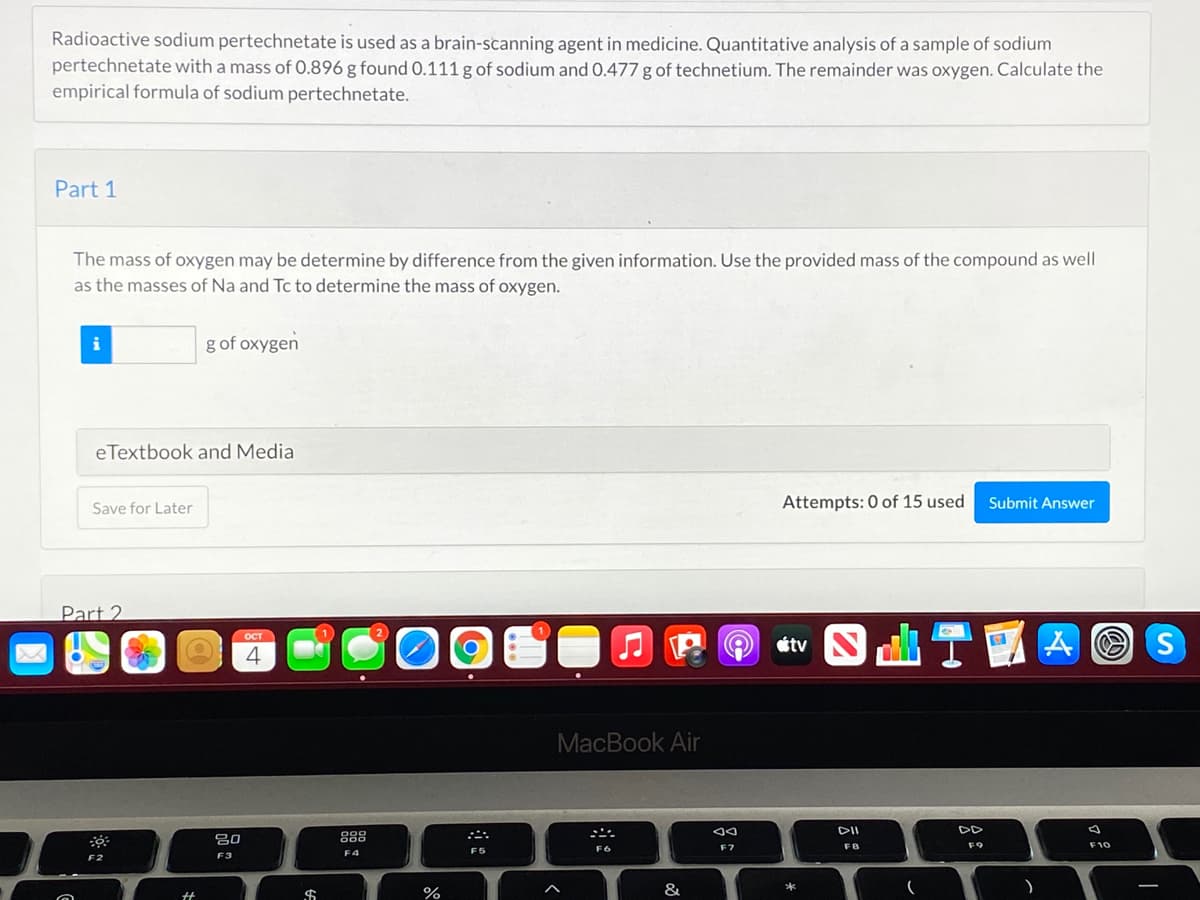
Radioactive sodium pertechnetate is used as a brain-scanning agent in medicine. Quantitative analysis of a sample of sodium pertechnetate with a mass of 0.896 g found 0.111 g of sodium and 0.477 g of technetium. The remainder was oxygen. Calculate the empirical formula of sodium pertechnetate. Part 1 The mass of oxygen may be determine by difference from the given information. Use the provided mass of the compound as well as the masses of Na and Tc to determine the mass of oxygen. g of oxygen eTextbook and Media
Radioactive sodium pertechnetate is used as a brain-scanning agent in medicine. Quantitative analysis of a sample of sodium pertechnetate with a mass of 0.896 g found 0.111 g of sodium and 0.477 g of technetium. The remainder was oxygen. Calculate the empirical formula of sodium pertechnetate. Part 1 The mass of oxygen may be determine by difference from the given information. Use the provided mass of the compound as well as the masses of Na and Tc to determine the mass of oxygen. g of oxygen eTextbook and Media
Chemistry: An Atoms First Approach
2nd Edition
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Steven S. Zumdahl, Susan A. Zumdahl
Chapter1: Chemical Foundations
Section: Chapter Questions
Problem 35E: Early tables of atomic weights (masses) were generated by measuring the mass of a substance that...
Related questions
Question
Transcribed Image Text:Radioactive sodium pertechnetate is used as a brain-scanning agent in medicine. Quantitative analysis of a sample of sodium
pertechnetate with a mass of 0.896 g found 0.111 g of sodium and 0.477 g of technetium. The remainder was oxygen. Calculate the
empirical formula of sodium pertechnetate.
Part 1
The mass of oxygen may be determine by difference from the given information. Use the provided mass of the compound as well
as the masses of Na and Tc to determine the mass of oxygen.
g of oxygen
eTextbook and Media
Attempts: 0 of 15 used
Submit Answer
Save for Later
Part 2
S T國囚
MacBook Air
DD
吕0
FB
F9
F10
F5
F6
F7
F4
F2
F3
$
%
&
*
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning