QUESTIONS 1. For each field, which problem is being addressed by the consumer product? 2. What are some chemical components of the consumer products?
QUESTIONS 1. For each field, which problem is being addressed by the consumer product? 2. What are some chemical components of the consumer products?
Chapter3: Statistical Tests With Excel
Section: Chapter Questions
Problem 1P
Related questions
Question
For each field (field of medicine, transportation, communication, agriculture), which problem is being addressed by the consumer product?
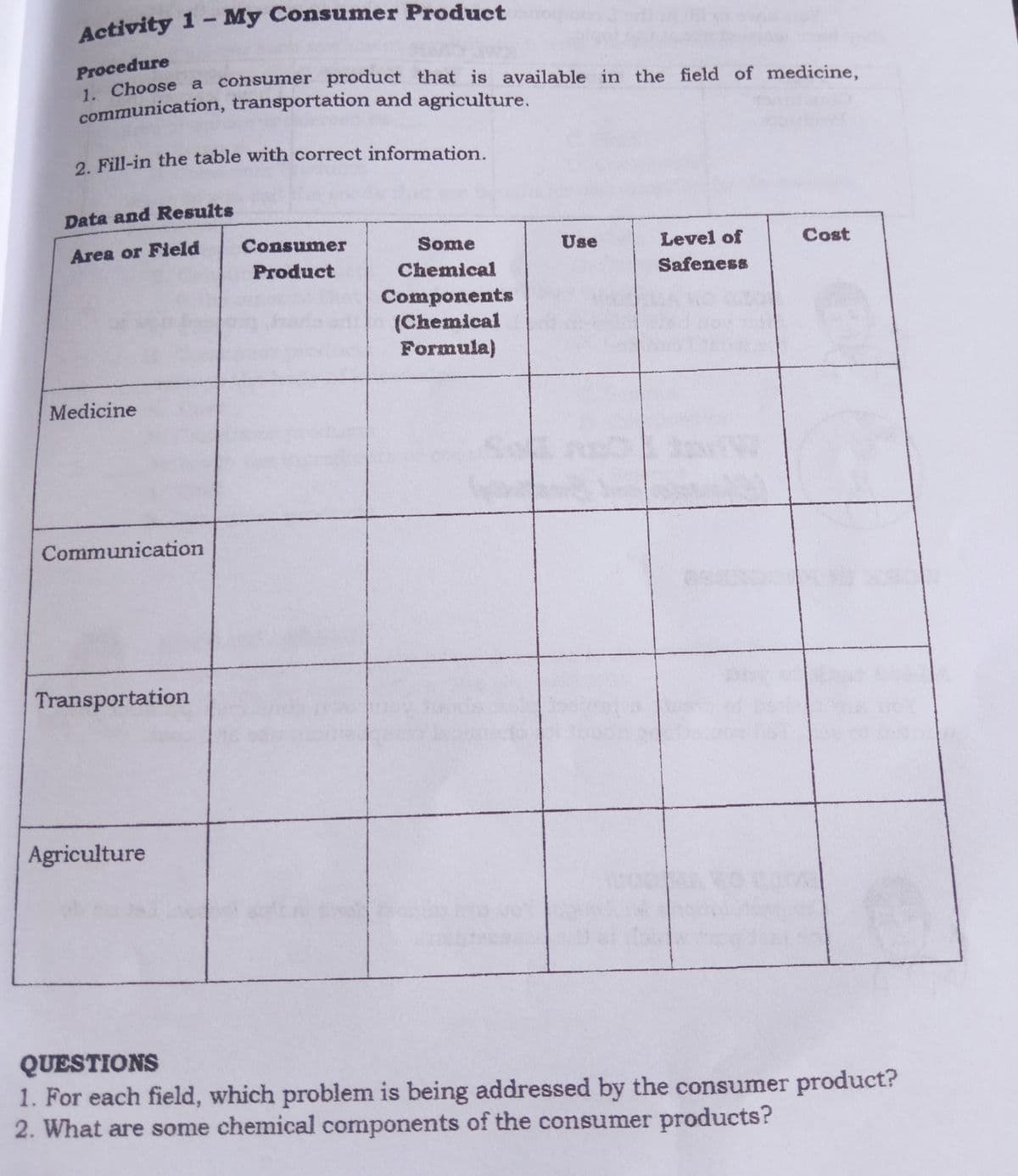
Transcribed Image Text:Activity 1- My Consumer Product
Procedure
Choose a consumer product that is available in the field of medicine,
communication, transportation and agriculture,
2. Fill-in the table with correct information.
Data and Results
Area or Field
Consumer
Some
Use
Level of
Cost
Product
Chemical
Safeness
Components
(Chemical
Formula)
Medicine
Communication
Transportation
Agriculture
QUESTIONS
1. For each field, which problem is being addressed by the consumer product?
2. What are some chemical components of the consumer products?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you



