Q: Define the term SDS page.
A: The term SDS page has a full form Sodium dodecyl sulphate. The term belongs to electrophoresis.…
Q: In general, as you go across a period in the periodic table from left to right: (1) the atomic…
A: Given, 1) The atomic radius across in the periodic table from left to right is:
Q: 4. Given: H₂O 1 CH + 1 HNO3 → 1 CH₂NO₂ + 1 6 If a student wishes to make at least 6.66 x 10 24…
A: Avogadro's number = 6.022 × 1023 Number of molecules in 1 mol of a substance equals Avogadro's…
Q: X ⒸAE=AE²+2·3RTlog (ox)* (red) a MF mils/y₁ = 534 W DAT off + p
A:
Q: VARIANT 6 1. Calculate redox potential value for the system obtained by mixing 10 mL of K3 [Fe(CN)6]…
A: 10 mL of 0.01 mole/L K3FeCN6 is mixed with 10 mL of 0.05 mole/L K4FeCN6 solution. The standard…
Q: (d) 1. Hg(OAc)2, H2 2. NaBH4
A: It is elimination addition reaction which follows Markovnikoff rule. So, OH group is added to more…
Q: 2 H3PO4(s) --> 3 H2(g) + 2 P(s) + 4 O2(g) Using the reaction above, show step by step how many…
A: Given, 2 H3PO4(s) --> 3 H2(g) + 2 P(s) + 4 O2(g) mass of H3PO4 reacts = 0.7156 grams Volume of…
Q: 14. Which one of the following A) Mn²* and Ar B) Zn²+ and Cu²+ C) Na* and K pairs are isoelectronic?…
A:
Q: STARTING AMOUNT X A shop-vac is capable of pulling in air at a rate of 210 ft3/min. What is the rate…
A: We have to convert the rate in units of L/s
Q: Calculate the Binding energy in Joules/mol for Ni-58 (nuclear mass of Ni-58 = 57.93535 amu) (mass of…
A:
Q: In the upcoming chapters, we will learn a two-step method for achieving the following…
A: In this question, we will write the all reagents sequence ( which is used for the given…
Q: Predict the most likely mechanisms which occur when 2-iodo-2-methylhexane is heated in ethanol.
A:
Q: What is IUPAC name of this chiral molecule below? (Hint: include its chiral configuration in the…
A:
Q: True or False Cadmium ions precipitate as cadmium hydroxide with hydrogen sulfide
A: Analytical chemistry is branch of chemistry in which we deal with identification, purification and…
Q: Which is a major product of this reaction? DCH, .) CH-CH, OH o LCH CHỊCH, OH 1) CH₂CH₂MgBr 2) Н,0²…
A:
Q: 3) what do you mean by ion exchange capacity? Provide the functional groups present for weakly…
A:
Q: 13.How many protons and electrons are in one Br ion? A)35 p. 35e B)80 p. 81 e C)35 p. 34 e D)35p,…
A: In neutral element , number of protons and electrons are same but for ionic species number of…
Q: For the equilibrium reaction N2(g) + 3H2(g) « 2NH3(g) DH = -92 kJ, Describe how the equilibrium…
A:
Q: A system at constant pressure experiences the following: The thermal energy increases by a magnitude…
A: If energy is absorbed by the system, the sign will be positive. If energy released by the system,…
Q: Propose a detailed mechanism for the reaction below. لعلام , AICI3
A:
Q: The diagrams 1 to 3 below can be arranged to show the mechanism involved in a familiar reaction. The…
A:
Q: Case problem. Describe theoretical basis and procedure of Practical «The study of chemical…
A: Ferric ion and iodide reacts to form ferrous and Iodine. In pharmacy various physical or chemical…
Q: A chemist is studying the following equilibirum, which has the given equilibrium constant at a…
A:
Q: Does that mean that Average molarity of EDTA standard solution = Average molarity of EDTA? Will…
A: Recall the given reaction, Ca2+ aq + EDTA4- aq →CaEDTA2- aqWe have to…
Q: I III Which of the following fatty acids is an Omega-3 fatty acid? II IV A) Compound I B) Compound…
A: We have to select the Omega-3 fatty acid
Q: 28. What is the coefficient of H₂O when the following equation is properly balanced with smallest…
A:
Q: 20. The number of lone electron pairs in the N₂ molecule is A) 1 B) 2 C) 3 21.The Lewis structure…
A: Number of lone pair of N2 and Lewis structure of CS2
Q: 9) What will be the color and structure of methyl orange and phenolphthalein in highly acidic water…
A: The following indicators shows colour in acidic medium are as follow.
