b) The B3LYP/6-31G(d) energy-minimized structure of 3-(4'-nitrophenyl)furan is shown below on an overlay of the conformational potential energy surface (PES). The inter-ring dihedral angle is approximately 22°. Briefly explain why the molecular structures with inter-ring dihedral angles of 0° and 90° are energy maxima. 3.50 3.00 2.50 2.00 22° 1.50 1.00 0.50 0.00 0.0 60.0 120.0 180.0 240.0 300.0 360.0 Inter-ring dihedral angle (°)
b) The B3LYP/6-31G(d) energy-minimized structure of 3-(4'-nitrophenyl)furan is shown below on an overlay of the conformational potential energy surface (PES). The inter-ring dihedral angle is approximately 22°. Briefly explain why the molecular structures with inter-ring dihedral angles of 0° and 90° are energy maxima. 3.50 3.00 2.50 2.00 22° 1.50 1.00 0.50 0.00 0.0 60.0 120.0 180.0 240.0 300.0 360.0 Inter-ring dihedral angle (°)
Organic Chemistry
8th Edition
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Chapter9: Nucleophilic Substitution And Β-elimination
Section: Chapter Questions
Problem 9.41P: Elimination of HBr from 2-bromonorbornane gives only 2-norbornene and no 1-norbornene. How do you...
Related questions
Question
PES Maximum at 0°:
PES Maximum at 90°:
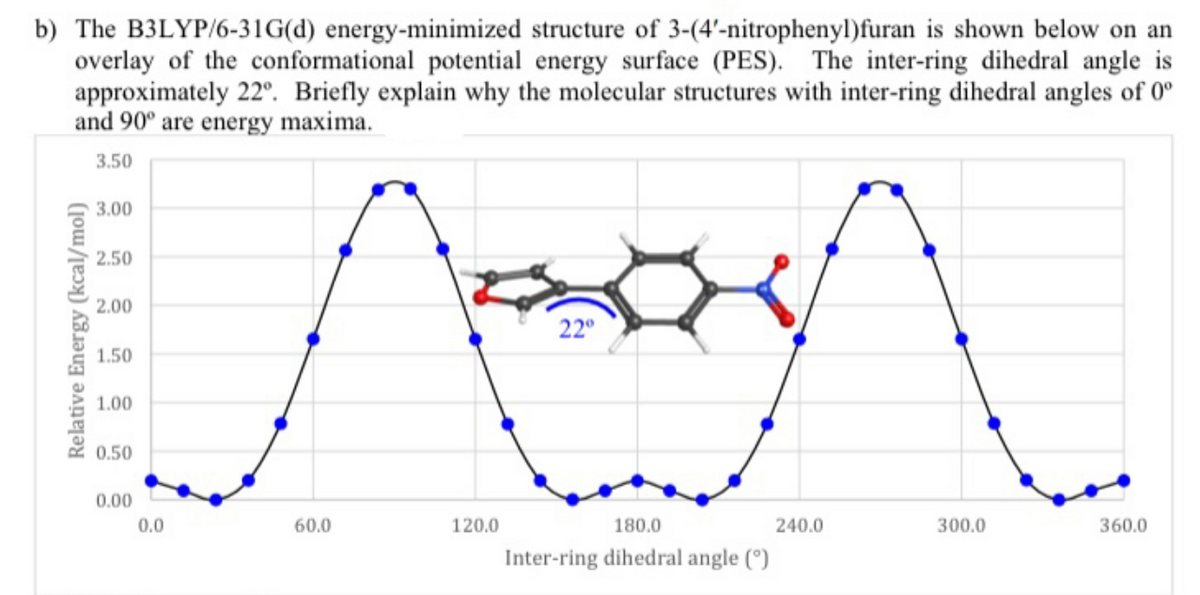
Transcribed Image Text:b) The B3LYP/6-31G(d) energy-minimized structure of 3-(4'-nitrophenyl)furan is shown below on an
overlay of the conformational potential energy surface (PES). The inter-ring dihedral angle is
approximately 22°. Briefly explain why the molecular structures with inter-ring dihedral angles of 0°
and 90° are energy maxima.
3.50
3.00
2.50
2.00
22°
1.50
1.00
0.50
0.00
0.0
60.0
120.0
180.0
240.0
300.0
360.0
Inter-ring dihedral angle (°)
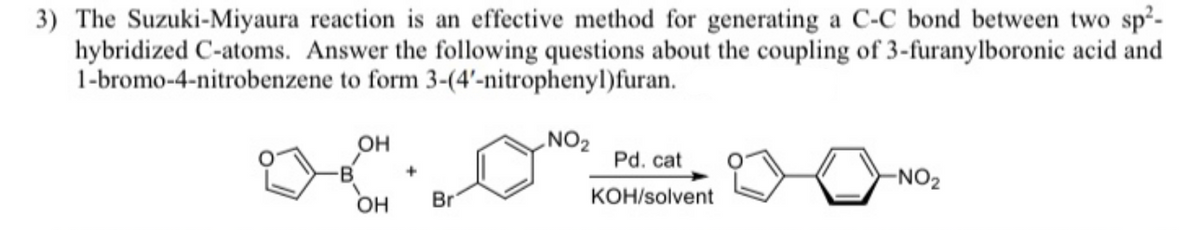
Transcribed Image Text:3) The Suzuki-Miyaura reaction is an effective method for generating a C-C bond between two sp²-
hybridized C-atoms. Answer the following questions about the coupling of 3-furanylboronic acid and
1-bromo-4-nitrobenzene to form 3-(4'-nitrophenyl)furan.
OH
NO2
Pd. cat
B
-NO2
OH
Br
KOH/solvent
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning