Chapter3: Stoichiometry
Section: Chapter Questions
Problem 36Q: Consider the following generic reaction: Y2+2XY2XY2 In a limiting reactant problem, a certain...
Related questions
Question

Transcribed Image Text:Run 2: Initial grams of NaHCO3
Theoretical yield (grams) of Na2CO3:
Percent yield (Run 2)
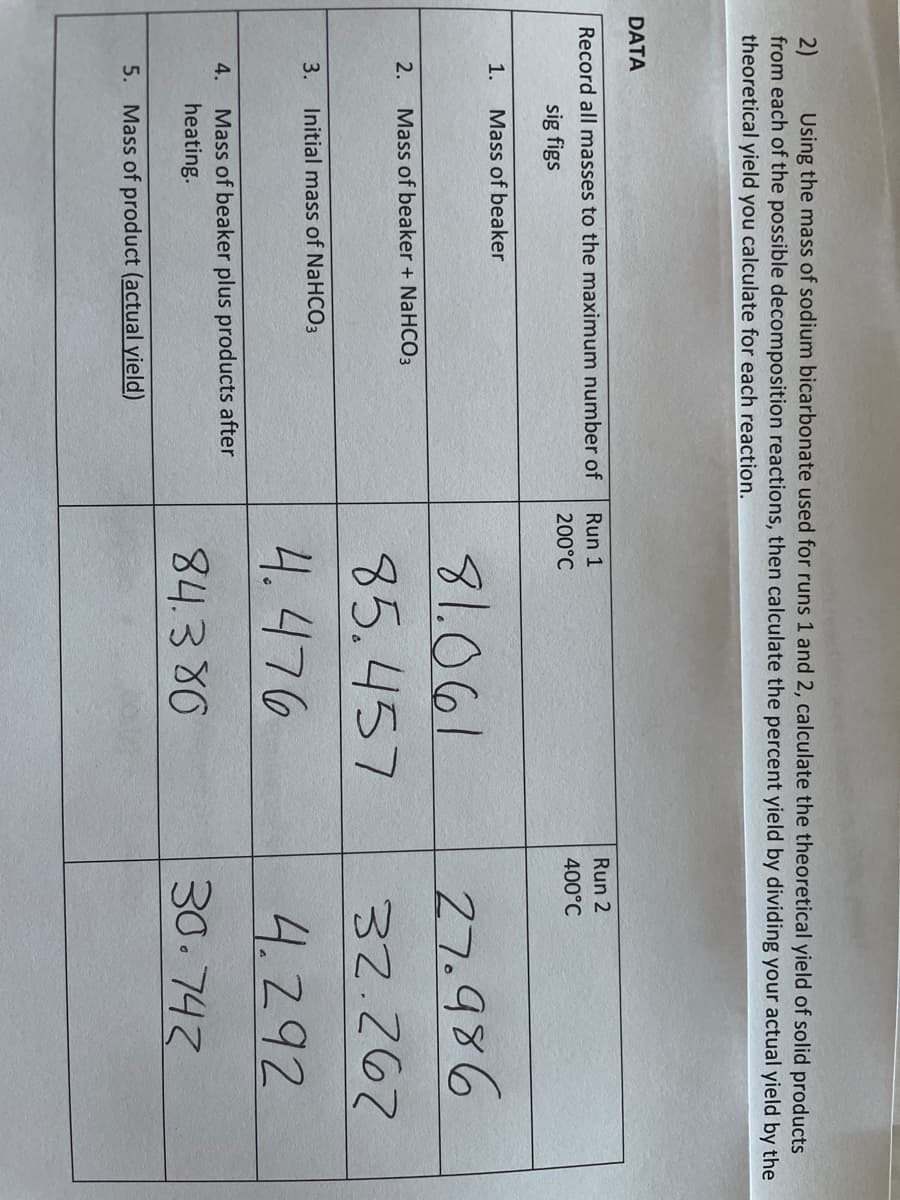
Transcribed Image Text:2)
from each of the possible decomposition reactions, then calculate the percent yield by dividing your actual yield by the
theoretical yield you calculate for each reaction.
Using the mass of sodium bicarbonate used for runs 1 and 2, calculate the theoretical yield of solid products
DATA
Record all masses to the maximum number of
Run 1
Run 2
200°C
400°C
sig figs
1.
Mass of beaker
81.061
27.986
2.
Mass of beaker + NaHCO3
85.457
32.267
3.
Initial mass of NaHCO3
4. 476
4.292
Mass of beaker plus products after
heating.
4.
84.380
30.742
5. Mass of product (actual yield)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 4 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning