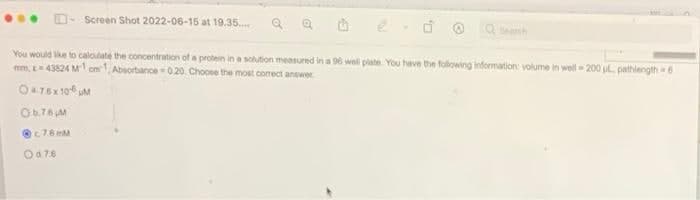
& protein in a solution measured in a 96 well plate You have the following information: volume in well-200 ul., pathlength 6 mm, -43824 M1cm1, Absorbance 020. Choose the most correct answer 0476x 100 M Ob.76 M c76mM Od 7.6
Q: ncreasing the concentration of the salt in a colloidal system will: a) Increase the radius of sheer.…
A: Given in following question a concentration of the salt in a colloidal system will increase the .…
Q: Analytes than can be trapped inside Sephadex G-25 (1,000-5000) Cytochrome C (MW=12384) Aprotinin…
A: Sephadex is a bead-formed gel prepared by cross linking dextran with epichlorohydrin. It is supplied…
Q: What impact would the increased use of electric vehicles have on environmental damage caused by acid…
A: Please find your solution below : Acid rain is a form of precipitation that consists of highly…
Q: Determine the number of valence electrons for the following elements potassium " calcium . arsenic W…
A: The outermost shell electrons are called as valence electrons.
Q: How many milliliters of 10.0 M HCl(aq) are needed to prepare 920.0 mL of 1.00 M HCl(aq)?
A: • The values provided in the question are:- i) Initial concentration of HCl solution, M1 =…
Q: A 0.530 mol sample of carbon dioxide gas occupies a volume of L at STP.
A: The answer to the following question is-
Q: I need help with problem #4
A: Given Synthesis the following reactions Answer only question no 4 as you ask According to the…
Q: Match this condensed formula, CH₂CH₂CH₂CH₂CHO, with the correct skeletal drawing.
A:
Q: The validation process comprises four main steps that are : hardware validation, software…
A: Validation in analytical chemistry is the process of verifying that a procedure yield acceptable…
Q: What effect does an increase in the atmospheric carbon dioxide levels (and therefore rising…
A: Carbon dioxide is a green house gas. The increased amount of atmospheric carbon dioxide level…
Q: CH₂OH/H Na 2 H₂O NaCN/H,O LIAIH4 1. CH3MgBr 2. H₂O H₂O/H30* CH3ONa _CH3OH_ NH3
A: Ring opening of epoxides takes place with different nucleophilic reagents. In acidic media, media…
Q: CO₂H OH EtOH, H* nd me structure of the product of this Fischer esterification reaction. Use the…
A:
Q: Select the product expected for the reaction shown. H₂ 1) NaOEt 2) 4CH₂Br C EtO₂C CO₂Et
A:
Q: Provide the correct IUPAC name for the compound shown here. CH3 G Ō. CH3
A:
Q: 7. Draw the product is formed when each alkene is treated with CH₂I2 and Zn(Cu)? a. I b. < C.
A: Since you have asked multiple question,we will solve the first question for you.If you want any…
Q: Epoxides are highly strained and can alleviate the strain by reacting under acidic and basic…
A: When a nucleophile(:Nuc) attacks an unsubstituted epoxide, it forms a nucleophile substituted…
Q: A high altitude weather balloon filled with helium, He, collects data as it rises through the…
A:
Q: At 60oC, the vapor pressure of ethanol is 352.7 mm Hg and that of methanol is 625.0 mm Hg. These two…
A:
Q: What is the first p element in the 4th period to which Hund's rule must be applied when writing the…
A: Given that, period 4th element = first block = p block
Q: of the molecules shown can be made using the Williamson ether synthesis. Identify the ether and draw…
A: Simple as well as mixed ethers can be prepared using Williamson ether synthesis reaction.
Q: what is the binding energy (in MeV) for an iron-56 nucleus? electron=0.000549 mass amu…
A: Atomic number of iron(Fe) is 26 mass number of Iron-56 is 56 Hence Iron-56 has 26 protons and 56-26…
Q: Explain why the amount of dissolved oxygen in a lake is often considered an indicator of the…
A: Dissolved oxygen (DO) is the amount of oxygen gas present in water. Running water has more amount of…
Q: Consider the diagram of galvanic cell, which consists of Ag*(aq)/Ag(s) and Sn²+ (aq)/Sn(s)…
A:
Q: Five hundred grams of a 0.100 molal solution of contains how many grams of NaF?
A: 500g of 0.100 molal Solution is given Now formula of Molality is as Molality = moles of solute…
Q: 1) For the following cell at 25°C: a) Zn(s) | ZnCl₂(aq) || AgCl (aq) | Ag(s) Write the…
A:
Q: Identify two nonrenewable and two renewable resources used to generate electricity.
A: Nonrenewable and renewable both resources are present in the nature, from where electricity has…
Q: Give specific organic reactions for hydrohalogenation. Draw the skeletal or condensed structure of…
A:
Q: Which statement is false? O A gas is composed of molecules that are separated from each other by…
A: Postulates of kinetic theory of gases, • Gases are composed of molecules separated from each other…
Q: Silver ions can be precipitated from aqueous solutions by the addition of aqueous chloride:…
A:
Q: CO₂H OH EtOH, H' Not Submitted Drew the structure of the product of this Fischer esterification…
A:
Q: same compounds. and (a) 0-H O +0-H (c) and 0- CH₂-C-H (e) CH₂=C-H and CH₂-C-H
A:
Q: Much of the U.S. electricity is generated in thermal generating stations that burn fossil fuels such…
A: This is a very good move as burning of fossil fuels produces very harmful gases such as carbon…
Q: Protons Ha and Hb in this compound are На, Нь O diastereotopic O mesotopic O magnetotopic O…
A: To check this replace Ha and Hb with other atom say D.
Q: -Draw an (a) expanded structural formula and (b) a line formula cor lowing condensed structural…
A:
Q: In Scotland, the legal blood alcohol limit is 220 μg of alcohol per 1 L of breath. What is this…
A: The parts per billion is the concentration measurement of a solution. It measures the concentration…
Q: Which carbonyl contains the most acidic proton on its alpha carbon?
A: Acidity means release of hydrogen ions when dissolved in water. Organic reactions are those in which…
Q: (H₂O) Wine goes bad soon after opening because the ethanol (CH3CH₂OH) in it reacts with oxygen gas…
A:
Q: Consider the following method for the analysis of cocaine (MW 303.35) powder. "Dissolve about 600 mg…
A: For acid base titartion: Nacid x Vacid = Nbase x Vbase Since the given acid is monoprotic and there…
Q: What are the major biomolecules essential for life?
A: Biomolecules: The molecules that are present in living organisms are called biomolecules. The main…
Q: The U.S. is one of several countries committed to reaching “net-zero” carbon emissions by 2050. This…
A: This step should be taken by all the countries because carbon dioxide is the main reason behind most…
Q: What is the formal charge of O in the molecule below? a. 0 O b.-2 O C. -1 O d. +2 :0:- :C1:
A:
Q: The reaction shown can give two different mono-alkylation products depending on the conditions. Draw…
A: Here we are required to predict the product of the reaction.
Q: What about 4,5,6
A: The following reaction is done by appropriate reagent by applying right mechanism.
Q: Draw the products of each nucleophilic substitution reaction. CN Br a. (CH3)3C- b. (CH3)3C- acetone…
A:
Q: A solution is made by dissolving 23.4 g of potassium sulfate, K₂SO4, in enough water to make exactly…
A: Given, The mass of K2SO4 = 23.4 g. The volume of the solution = 500. mL The molarity of K2SO4 , K+…
Q: Identify mitigating measure/s What should be the appropriate response to minimize harm During the…
A: A question based on general chemistry that is to be accomplished.
Q: Determine the oxidation number of Phosphorus in the following. Show full calculations. a.Na3PO4 b.…
A: In a neutral compound, the oxidation numbers of all the elements add up to zero. In an ion, the…
Q: Draw the products of each SN1 reaction and indicate the stereochemistry when necessary. CH3CH2 CH3…
A: In SN1 reactions at first a carbocation is formed from the substrate molecule. Then nucleophile can…
Q: What is the % (weight/volume) of acetic acid in a vinegar that, when titrated, found a concentration…
A:
Q: ||| H H CH3 Which of the following wedge-and-dash structures represents the chair structure shown…
A:
Step by step
Solved in 2 steps with 2 images

- analyte concentration(C)(mg/ml) injection volume (ul) elution time (time) peak DAD signal(mAU) caffeine 1 1 4.67 302.85 aspartame 5 1 7.53 15.83 benzoic acid 1 1 8.14 89.98 saccharin 1 1 1.91 84.86 mixture(add everything above with 1:1:1:1 ratio) 1 4.47 69.58 How to get the concentration of the mixture in this case?A 5.00-mL sample of blood was treated with trichloroacetic acid to precipitate proteins. After centrifugation, the resulting solution was brought to pH 3 and extracted with two 5-mL portions of methyl isobutyl ketone containing the lead-complexing agent APCD. The extract was aspirated directly into an air/acetylene flame and yielded an absorbance of 0.527 at 283.3 nm. Five-milliliter aliquots of standard solutions containing 0.400 and 0.600 ppm of lead were treated in the same way and yielded absorbances of 0.396 and 0.599. Find the concentration of lead in the sample in ppm assuming that Beer’s law is followed.A 5.00-mL sample of blood was treated with trichloroacetic acid to precipitate proteins. After centrifugation, the resulting solution was brought to pH 3 and extracted with two 5-mL portions of methyl isobutyl ketone containing the lead-complexing agent APCD. The extract was aspirated directly into an air/acetylene flame and yielded an absorbance of 0.569 at 283.3 nm. Five-milliliter aliquots of standard solutions containing 0.400 and 0.600 ppm of lead were treated in the same way and yielded absorbances of 0.396 and 0.599. Please calculate the concentration of lead in the sample in ppm.
- An HPLC method was developed for the separation and determination of ibuprofen in blood samples. A 3.00 mL blood sample dissolved in 70.00 mL solvent was prepared and 10.0 mL was injected into the HPLC. Standard ibuprofen solutions were also run.Calculate the ibuprofen present in the 3.00 mL blood sample The following data were obtained. Table po itoo: Ibuprofen concentration, mg/ml0.50 1.0 2.0 3.0 Blood sample Peak area (Right column)5.29.617.419.911.4You obtained the following raw data when setting up a Biuret standard curve: BSA (mg/ml) Absorbancy 540nm 0 0.158 1 0.210 2 0.260 3 0.305 4 0.360 5 0.410 6 0.455 7 0.510 8 0.530 9 0.550 10 0.554 After blanking against a biuret-dH2O sample, the protein concentration of an unknown sample was determined using the same method and an absorbancy of 0.251 was obtained. Set up a standard curve, excluding outliers (experimental and statistical) and determine the protein concentration in the unknown sample in mg / ml (up to 3 significant figures).You obtained the following raw data when setting up a Bradford standard curve: BSA (mg/ml) Absorbancy 595nm 0 0.225 1 0.310 2 0.420 3 0.510 4 0.610 5 0.720 6 0.810 7 0.915 8 0.950 9 0.980 10 0.990 After blanking against a bradford-dH2O sample, the protein concentration of an unknown sample was determined using the same method and an absorbancy of 0.523 was obtained. Set up a standard curve, excluding outliers (experimental and statistical) and determine the protein concentration in the unknown sample in mg / ml (up to 3 significant figures).
- FT-IR technique can be utilized for the analysis of unknown analytes by matching it with__________. Both choices are correct Commercial library/database Reference standardUse the following information collected from a SIZE-EXCLUSION CHROMATOGRAPHY experiment to answer the questions below. MW Standards Molecular Weight (kDa) Elution Volume (mL) blue dextran 1000 85 myoglobin 17.2 200 TIM 53.3 178 hemoglobin 62.2 156 IgG 140 122 ATCase 307.9 94 Prepare a graph showing the linear relationship between log function of molecular weight against Ve/Vo. The R2value must be 0.9 or higher. What is the linear equation from the standards given? What is the R2value?The figure below shows a typical separation of several fat-soluble vitamins by HPLC. The x-axis is retention time in minutes. From the graph try to estimate retention times for peaks 2 and 3. Retention time peak 2 = Retention time peak 3 = Assume that w1/2 is 0.15 minutes for peak 2 and 0.16 minutes for peak 3. Assume that tm is 2.00 minutes. How much time does gamma-tocopherol spend in the stationary phase? From tr and w1/2 find N for the peak that elutes first. From tr and w1/2 find the resolution of peaks 2 and 3 Calculate α for these peaks. If the flow rate for the column is 1.2 mL/minute what is the retention volume for peak 4?
- A protein consists of two types of peptide chains (A and B) with an unknown stoichiometry (AxBy). When you ran this protein directly on reversed phase-HPLC with UV monitor set at 280 nm, two peaks were resolved. Mass spec determined that the peaks represented Chain A and Chain B, respectively. The peak area is 500,000 for Peak A (Chain A) and 100,000 for Peak B (Chain B). The molecular masses of Chain A and Chain B are 25,000 and 5000, respectively. The extinction coefficients for Chain A and Chain B are 1 mL/mg.cm and 0.5 mL/mg.cm, respectively. Please calculate x/y.A 0.0200 gram blood sample was decomposed by a microwave digestion technique followed by dilution to 100.0 mL in a volumetric flask. Aliquots of the sample solution were treated with a lead complexing reagent and water as follows: Solution 1: 10.0 ml blood sample + 20.0 mL complexing agent + 30.0 mL H20. Solution 2: 10.0 ml blood sample + 20.0 mL complexing agent + 26.0 mL H20 + 4.00 mL of 78 ppb Pb2+ standard. The resulting solutions were analyzed by UV/Vis at 375 nm. Absorbance for solution 1 = 0.155 and for solution 2 = 0.216. Calculate the concentration of lead (ppb) in the original sample.1) The ratio strength of a 10mL vial of neostigmine methylsulfate injection USP is 1:1500. Calculate the amount of neostigmine methylsulfate in milligrams contained in the 10mL vial. (Round to the whole number).

