Question 15 of 17 What type of hybrid orbitals are utilized by carbon in anthracene? How many o bonds and a bonds are there in an anthraceie molecule? o bonds: n bonds: How many valence electrons occupy o-bond orbitals, and how many occupy r-bond orbitals? valence e in o-bond orbitals: valence e in n-bond orbitals: Publisher Unilersity Science Bod Question Source: McQuarrie, Rock, And Gallogly 4e General Chemsito
Question 15 of 17 What type of hybrid orbitals are utilized by carbon in anthracene? How many o bonds and a bonds are there in an anthraceie molecule? o bonds: n bonds: How many valence electrons occupy o-bond orbitals, and how many occupy r-bond orbitals? valence e in o-bond orbitals: valence e in n-bond orbitals: Publisher Unilersity Science Bod Question Source: McQuarrie, Rock, And Gallogly 4e General Chemsito
Chemistry by OpenStax (2015-05-04)
1st Edition
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Chapter7: Chemical Bonding And Molecular Geometry
Section: Chapter Questions
Problem 109E: Draw the Lewis electron dot structures for these molecules, including resonance structures where...
Related questions
Question
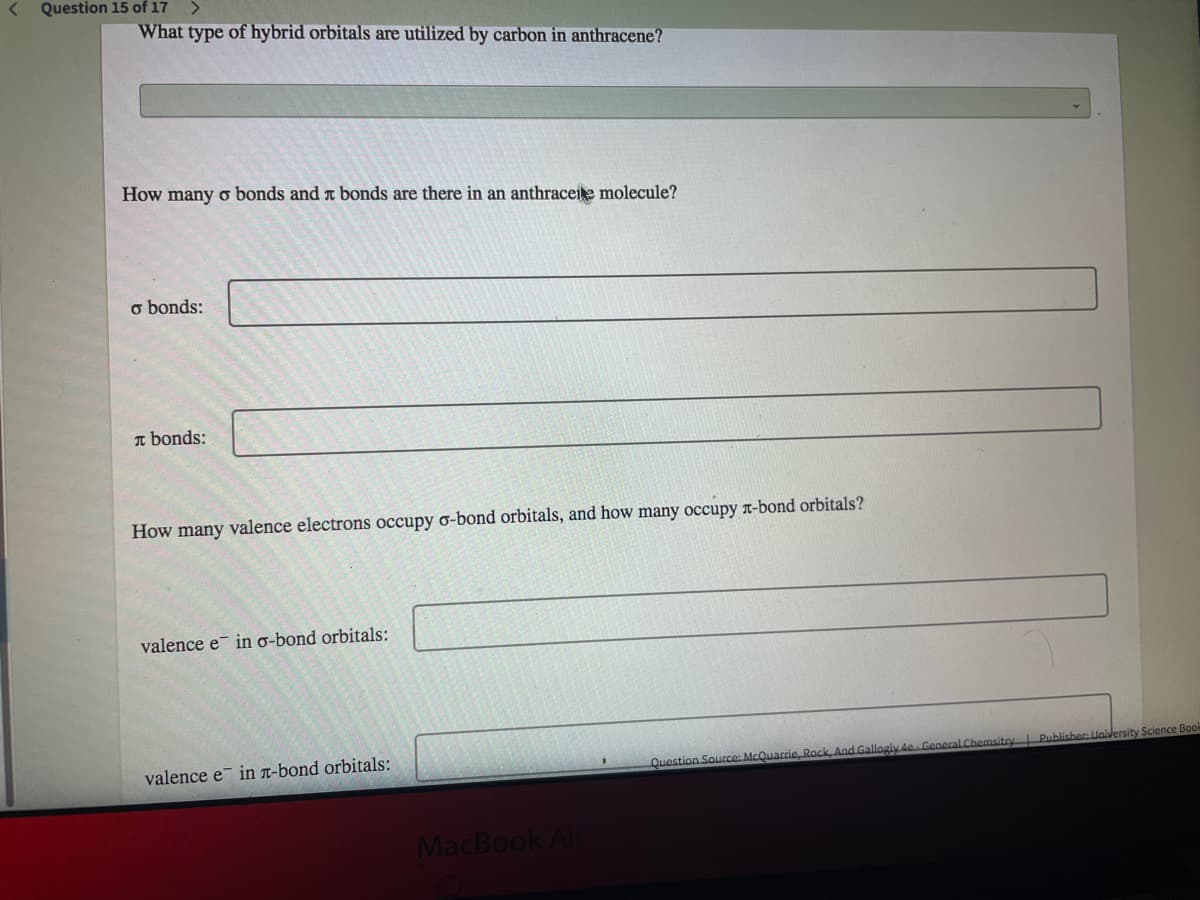
Transcribed Image Text:Question 15 of 17
What type of hybrid orbitals are utilized by carbon in anthracene?
