which give di-aldehyde Product. You…
Q: edict the major product for the following reaction, by proposing a plausible stepwise mechanism.…
A: In this question we want to draw major product with mechanism. You can see the major product in…
Q: AICI . (A), Product (A) is :- whynet Ph HO (B) HO (C) (D)
A:
Q: a) CI H b) .N.
A: Concept is based on aromatic electrophilic substitution reactions.
Q: Identify the major product. Assume excess of all reagents as appropriate. HO. NazCr2O7 H2SO4
A:
Q: 1.2 The reagent, (MO)2P=O(CH₂CO₂Me), resultant species is then reacted with the following compound:…
A: It is the modification reaction of wittig reaction which is known as Horner Wordsworth Emmons…
Q: What reagents are needed to prepare CH;CH,CH,CH,C=CH from CH3(CH,);CH=CH,? Select all that apply.…
A: This is an example of conversion of alkene to alkyne. This can be accomplished by halogenation…
Q: raw the expected major product for both compounds A and B of the following reaction uence HCI A
A: Major product is analyse by applying appropriate reaction mechanism on substrate.
Q: O https://ovg.cengagenow.com/iln/t Draw the structure(s) of the major organic product(s) of the…
A: The reaction taking place is given as,
Q: Whar change would you expect on the rate of the S2 reaction eof 1-lodo 2methybatane with cyanide…
A: The SN1 means unimolecular reaction and SN2 means bimolecular reaction. The rate of unimolecular…
Q: 18.65 Propose a plausible synthesis for the following trans- formation. CH3 CH3 CH₂
A:
Q: 17) (1) Obtain major product(s) for all electrophilic are HNO (A) H₂SO4 (B) (C) O₂N CO₂Me SO 3 H₂SC…
A: In the given question we have to do the following reaction.
Step by step
Solved in 2 steps with 2 images

- Select the major product for the reaction below.From the table of available reagents select the one(s) you would use to accomplish the transformations shown below.Which reagent would you use to accomplish each of the following transformations? Options (please type exactly as shown and chose the most relevant reagent): H2O BH3-H2O2 KMnO4-NaOH O3 H2O2 O3-Me2S MCPBA-H3O+ H2O-H2SO4 OsO4-H2O2 Reagent 1: Reagent 2:
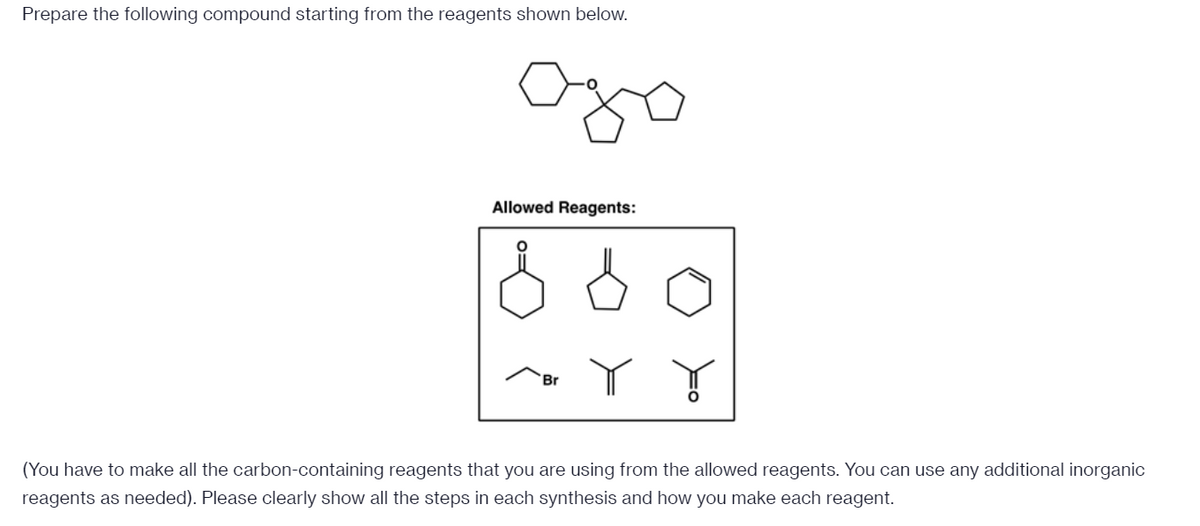
- Synthesize the following compounds from the given starting material. You can add on any alkyl/ alkyl halide under 8 carbons or any necessary inorganic reagent needed (this includes triphenyl phosphine (Ph3P). Please draw all intermediates and reagents necessary to get to the productWhich reagent would you use to accomplish each of the following transformations? Options (please type exactly as shown and chose the most relevant reagent): O3 MCPBA-H3O+ OsO4-H2O2 ROOR CH3CO3H H2O-H2SO4 O2-DMS KMnO4-NaOH O3-Me2S Reagent 1: Reagent 2:Show how to carry out the following transformation. Show all reagents and all molecules synthesized along the way. You do not have to use curved arrows. You are not allowed to use NBS/light or Br2/light. You MUST use SOCl2/pyr. for this drawing
- Choose the best reagents to complete the reaction shown below.Choose the best reagents to complete the following reaction. There's another answer choice not shown on screen: E) 1. CH3Li 2. H3O+ Hint: The correct answer is not option C.identify the reagents necessary to complete this synthesis. be sure to draw the product(s) for each step of your synthesis
- Select the necessary reagent(s) for the following transformation. a. I2 b. H2/Pt c. H+/H2O d. NaOH e. K2Cr2O7 f. Cu^2+Draw the major organic products for each reaction below.What is the best way to prepare the following ethers using an alkyl halide and an alkoxide ion?a. CH3CH2CHOCH2CH2CH3CH3 c. OCH3 b. CH3CH2OCH2CHCH2CH2CH3CH3 d. CH2O

