Chapter33: High-performance Liquid Chromatography
Section: Chapter Questions
Problem 33.14QAP
Related questions
Question
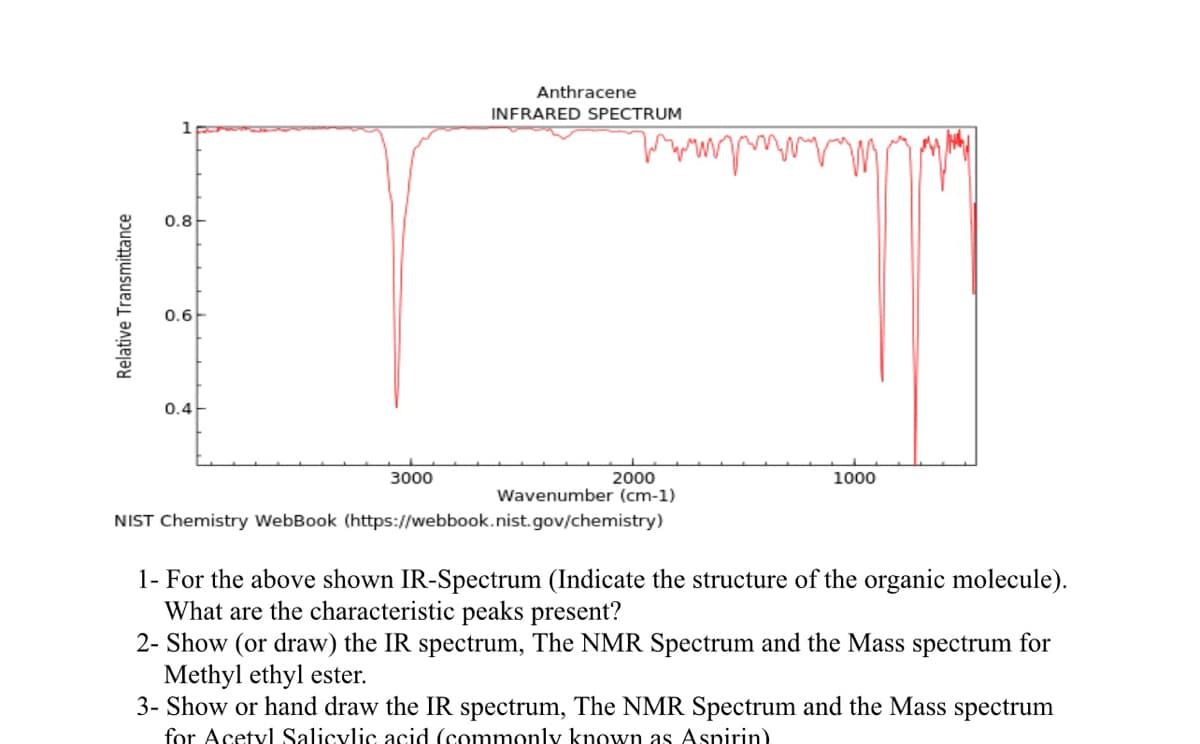
Transcribed Image Text:Relative Transmittance
1
0.8
0.6
0.4
3000
Anthracene
INFRARED SPECTRUM
www
2000
Wavenumber (cm-1)
NIST Chemistry WebBook (https://webbook.nist.gov/chemistry)
1000
1- For the above shown IR-Spectrum (Indicate the structure of the organic molecule).
What are the characteristic peaks present?
2- Show (or draw) the IR spectrum, The NMR Spectrum and the Mass spectrum for
Methyl ethyl ester.
3- Show or hand draw the IR spectrum, The NMR Spectrum and the Mass spectrum
for Acetyl Salicylic acid (commonly known as Aspirin)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning