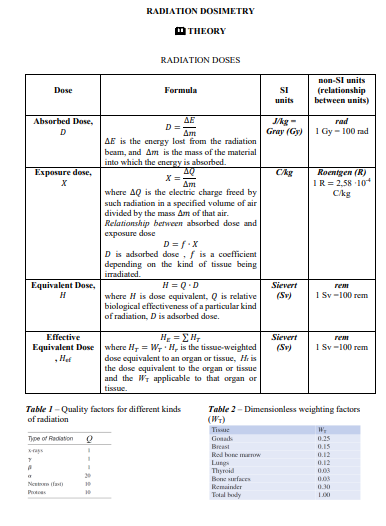
RADIATION DOSIMETRY O THEORY RADIATION DOSES non-SI units Dose Formula SI (relationship between units) units Absorbed Dose, J/kg - Gray (Gy) rad D%3D 1 Gy - 100 rad AE is the energy lost from the radiation beam, and Am is the mass of the material into which the energy is absorbed. 40 Ckg Roentgen (R) IR= 2.58 10 Ckg Exposure dose, х where AQ is the clectric charge freed by such radiation in a specified volume of air divided by the mass Am of that air. Relationship between absorbed dose and exposure dose D = f.X D is adsorbed dose, f is a coefficient depending on the kind of tissue being irradiated. Equivalent Dose, н H = Q.D where H is dose equivalent, Q is relative biological effectiveness of a particular kind of radiation, Dis adsorbed dose. Sievert геm (Sv) 1 Sy-100 rem Effective He = EH, Sievert Equivalent Dose where H, = W, H, is the tissue-weighted ,Het (Sv) 1 Sy-100 rem dose equivalent to an organ or tissue, Hr is the dose equivalent to the organ or tissue and the Wr applicable to that organ or tissue. Table 1- Quality factors for different kinds of radiation Table 2 - Dimensionless weighting factors Tissue Type of Rediation Gonads 0.25 Breast Rod une mnw 0.15 0.12 0.12 ras Lungs Thyrid Все меfе Nentn fast Remainder 0.30 Prtoas Total bly L00 DOSE RATES 1. Adsorbed dose rate: No Table 3 - Exposure constants Radionuclides ky(uCiy-mKGBq-h at I m 2. Dose equivalent rate: N 3. Exposure rate: Ny =- Exposure rate at a distance r from the source of "Mo radiation can be found as where is k, is exposure constant for a given radioisotope, A is activity of the radioactive source, r is distance from the source of the radiation. 88.11 15.95 12.16 39.46 20.54 41.89 55.41 37.03 15.16 58.65 154.05 Te "Co "P
Radioactive decay
The emission of energy to produce ionizing radiation is known as radioactive decay. Alpha, beta particles, and gamma rays are examples of ionizing radiation that could be released. Radioactive decay happens in radionuclides, which are imbalanced atoms. This periodic table's elements come in a variety of shapes and sizes. Several of these kinds are stable like nitrogen-14, hydrogen-2, and potassium-40, whereas others are not like uranium-238. In nature, one of the most stable phases of an element is usually the most prevalent. Every element, meanwhile, has an unstable state. Unstable variants are radioactive and release ionizing radiation. Certain elements, including uranium, have no stable forms and are constantly radioactive. Radionuclides are elements that release ionizing radiation.
Artificial Radioactivity
The radioactivity can be simply referred to as particle emission from nuclei due to the nuclear instability. There are different types of radiation such as alpha, beta and gamma radiation. Along with these there are different types of decay as well.
. (a) A 10 g tumour is irradiated with high energy gamma-rays and absorbs a total of 0.5 J of energy. What is the absorbed dose in gray and rad, and the dose equivalent in sievert and rem? (b) An alternate treatment for the same tumour is to administer a chemical solution containing a radioactive isotope which is preferentially absorbed by the tumour. If the isotope involved is an alpha emitter with an RBE of 20 and the tumour absorbs 0.10 J of energy, what is the absorbed dosein gray and rad, and the dose equivalent in sievert and rem?

Trending now
This is a popular solution!
Step by step
Solved in 6 steps with 6 images