How many o bonds and t bonds are there in an anthraceie molecule?
o bonds:
I bonds:
How many valence electrons occupy o-bond orbitals, and how many occupy r-bond orbitals?
valence e in o-bond orbitals:
valence e in n-bond orbitals:
Publisher University Science Book
Question Source: McQuarrie, Rock, And Gallogly 4e General Chemsito
MacBook Air
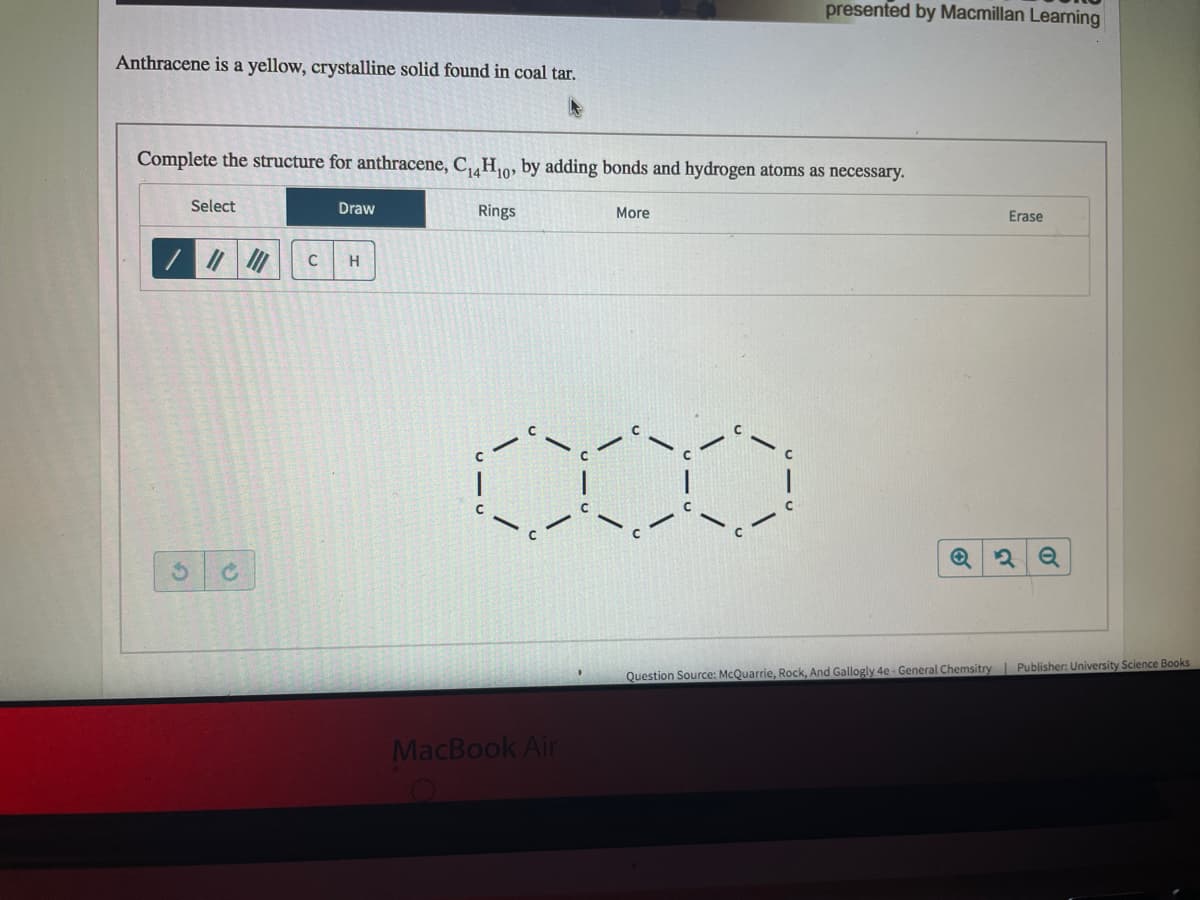
Transcribed Image Text:presented by Macmillan Leaming
Anthracene is a yellow, crystalline solid found in coal tar.
Complete the structure for anthracene, C,,H,o, by adding bonds and hydrogen atoms as necessary.
Select
Draw
Rings
More
Erase
C
H.
Question Source: McQuarrie, Rock, And Gallogly 4e - General Chemsitry Publisher: University Science Books
MacBook Air
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning