
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
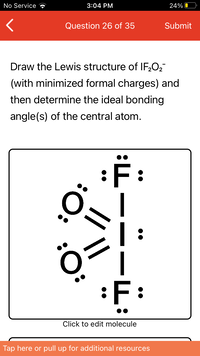
What have I done wrong here? And what is the bond angle?
Transcribed Image Text:No Service ?
3:04 PM
24% O
Question 26 of 35
Submit
Draw the Lewis structure of IF;O2
(with minimized formal charges) and
then determine the ideal bonding
angle(s) of the central atom.
F:
:F:
Click to edit molecule
Tap here or pull up for additional resources
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- What is the first thing you should do if you want to determine if a molecule is polar or not? Determine its hybridization. Draw the Lewis Structure. Draw the dipole arrows. Search for it on the internet.arrow_forward● X E X X View 1 View 2 Which one of your two Views is easier to understand? Explain why? How many different bond angles are there in this geometry. Hint, label each X with a number....e.g. X₁ through X5. A bond angle is then represented by any combination of XAX atoms.arrow_forward1. Briefly explain in your own words why the bond angle increases as the number of electron groups decrease? 2. How does electron domain geometry differ from molecular geometry? Briefly explain in your own words. 3. Write a complete list of steps you will utilize to predict the electron-domain geometry for a given species using only the Lewis structure. The only information you are provided with is the molecular formula and the net charge.arrow_forward
- Calculate the net molecular dipole of ammoniaarrow_forwardArrange the following in order of increasing polarity SF2, F2 and SCI2 O F2 < SF2 < SCI2 SF2 < F2 < SCI2 SF2 < SCI2 < F2 SCI2 < SF2 < F2 F2 < SCI2 < SF2arrow_forwardExplain why the CS2 molecule is linear while the H2S molecule is not linear.arrow_forward
- Why is the molecular geometry for water classified as bent and not linear?arrow_forwardWhat are the angles a and b in the actual molecule of which this is a Lewis structure? H H H C H a a = C b = ⁰ C H Note for advanced students: give the ideal angles, and don't worry about small differences from the ideal that might be caused by the fact that different electron groups may have slightly different sizes. H Xarrow_forwardis there any line structure explanation of this?arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY