Question 21 The number of protons in a neutral atom is equal to the of the atom. O mass O atomic weight atomic number O mass number O valence electron number Question 22 Which of these statements about Bohr's model of the atom is incorrect? O Electrons fill lower energy orbits first. O The greater the quantum number the higher the energy of an orbit. O Electrons occupy orbits with certain radii that correspond to discrete energies. O Elements with the same number of electrons in their outer orbit undergo similar chemical reactions. O Electrons in the innermost energy orbit are called valence electrons. Question 23 Which of these is the correct chemical symbol for carbon? O C O Ca O Cb
Question 21 The number of protons in a neutral atom is equal to the of the atom. O mass O atomic weight atomic number O mass number O valence electron number Question 22 Which of these statements about Bohr's model of the atom is incorrect? O Electrons fill lower energy orbits first. O The greater the quantum number the higher the energy of an orbit. O Electrons occupy orbits with certain radii that correspond to discrete energies. O Elements with the same number of electrons in their outer orbit undergo similar chemical reactions. O Electrons in the innermost energy orbit are called valence electrons. Question 23 Which of these is the correct chemical symbol for carbon? O C O Ca O Cb
Introductory Chemistry: A Foundation
9th Edition
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Donald J. DeCoste
Chapter11: Modern Atomic Theory
Section: Chapter Questions
Problem 106AP: lement X, which has a valence shell configuration of ns2np4 , was isolated in a laboratory. Which of...
Related questions
Question
I'm having some trouble on my homework and could really use some help
See attached:
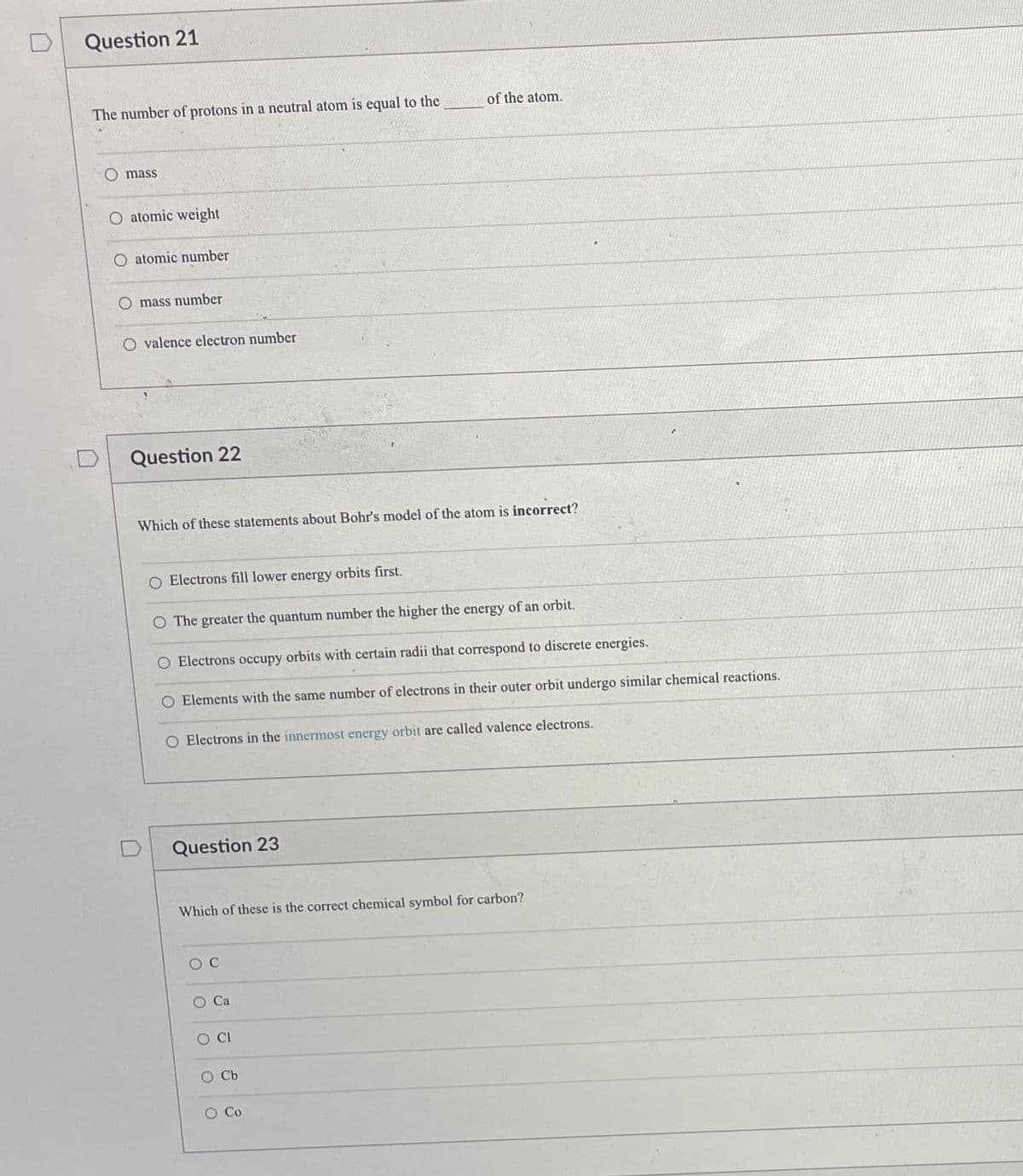
Transcribed Image Text:Question 21
The number of protons in a neutral atom is equal to the
of the atom.
mass
O atomic weight
O atomic number
mass number
O valence electron number
Question 22
Which of these statements about Bohr's model of the atom is incorrect?
O Electrons fill lower energy orbits first.
O The greater the quantum number the higher the energy of an orbit.
O Electrons occupy orbits with certain radii that correspond to discrete energies.
O Elements with the same number of electrons in their outer orbit undergo similar chemical reactions.
O Electrons in the innermost energy orbit are called valence electrons.
Question 23
Which of these is the correct chemical symbol for carbon?
O C
O Ca
CI
O Cb
O Co
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning