Q write mechansim Two reactions: (1) 1., H (1 equiv.) 2. a) LiAlH b) H2O work - up 3. H (final product only ) 1. 2. CH¸ MgBr 3. CH₂OH,H* Gwrite mechansim Two reactions: 0 ② CN 1. HO. OH H+ (1 equiv.) H 2. a) LiAlH4 b) H₂O work-up 3. H+ 1. HO (1 equiv.) H 2. CH3MgBr 3. CH3OH, H+ OH, H+ (final product only) OCH3 (±)
Q write mechansim Two reactions: (1) 1., H (1 equiv.) 2. a) LiAlH b) H2O work - up 3. H (final product only ) 1. 2. CH¸ MgBr 3. CH₂OH,H* Gwrite mechansim Two reactions: 0 ② CN 1. HO. OH H+ (1 equiv.) H 2. a) LiAlH4 b) H₂O work-up 3. H+ 1. HO (1 equiv.) H 2. CH3MgBr 3. CH3OH, H+ OH, H+ (final product only) OCH3 (±)
Chapter30: Orbitals And Organic Chemistry: Pericyclic Reactions
Section30.SE: Something Extra
Problem 41AP: In light of your answer to Problem 30-40, explain why a mixture of products occurs in the following...
Related questions
Question
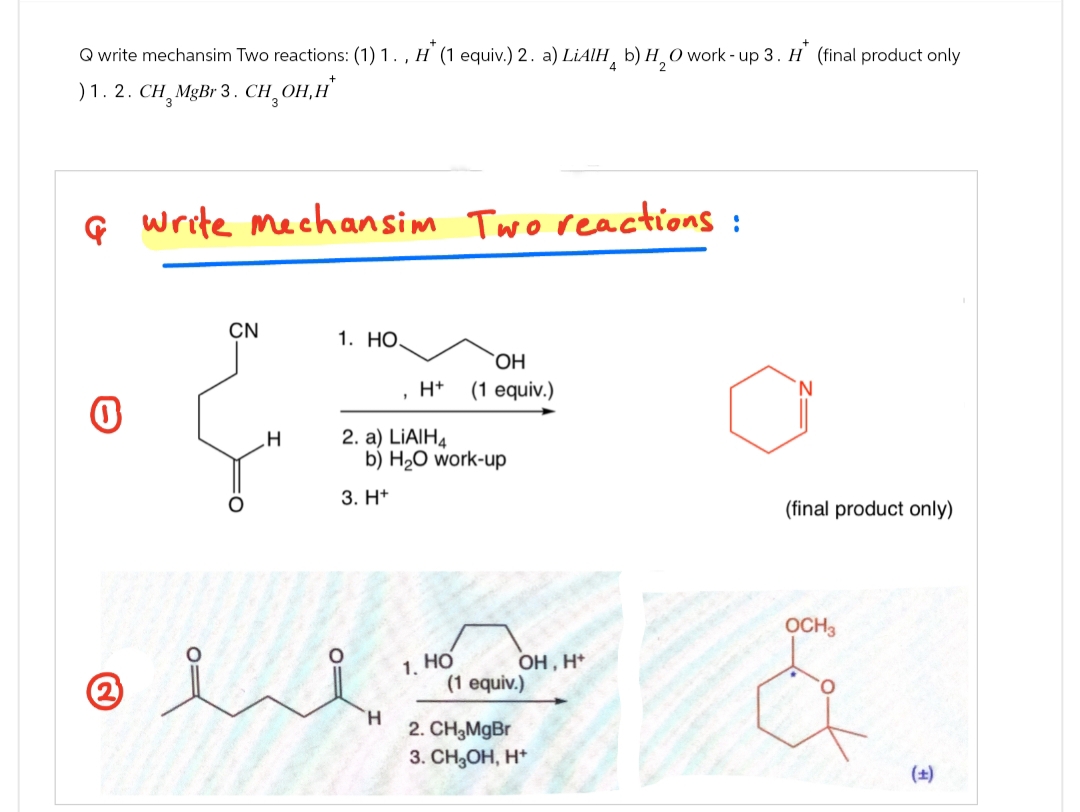
Transcribed Image Text:Q write mechansim Two reactions: (1) 1., H (1 equiv.) 2. a) LiAlH b) H2O work - up 3. H (final product only
) 1. 2. CH¸ MgBr 3. CH₂OH,H*
Gwrite mechansim Two reactions:
0
②
CN
1. HO.
OH
H+ (1 equiv.)
H
2. a) LiAlH4
b) H₂O work-up
3. H+
1. HO
(1 equiv.)
H
2. CH3MgBr
3. CH3OH, H+
OH, H+
(final product only)
OCH3
(±)
AI-Generated Solution
Unlock instant AI solutions
Tap the button
to generate a solution
Recommended textbooks for you


Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax


Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax