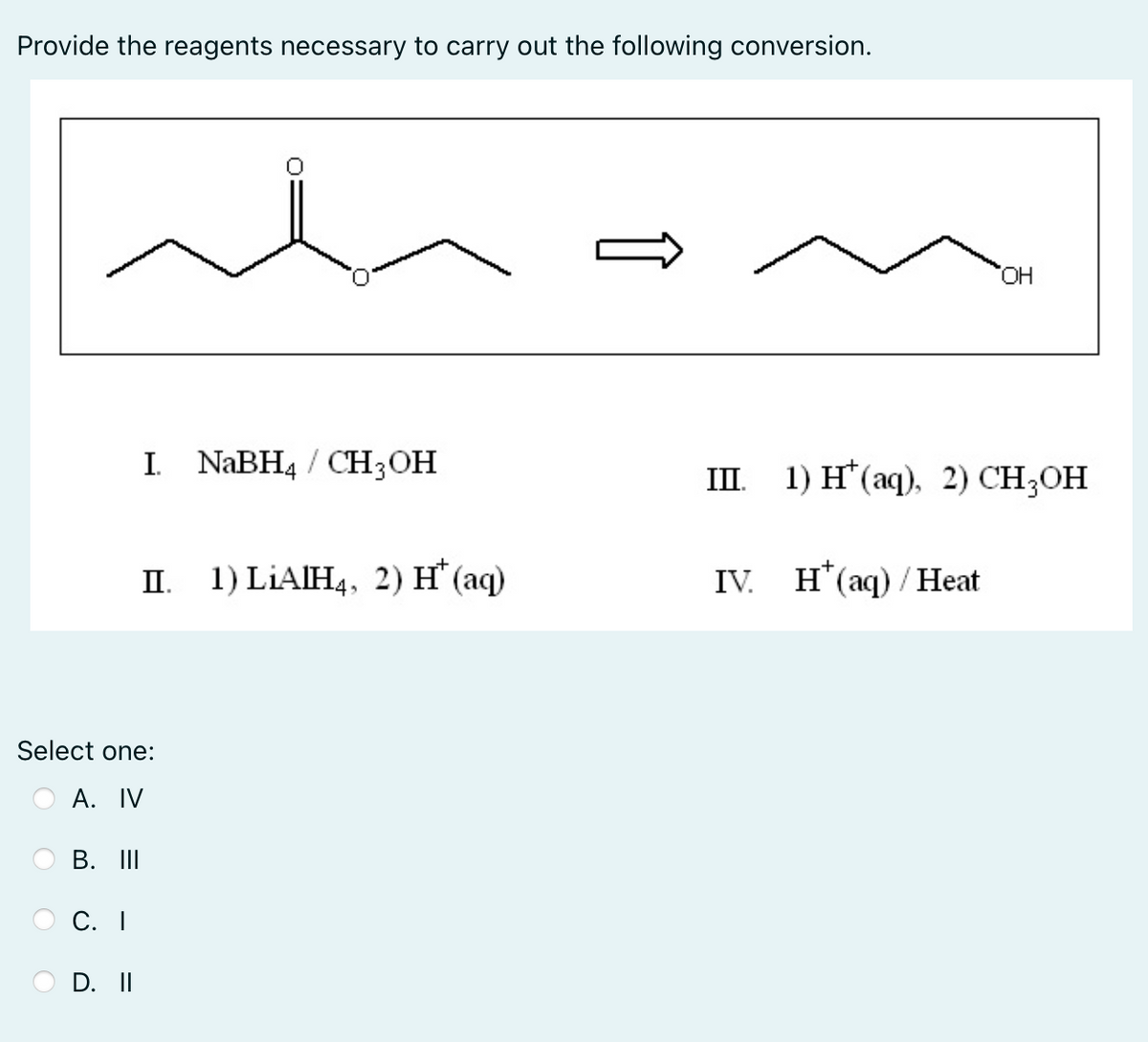
Provide the reagents necessary to carry out the following conversion. it I. NaBH4 / CH3OH II. Select one: A. IV B. III C. I D. II 1) LIAIH4, 2) H* (aq) OH III. 1) H(aq), 2) CH3OH IV. H(aq)/Heat
Q: Given the following equation: 2 C4H10 + 13 0₂ → 8 CO₂ + 10 H₂O How many grams of C4H₁0 are needed to…
A:
Q: 4) The percent moisture content of the following results weight of crucible and wet sample is…
A:
Q: Consider a galvanic cell with a cadmium electrode immersed in 1.0 M Cd2+ (aq) and a lead electrode…
A:
Q: Calculate [H3PO4], [H₂PO4], [HPO4²-], [PO4³-], [H3O+], [OH-], and [CI-] in a mixture that is…
A: H3PO4 is a triprotic weak acid with pKa values of 2.12, 7.21, and 12.32. Concentration of HCl =…
Q: A flashlight dims. Curious, Marinette removes the alkaline battery where two electrons are…
A: The solution is given below -
Q: The total energy eigenfunctions (called orbitals) for the hydrogen atom contain three quantum…
A: Given that, the total energy eigenvalue is En = -e28πε0a0n2, where n is the principal quantum…
Q: Calculate the value of K at 25°C for the reaction: 5Fe²+ (aq) + MnO (aq) +8H+ (aq) 4 Use the…
A: The equilibrium constant, K is related to the standard cell potential, E°cell by the following…
Q: What is the organic product formed in the following sequence of reactions? Br (1) Mg/ether, (2) CO₂…
A: The three step synthesis is shown in the following step.
Q: 3 points Save Answer Consider the following reaction with vanadium and iron: 2 Fe²+ (aq) + √²+ (aq)…
A: Fe3+(aq) + e- -----> Fe2+. E° = +0.77 V V2+. + 2e- -----> V E° = -0.49 V
Q: Calculate the % of Cd(OH)4² among all the cadmium complexes below in a pH 13 solution saturated with…
A:
Q: Calculate the value of the free energy change, ΔG, for the reaction below at 600.0ºC when the…
A:
Q: When 1.3 g of C4H9NO is dissolved in 150 mL water, the resulting solution is 0.50% ionized at…
A: C4H9NO is a weak base. Let us discuss its chemistry.
Q: 6. Assume you have 1kg of an alloy consisting of 85 wt% Sn and 15 wt% Pb at a temperature of 184 °C.…
A: To find out the mass of Sn needed to be added to 1 kg of alloy consisting of 85 % of Sn and 15 % of…
Q: Q1/ Determine the heating area required for the production of 1000 lb/hr of 50% from 10% NaOH feed…
A: given data production rate F=10000 lb / hr = 4535.92 kg/hr concentration of feed xf=10% NaOH…
Q: what is dynamic equilibrium?
A: Chemical equilibrium: The chemical equilibrium is the state in which the both the reactants and…
Q: a. b. Br Br MeO OMe
A:
Q: ga Question 12 How many milliliters of 0.246 M HNO3 should be added to 213 mL of 0.006 66 M…
A:
Q: Why does ethane prefer the staggered conformer over the eclipsing conformer? Ligands must be as far…
A: Ethane molecule exist in two conformations namely eclipsed and anti or staggered conformation. This…
Q: An injection of amphotericin B contains 50mg/10ml. What volume must be added to 500ml of normal…
A:
Q: Performing control experience in determining nitrogen using the Keldal method Choose one answer: a.…
A: Solutions- Performing control experience in determining nitrogen using the Keldal method. The…
Q: For the reaction below, A+2B → AB2 if AG = -19.9 kJ.mol-1, what is the value of K at 298 K ?
A:
Q: Discuss Spontaniety and give examples.
A: Spontaneous reaction is known as spontaneity. Here we have to define spontaneous reaction with…
Q: (b) Write an expression of the vibrational partition function. Obtain the relation of the…
A: The answers are given below:
Q: Instruction: Show your solutions. 1. A 0.917 g sample of canned tuna was analyzed by the Kjeldahl…
A: Given: The mass of the sample = 0.917 g In the Kjeldahl method, a volume of 20.59 mL of 0.1249 M HCl…
Q: Br H O
A: alkyl halide with bulky base will give less substituted alkene alkene undergo hydroboration will…
Q: for this question. The equilibrium constant, K, for the following reaction is 1.20x 10-2 at 500 K.…
A:
Q: How many millimoles of solute are contained in a) 250.0 mL of 0.0423 M KSCN? b) 500.0 mL of a…
A: Given Multiple question here.
Q: Given the information A+B 2D CID calculate AG at 298 K for the reaction A+B 2C AH=740.5 kJ AH"=383.0…
A:
Q: Two students were performing an experiment of the solubility equilibria of Ca(OH)2 which is…
A: GIVEN Experiment: The solubility equilibria of Ca(OH)2 which is exothermic by titrating it with HCl…
Q: Arrange the following electron rich species in the increasing order of their stability with an…
A: Electron withdrawing group are those which decreases the electron density and electron releasing…
Q: ou!- complete the following reaction ? MaSH 9 <CO₂EE LIAIHA H NIH 200² (HN) ANTH thot
A:
Q: How much water can you extract from 1.00 L of 0.35 M solution of sugar at 40 C by exerting a…
A:
Q: 1. Name the chemical and physical characteristics of polymers.
A: 1. Chemical characteristics of polymers; a. Reactivity and bonding: Polymers have covalent bonding…
Q: Jaya needs to measure the unknown concentration of Sn2+. A Galvanic cell is set up in a room with a…
A:
Q: Molar Mass NaHCO3 84.01 g/mol. Molar Mass Na2CO3 105.99 g/ moo. A 1.375- gram sample of baking soda…
A:
Q: The pOH of this solution is
A:
Q: Write the complete stepwise mechanism for each of the following reactions. Show all electron flow…
A:
Q: A galvanic cell is set up with iron and aluminum, according to the unbalanced reaction below. If the…
A:
Q: Calculate the volume of 0.0602 M EDTA needed to titrate 11.63 mL of 0.0725 M Mg(NO3)2.
A:
Q: What is the coefficient for H* after balancing the following reaction in acidic medium? (Please note…
A:
Q: In the laboratory you are given the task of separating Ag+ and Co2+ ions in aqueous solution. For…
A: Here we are required to find to separate two cation Ag+ and Co2+ using various reagent
Q: Balance the following chemical equations and indicate the type of reaction taking place. If the…
A: we need to balance equation 3 and 4
Q: Find the oxidizing agent in the following balanced reaction (Please note that element X and Y are…
A: Detail description is given below
Q: Solid potassium sulfide is slowly added to 50.0 mL of a 0.0321 M silver nitrate solution. The…
A:
Q: (c) Define self-assembly in supramolecular chemistry. Explain the metal-directed self-assembly…
A: According to guidelines I will answer only one of those questions!!
Q: The Zn in a 0.6803-g sample of foot powder was titrated with 27.3 mL of 0.04246 M EDTA. Calculate…
A: To calculate the percentage of zinc in the foot powder sample, use the EDTA titration. First…
Q: (b) In the pH metric titration, calomel and glass electrodes are used for measuring. What are the…
A: The answers are given below as follows:
Q: Which compound has the element X with the oxidation number of +6. (select all that are correct…
A: Determination of oxidation number of various molecules.
Q: Which structure satisfies the octet rule for a compound made of generic element X? O:X-X: O :X=X:…
A: Octet rule states that the atom of an element wants to have 8 electrons in its valence shell.
Q: 2. 1. What does Le Chatelier's principle state about chemical equilibrium? A. If stress (changes in…
A: Le chatelier principle is used to determine the direction of reaction. It will tell the effect of…
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

- Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides. One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(CzHs)3). 3SnCl4 + 4Al(C2H5)3 → 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.160 L of SnCl4 (d= 2.226 g/mL) was treated with 0.346 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). What is the theoretical yield in this experiment (mass of tetraethylstannane, Sn(C2H5)4)? If 0.257L of tetraethylstannane (d= 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?For butenafine, develop a synthesis that starts with napthalene and benzene. (aromatic methyl groups can be converted to alkyl bromides using Br2/light, and aldehydes by heating the alkyl bromide in DMSO, eg. ArCH3 to ArCH2Br then to ArCHO).Explain wurtz reaction with suitable reaction.
- Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides.One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(C2H5)3). 3SnCl4 + 4Al(C2H5)3 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.230 L of SnCl4 (d = 2.226 g/mL) was treated with 0.396 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). If 0.335 L of tetraethylstannane (d = 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?Write out the reaction of Bromination of E-stilbene using chemdraw (ACS 1996 format). Include all reagents, products, solvents, reaction temperature and yield. Reagents used include glacial acetic acid, E-stilbene, and pyridinium perbromide, otherwise known as PHPB. Glacial acetic acid: 10mL Stilbene: 0.5g Perbromide: 1.0g Product yield (Precipitate) weight: 0.63gDescribe how the product is purified. 4,4'-DIBROMOBIPHENYL [Biphenyl, 4,4'-dibromo-] Submitted by Robert E. Buckles and Norris G. Wheeler1. Checked by R. S. Schreiber, Wm. Bradley Reid, Jr., and Robert W. Jackson. 1. Procedure In a 15-cm. evaporating dish is placed 15.4 g. (0.10 mole) of finely powdered biphenyl (Note 1). The dish is set on a porcelain rack in a 30-cm. desiccator with a 10-cm. evaporating dish under the rack containing 39 g. (12 ml., 0.24 mole) of bromine. The desiccator is closed, but a very small opening is provided for the escape of hydrogen bromide (Note 2). The biphenyl is left in contact with the bromine vapor for 8 hours (or overnight). The orange solid is then removed from the desiccator and allowed to stand in the air under a hood for at least 4 hours (Note 3). At this point, the product weighs about 30 g. and has a melting point in the neighborhood of 152°. The crude 4,4'-dibromobiphenyl is dissolved in 75 ml. of benzene, filtered, and cooled to 15°. The…
- Why do we need to allow the solution of KF reagent to stand overnight before standardizing? if copied from google please paraphraseII. Alcohols in Synthesis B.) How are alcohols used to make ethers in the Williamson ether synthesis? How do you design the synthesis of ethers using both acidic and basic Williamson ether synthesis strategies?The product of reaction between acetone and bromine (excess) with KOH excess is A. Ch2BrCOCh3 B. ChBr3 C.ChBr2COCH3 D.CBr3COCH3 E.Ch2BrCOCHBr2
- Please complete reactions in clear handwritten of all subpartsChemistry please provide the flow of electrons aswell!! thank you! using the starting material (on the left) to determine the sythetic route which will be the most reaosnable and effective to theres none, you have to start witth the begin products to get to finish needed by using the minmium reagents and reactions needed to get to the final productplease quickly thanks ! 3.Please write out the major reaction and side-reaction in the preparation of ter-butyl.chloride, and write out the key points to use separation funnel in this process.

