Propose the structure that corresponds to the following BC NMR spectra 13C-NMR, CDCI; Solvent, Molecular Formula: C5H1002 DEPT-90 CH DEPT-135 CH, CH they are positive CH2 es is negative broadband decoupled C, CH, CH2, CH3 ppm 170 160 150 140 130 120 110 100 90 80 70 60 50 40 30 20
Propose the structure that corresponds to the following BC NMR spectra 13C-NMR, CDCI; Solvent, Molecular Formula: C5H1002 DEPT-90 CH DEPT-135 CH, CH they are positive CH2 es is negative broadband decoupled C, CH, CH2, CH3 ppm 170 160 150 140 130 120 110 100 90 80 70 60 50 40 30 20
Chapter13: Structure Determination: Nuclear Magnetic Resonance Spectroscopy
Section13.SE: Something Extra
Problem 59GP: The mass spectrum and 13C NMR spectrum of a hydrocarbon are shown. Propose a structure for this...
Related questions
Question
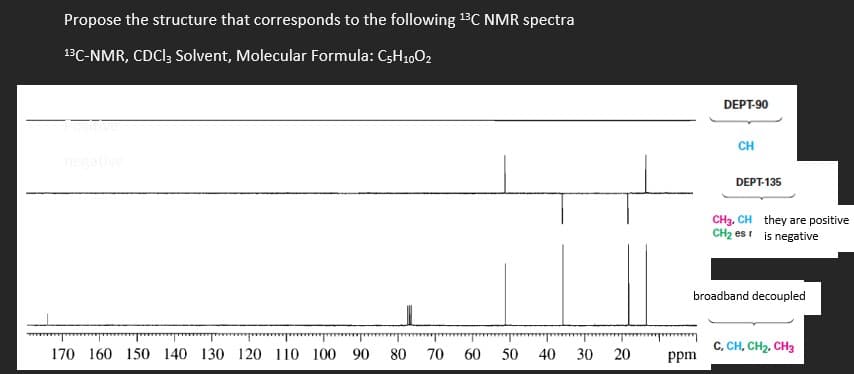
Transcribed Image Text:Propose the structure that corresponds to the following 13C NMR spectra
13C-NMR, CDCI; Solvent, Molecular Formula: C3H1,02
DEPT-90
CH
negative
DEPT-135
CH3, CH they are positive
CH2 esi is negative
broadband decoupled
C, CH, CH2, CH3
170 160 150 140 130 120 110 100 90
80
70
60
50 40
30
20
ppm
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

