Select the reagents you would use to synthesize the compound below from benzene. (More than one step is required. If no third step is needed, choose (none).) Reagents Available а. Brz. FeBrg f. KMNO4, H2O b. Cl2, FeCl3 g. H2, Pd/C с. HNO3. H2SO4 h. SO3, H2SO4 d. CH3CI, AICI3 i. NaOH, H2O e. CH3COCI, AICI3 j. N-bromosuccinimide (NBS) ball & stick v labels Step 1: Step 2: Step 3:
Select the reagents you would use to synthesize the compound below from benzene. (More than one step is required. If no third step is needed, choose (none).) Reagents Available а. Brz. FeBrg f. KMNO4, H2O b. Cl2, FeCl3 g. H2, Pd/C с. HNO3. H2SO4 h. SO3, H2SO4 d. CH3CI, AICI3 i. NaOH, H2O e. CH3COCI, AICI3 j. N-bromosuccinimide (NBS) ball & stick v labels Step 1: Step 2: Step 3:
Chapter11: Reactions Of Alkyl Halides: Nucleophilic Substitutions And Eliminations
Section11.SE: Something Extra
Problem 80AP: Amines are converted into alkenes by a two-step process called Hofmann elimination. SN2 reaction of...
Related questions
Question
3
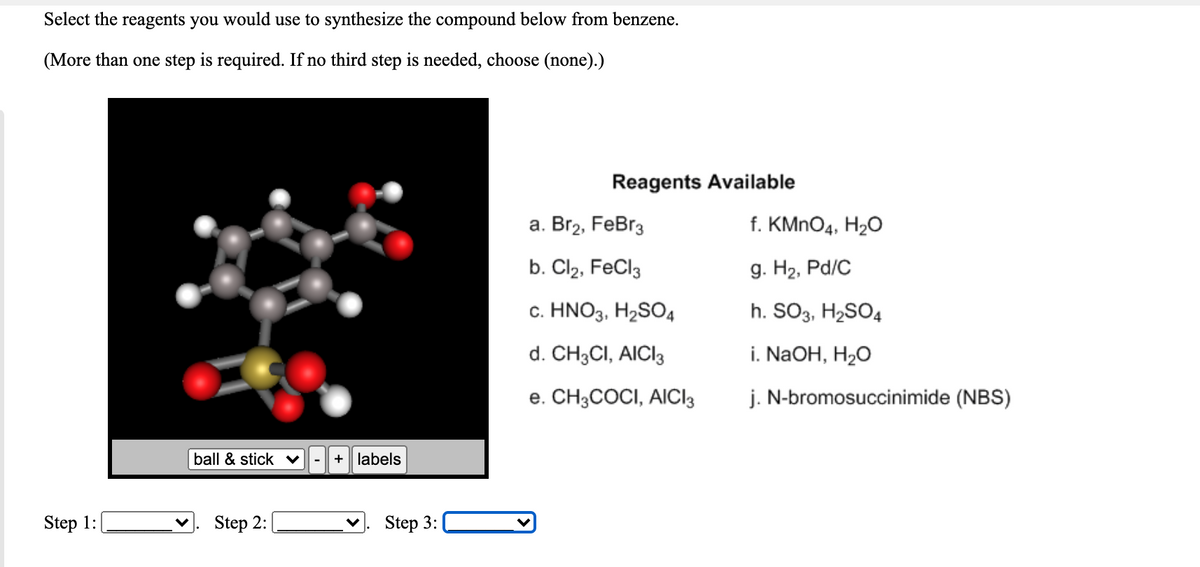
Transcribed Image Text:Select the reagents you would use to synthesize the compound below from benzene.
(More than one step is required. If no third step is needed, choose (none).)
Reagents Available
а. Вг2, FeBrg
f. KMNO4, H2O
b. Cl2, FeCl3
9. На, Pd/C
c. HNO3, H2SO4
h. SO3, H2SO4
d. CH3CI, AICI3
i. NaOH, H2O
e. CH3COCI, AICI3
j. N-bromosuccinimide (NBS)
ball & stick v
+ labels
-
Step 1:
Step 2:
Step 3:
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning



Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning
