Side chains of which two amino acids may participate in peptide bonding? A. Alanine and Glycine B. Phenylalanine and Tryptophan C. Histidine and Tyrosine D. Glutamic acid and Lysine O E. Leucine and Isoleucine
Q: 3. The rates of a carbonic anhydrase catalyzed reaction of CO2 and H2O, in the absence and presence ...
A: Carbonic anhydrases catalyze the interconversion between CO2 and H2O, maintain acid base homeostasis...
Q: Which of the following differentiates erythrose with erythrulose? Erythrose has 2 chiral carbon whil...
A: Erythrose and erythrulose are the monosacharide sugars that consists of four carbon atoms. They diff...
Q: hat is the pH of 0.6M acetic acid?
A: Acetic acid is a weak acid, to calculate the pH we need acid dissociation constant (ka) , for this a...
Q: Statements: (1) Polysaccharides usually give a negative Benedict's test. (2) The hydrolysis of cel...
A: We will discuss the statements first and then see which is the correct option
Q: Which of the following is the correct statement for the a anomer and B anomer of D-arabinose? O They...
A: D-Arabinose: Arabinose is a monosaccharide with 5-carbon atom. It has a aldehyde functional group he...
Q: How to identify the Vmax and the Michaelis constant (Km) in an enzyme kinetics graph?
A: Vmax and KM are the two parameters that define the kinetic behavior of an enzyme as a function of [S...
Q: Choose from the following pairs. Which are constitutional isomers? Pair A Br
A: constitutional isomers are structural isomers that they differ in structure but have the same formul...
Q: C6H12O6 is the chemical formula for a ________.a. polymer of carbohydrateb. pentose monosaccharidec....
A: Carbohydrates, abbreviated as carbs, are sugar molecules. Carbohydrates, together with proteins and ...
Q: Which of the following is true of glucitol? It exists as both ring and open-chain structures with t...
A: Introduction: Monosaccharides can be reduced by turning the carbonyl group into an alcohol group. It...
Q: .Linoleic acid and linoleic acid cannot be synthesized in human tissues, because of _____. * a.Hum...
A:
Q: Do you feel that laboratory animals should be used in experiments in toxicology? Why or why not?
A: People and our quality of life benefit from research using laboratory animals. Most people on the ea...
Q: What is Intron retention type of alternativesplicing ?
A: A portion in mRNA that codes for protein is termed as exon. The non-coding region is termed as intro...
Q: Heteropolysaccharide structures are defined by: Question 23 options: Two or more different mon...
A: Carbohydrates are made up of carbon, hydrogen, and oxygen in the ratio of 1:2:1. The carbohydrates w...
Q: 1.What is the molar concentration of a 15% glycerol solution? (Glycero...
A: For the first question, 15% glycerol solution represents that 15g of glycerol is dissolved in 100mL ...
Q: Discuss the mechanism of action-potential in neurons?
A: Introduction: All excitable tissue like nerve and muscle tissue have membrane potential. The membran...
Q: Its principal function is to increase the concentration of glucose in the blood by speeding the glyc...
A: Glucose is the simplest source of energy for the body. The glucose which is obtained through diet is...
Q: Please explain in detail a chemical reaction between rRNA and another biomolecule
A: rRNA are the structural components of ribosomes. Hence rRNA does not generally participate in chemic...
Q: 4: CASE ANALYSIS Over the course of many months, a previously healthy 58-year-old lady develops inc...
A: The condition from which she is suffering is:
Q: Cellulose is indigestible but necessary for O aiding in elimination O preventing stomach cancer O ho...
A: Cellulose a linear polysaccharide composed of glucose molecules linked via β-1,4 glycosidic bond. Hu...
Q: Choose the order of reactions that best describes a metabolic pathway catalyzed by enzymes
A: In living cells the reactions occur at normal body temperature and pressure due to the presence of s...
Q: 15. During vigorous physical activity, a lot of pyruvate is generated inside muscle cells. Show, thr...
A: Hi. Thank you for the question. As per the honor code, we'll answer the first question since the exa...
Q: What are the important considerations that you have to remember in antibody screening?
A: Since you have asked multiple question, we will solve the first question for you. If you want any sp...
Q: Evaluating: Evaluate the following statements and determine which of them is FALSE about “agarose" *...
A:
Q: need answer asap
A: Inhibitors are substances which have the capability to inhibit the enzyme activity by binding to the...
Q: 1. Which of the following is the example of defense proteins? A. Dehydrogena B. Myoglobin C. Hemo...
A: Proteins are classified in different ways. The basis for the classification is function, chemical na...
Q: These are assembled primarily in the intestine and contain a smaller version called apoB-48.
A: Apolipoprotein B or apoB are proteins that binds oil soluble substances like fat and cholesterol bas...
Q: Answer the following questions. 1. List the complementary non-coding DNA sequence. This refers to th...
A: The messenger RNA (mRNA) sequence is given, and we are asked to determine the complementary non-codi...
Q: SSBs are: single-stranded DNA binding proteins that prevent re-annealing supercoil stabilizing...
A:
Q: Size exclusion chromatography is a protein purification method based on size. Larger proteins exit t...
A: Size exclusion chromatography: A chromatographic technique in which molecules in the solution get se...
Q: 2. Consider the experiment conducted by Meselson and Stahl in which they used 14N and 15N in culture...
A: Literally, replication means the system of duplication. In molecular biology, DNA replication is the...
Q: INTRUCTIONS: - Do not copy here in BARTLEBY or GOOGLE - PLEASE ANSWER PROPERLY Failed to follow inst...
A: Carbohydrates are divided into 3 classes monosaccharide, disaccharide, and polysaccharide. Pol...
Q: What is the likely mechanism of spread of NDM-1?
A: NDM-1 is the protein product of a gene that some bacteria produce. A bacterial strain containing NDM...
Q: . Which type of proteins are also known as denatured proteins? A. Primary proteins B. Secondary pr...
A: Since you have asked multiple questions, we will solve the first question for you. If you want any s...
Q: INTRUCTIONS: - Do not copy here in BARTLEBY or GOOGLE - PLEASE ANSWER PROPERLY Failed to follow inst...
A: Soap making is an ancient process that used to be done using rainwater, potash, and animal tallow. T...
Q: Understanding: Non-polar molecules are insoluble in water. This is because:
A: "Since you have asked multiple question, we will solve the first question for you. If you want any s...
Q: Compare and contrast the location and pH sensitivity of receptor-ligand interaction in the LDL and t...
A: Endocytosis occurs when a coat protein (typically clathrin) on the cytoplasmic side of the plasma me...
Q: QUESTION 4 Depolymerization of disaccharides into two monosaccharides involves alan reaction. amidat...
A: Disaccharides are the Carbohydrates that are made up of two monosacharide units joined together by G...
Q: Explain why oils become rancid. Provide short and concise answer with relevant examples.
A: Fats and oils are made up of triglycerides, which are esters made up of three fatty acid units bound...
Q: If the inoculation of yeast during the process of vinegar making are not done, can we still produce ...
A: Vinegar is nothing but acetic acid which is present in aqueous state. This can be used as flavouring...
Q: List some important types of molecular interactions involved in recognition and binding of polysacch...
A: Polysaccharides, also called as glycan are the abundant carbohydrates found in food. They are compos...
Q: From natural to synthetic ingredients in perfumery Explain and give example
A: When we smell perfume, we recognise certain fragrances. Scents of flowers, fruits, some foods, leath...
Q: THE MAIN BUFFERS OF THE CELLULAR FLUIDS ARE EXCEPT a. HPO4 & H2PO4- b. НСОз- & Н2СОЗ c. NH40H & NH4C...
A: Introduction: The buffering system in body fluids provide an immediate response in fluctuations in p...
Q: 1. In a protein, why does when Ala is replaced with Ile, it loses its activity but when Lys is repla...
A: Note : Hi ! Thank you for the question. We are authorized to answer one question at a time. Since yo...
Q: A substance that affect the rate of the reaction without being consumed Select one: a. enzymes b. a...
A: The study of chemical reactions catalysed by enzymes is known as enzyme kinetics. Enzymes, which act...
Q: 1. How do you prepare 25 mL of a 0.4X solution of SDS from a ...
A: Using formula C1V1= C2V2, we can calculate the Molarity, w/v, %, or any other concentration of the s...
Q: CH3 CH2-CH,-CH2 CH2-CH,CH2-CH2-CH2 CH2-CH2 ĉ-SCOA
A: Beta oxidation is the process by which long chain acyl CoA are broken down to acetyl CoA. Each round...
Q: Why are some sugars sweeter than others? Explain in 3 sentences. *
A: Sugars are simple Carbohydrates that act as building blocks for polysaccharides. They are used as fl...
Q: An enzyme following Michaelis-Menten kinetics was found to have highest activity at 37° C and pH 7. ...
A: Values given: temperature: 37°C pH: 7 Maximum velocity or Vmax= 2.4×10-4 mol.lit-1s-1 Enzyme concen...
Q: write the introduction for the effect of denaturing agent on the structure of phycocyanin on two pag...
A: Phycocyanin is a water-soluble pigment protein. It comes from microalgae. It is non-toxic. It has a...
Q: . The buffer that is adjusted to control acid-base balanceis ________.a. plasma proteinb. hemoglobin...
A: Carbon dioxide is produced in huge quantities in tissues with a high metabolic rate. Carbon dioxide ...
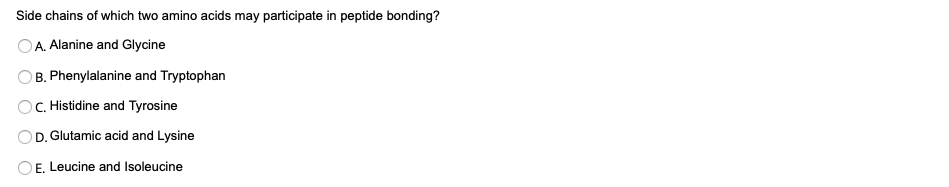
Step by step
Solved in 2 steps with 2 images

- Which of the following pairs of amino acids can have intermolecular hydrogen bonding between the functional groups in their side chains? Select all that apply. phenylalanine and tyrosine two tyrosine residues serine and threonine alanine and threonineDuring the formation of the peptide bond which of the following takes place? Select one: a. Hydroxyl group is lost from its carboxyl group of one amino acid and a hydrogen atom is lost from its amino group of another amino acid b. Hydrogen atom is lost from its carboxyl group of one amino acid and a hydroxyl group is lost from its amino group of another amino acid c. Hydroxyl group is lost from its carboxyl group of one amino acid and a hydroxyl group is lost from its amino group of another amino acid d. Hydrogen atom is lost from its carboxyl group of one amino acid and a hydrogen atom is lost from its amino group of another amino acid Clear my choiceWhich of the amino acids does NOT have an aliphatic side chain? a. Lysine b. Alanine c. Leucine d. Isoleucine e. Valine
- What determines the characteristics of an amino acid? Group of answer choices the identity of its side chain its molecular weight both a and b neither a nor bBiochemistry,based on the example,need help to 1)hand draw the dipeptides that contains Glutamic acid and Proline. 2)hand draw the tetra peptides that contains Glycine , Alanine,Glutamic acid and Proline.Which of the following amino acid pairs may form hydrogen bonds between eachother (select all that apply)? A. Proline and Proline B. Arginine and Aspartate C. Lysine and Glutamate D. Alanine and Aspartate E. Lysine and Asparagine
- Between which two amino acids do disulfide bonds form? A.Alanine-Glycine B.Serine-Threonine C.Cysteine-cysteine D.Methionine-methionineLocate the parts of the amino acids that participate in the chemical reaction that links the amino acids together in a covalent bond. Serine Cysteine LysineWhich of the following amino acid pairs may form hydrogen bonds between their sidechains (select all that apply)? A. Arginine and Aspartate B. Lysine and Glutamate C. Glycine and Cysteine D. Alanine and Aspartate E. Lysine and Asparagine
- Which of the following combination is are non-essential amino acids? A. Leucine and Isoleucine B. Isoleucine and Methionine C. Methionine and Asparagine D. Proline and AlanineDuring the formation of the peptide bond which of the following takes place? A. Hydroxyl group is lost from its Carboxyl group of one amino acid and a hydrogen atom is lost from its amino group of another amino acid B. Hydrogen atom is lost from its Carboxyl group of one amino acid and a hydroxyl group is lost from its amino group of another amino acid C. Hydroxyl group is lost from its Carboxyl group of one amino acid and a hydroxyl group is lost from its amino group of another amino acid D. Hydrogen atom is lost from its Carboxyl group of one amino acid and a hydrogen atom is lost from its amino group of another amino acidWhich of these are considered “part” of each amino acid? A. Carboxyl acid group B. All of the options are correct C. R Group D. Amino group