Q: 5. If a 7.0 mL sample of vinegar was titrated to the stoichiometric equivalence point with 7.5 mL of…
A:
Q: If Kp = 927 for the reaction below at 310.0 K, then what is the value of Kc?(R = 0.08314…
A: Given, 2A(g) + B(s) ⇌ 2C(s) + D(g) Kp = 927 Temperature (T) = 310.0 K Note: 1 bar = 0.987 atm…
Q: 1. Calculate the concentration (mol/L or M) of calcium ions in each water sample using the molar…
A: The balanced chemical equation for the reaction between Ca2+ and EDTA is shown below: Ca2+ +…
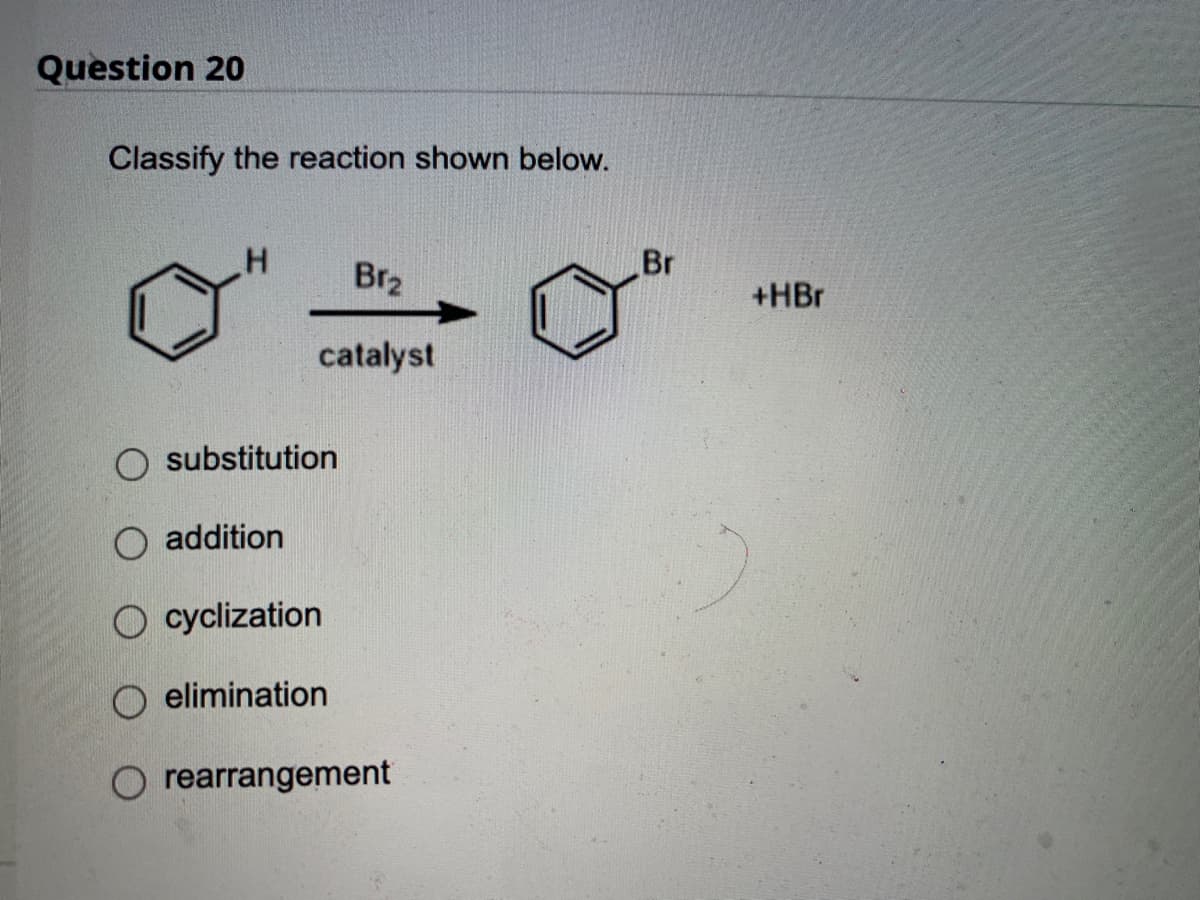
Q: ALKENE Br₂ Br H... H3C Br ""H CH3
A: This is actually bromination of alkene in antiposition.
Q: 1.1 Provide IUPAC names of the following aromatic compounds: OH Br Br 6
A: IUPAC is International Union of Pure and Applied Chemistry. This nomenclature is accepted all over…
Q: What is the product of this reaction? a) c) CH₂ MgCI CH3 CH3 1) CH₂MgCI 2) H₂O* b) d) LOH CH₂ LOH…
A:
Q: Two metals X and Y are mutually soluble in all proportions in the liquid state, but are completely…
A: We have find out the answer by drawing binary X-Y phase diagram.
Q: Considering the C-NMR below, choose the compound from the list provided that gave rise to the…
A: The carbons with different environment will give different signal in C-13 spectrum. This shows the…
Q: at is the correct order of decreasing reactivity ost reactive first) of these compounds toward…
A: When electron donating groups are attached to benzene ring.so, they increase the reactivity towards…
Q: For the question that follows, consider the following equation. 2Mg +02-2MgO The number of grams of…
A: According to Bartlby guidelines I solve only first question. Second question post separately.
Q: Report Sheet-Lab 4 Questions and Problems Q.3 A neutral atom has a mass number of 80 and has 45…
A: In chemistry, the general representation of an element or atom is given below: MzA Here, M is the…
Q: 1. Provide systematic names (assigning absolute stereochemistry if necessary) for the four molecules…
A:
Q: ata Table 1. Analysis of Alka-SelltzerⓇ Mass of acetic acid Density of acetic acid Concentration of…
A:
Q: Which of the following reaction conditions results in Markovnikov addition to an unsymmetric alkene?…
A:
Q: Which of the following is a tertiary amine? C) CH, N H CH₂ N CH, H₂C b) NH₂ CH, CH, H C Ф НС-С -N…
A:
Q: 4. Which of the following is not a correct description of 16.0 grams of methane, CH₂? A. It is one…
A: The one mole of compund or molecule is equal to the molecular mass of that compund or molecule .…
Q: I OH CH2OH Н OH Н OH I Н ОН Н
A: Alpha anomer Hydroxyl group at the anomeric carbon is cis to the exocyclic oxygen. Beta anomer…
Q: Provide the correct common name for the compound shown here. CH3 para- 2- 4- Br ortho- meta- 1- 2,4-…
A: The common name of a compound will have the substituent at the first position, and the other…
Q: ouTube Maps 4 6 5 CH2OH ОН ОН ОН 3 The Haworth projection of D-mannose is shown. 1 Question 10 of 37…
A:
Q: 4. Write the product and the mechanism for reaction. Write the names of starting material and…
A:
Q: Question1 12 UI 12 Provide the correct IUPAC name for the compound shown here. SH
A: For IUPAC naming select the functional group & principle chain i.e longest possible carbon…
Q: Unknown Code Sample A Sample D Sample E Sample G Sample F Calculated Density using the Ruler…
A: Given , Unknown sample(s) : Sample A Sample D Sample E Sample F Sample G
Step by step
Solved in 2 steps with 2 images

- Are there any other possible products for the following reaction in catalytic H2SO4 and H2O?In the reaction (E) 2-methyl -2,4-hexadiene with hydrogen bromide at room temperature, two isomeric products are isolated. Draw the structures for these isomeric products. Which is the kinetic isomer and which is the thermodynamic isomer.The rate of hydration of 2-butene and 2-methylpropene differ by several orders of magnitude. Whcih alkene is more rapidly hydrated and why?
- Pls ignore the highlighted part __________ is reduced in thefollowing reaction: Cr2O72- + 6S2O32-+ 14H+→ 2Cr3+ + 3S4O62- + 7H2O[Rh(PPh3)3Cl] is a precatalyst that can be activated by dissociation of a phosphine ligand to form an active catalyst, B, which is used in the hydrogenation of alkenes. The active catalyst can then undergo oxidative addition in the presence of H2 to form complex C. Propene coordinates to C to form complex D, which then undergoes a 1,2- insertion step to form E. Reductive elimination of propane from E regenerates the active catalyst B. Draw complexes B – E and hence provide a full catalytic cycle, including the activation step, for the hydrogenation of propeneExplain your reason for the answer1.All the following parameters would affect the rate and kinetics of the reaction exceptA.Solvent mediumB.Reaction temperatureC.Reaction timeD.Geometry and structure of the alkyl halideE.pH
- Draw the mechanism fot his reaction. B = [IrCl(CO)(PPH3)2]Examine the reaction below. Write a mechanism for the reaction beforr answeringn the question. What reaction mechanism is responsible for the ring formation in the mechanism seauence? a) beta hydrate elimination b) oxidative addition c) reductive elimination d) transmetallation e) none of the aboveAnswr this question DDDDDDDDraw the structureeeeeee after eacccccccch step & giveeeeeeee aaaaaaaaaaaaaaaaaaa ssssssssssshorttttttttttt explanation of step 2.
- One possible way of determining the identity of an alkene, is to let itundergo an oxidative cleavage reaction in the presence of hot basicpotassium permanganate. You are given two containers said to containdifferent alkenes. Container A is marked as cis / trans‐2‐butene andcontainer B as 2‐methyl‐1‐butene. Explain by referring to the formation ofproducts, how you would verify the identity of the alkenes.A mixture propane and bromine is exposed to sunlight. The reaction equation is given below. ??3??2??3 + ??2 + ?? → ? + ? a. State the type of the reaction above. b. What is the function of sunlight in the reaction? c. Draw the structure of the monosubstituted products, A and B. identify the major products. d. Write the mechanism for the formation of the major product. e. What would happen if the mixture is kept in the dark at room temperature?Rank thr stability of the following sets of reaction intermediates.

