Similar to alkanes, hydrogen gas can undergo radical halogenation according to the following reaction, where X = F, CI, Br, or I. Propose a chain-reaction mechanism for this reaction, including an initiation step, propagation steps, and two plausible termination steps. H-H + X-X 2 H-X hv
Similar to alkanes, hydrogen gas can undergo radical halogenation according to the following reaction, where X = F, CI, Br, or I. Propose a chain-reaction mechanism for this reaction, including an initiation step, propagation steps, and two plausible termination steps. H-H + X-X 2 H-X hv
Chapter10: Organohalides
Section10.SE: Something Extra
Problem 21MP: Alkyl halides can be reduced to alkanes by a radical reaction with tributyltin hydride, (C4H9)3SnH,...
Related questions
Question
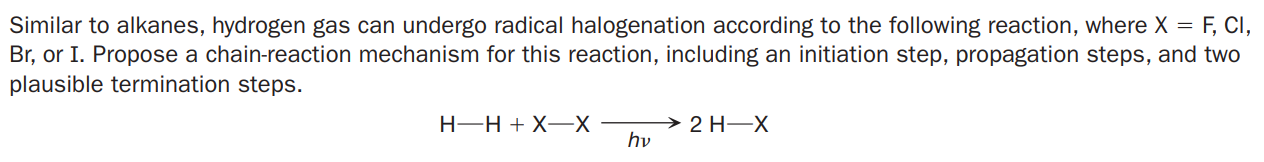
Transcribed Image Text:Similar to alkanes, hydrogen gas can undergo radical halogenation according to the following reaction, where X = F, CI,
Br, or I. Propose a chain-reaction mechanism for this reaction, including an initiation step, propagation steps, and two
plausible termination steps.
H-H + X-X
2 H-X
hv
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning