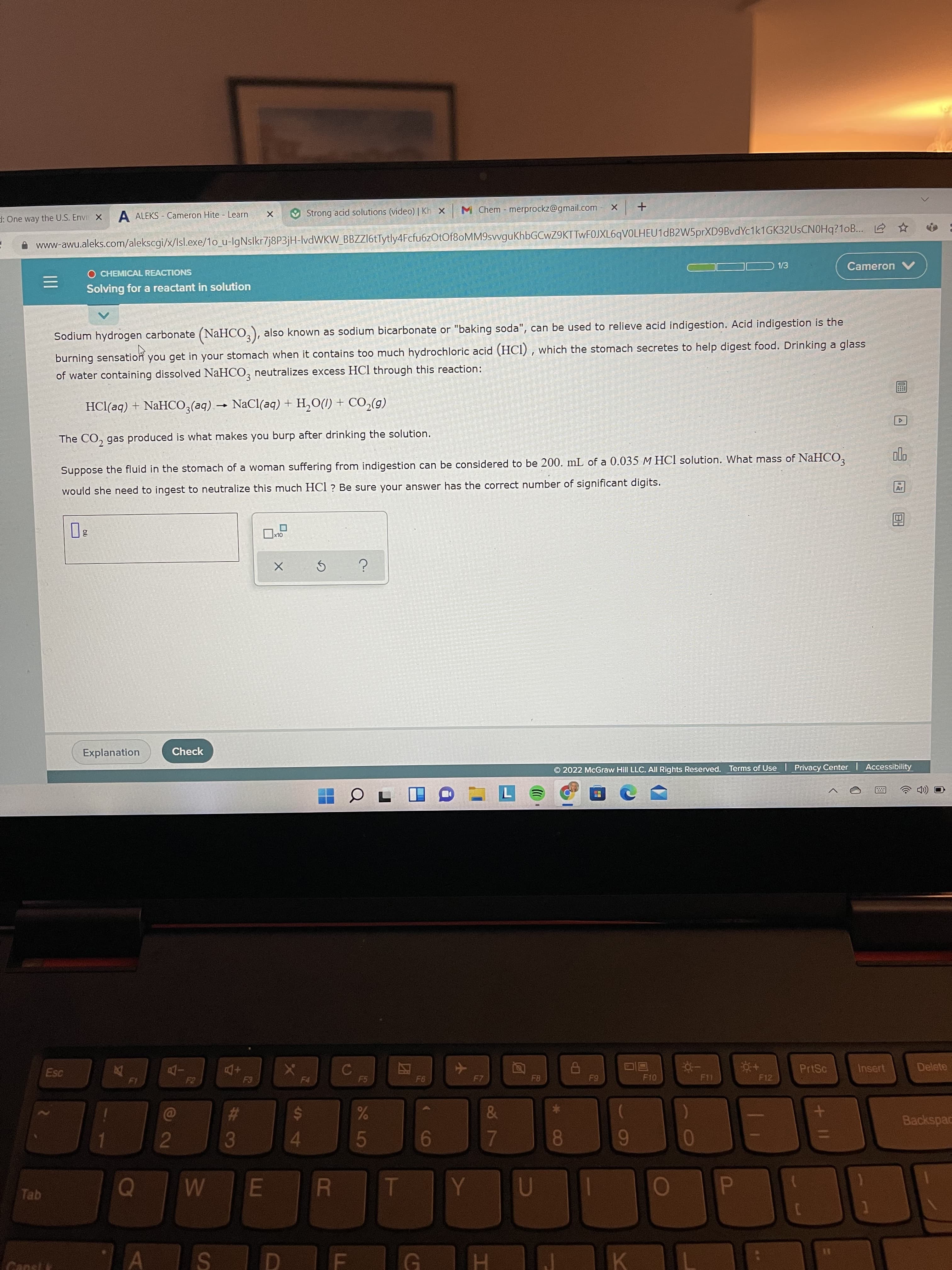
Sodium hydrogen carbonate (NaHCO, ), also known as sodium bicarbonate or "baking soda", can be used to relieve acid indigestion. Acıd ihdlgestloH burning sensation of water containing dissolved NaHCO, neutralizes excess HCl through this reaction: you get in your stomach when it contains too much hydrochloric acid (HCI) , which the stomach secretes to help digest food. Drinking a glass HCl(aq) + NAHCO,(aq) → NaCl(aq) + H,O(1) + C0,(g) The CO, gas produced is what makes you burp after drinking the solution. Suppose the fluid in the stomach of a woman suffering from indigestion can be considered to be 200. mL of a 0.035 M HCI solution. What mass of NaHCO, olo would she need to ingest to neutralize this much HCl ? Be sure your answer has the correct number of significant digits. Ar
Sodium hydrogen carbonate (NaHCO, ), also known as sodium bicarbonate or "baking soda", can be used to relieve acid indigestion. Acıd ihdlgestloH burning sensation of water containing dissolved NaHCO, neutralizes excess HCl through this reaction: you get in your stomach when it contains too much hydrochloric acid (HCI) , which the stomach secretes to help digest food. Drinking a glass HCl(aq) + NAHCO,(aq) → NaCl(aq) + H,O(1) + C0,(g) The CO, gas produced is what makes you burp after drinking the solution. Suppose the fluid in the stomach of a woman suffering from indigestion can be considered to be 200. mL of a 0.035 M HCI solution. What mass of NaHCO, olo would she need to ingest to neutralize this much HCl ? Be sure your answer has the correct number of significant digits. Ar
Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter1: Chemical Foundations
Section: Chapter Questions
Problem 1RQ: Define and explain the differences between the following terms. a. law and theory b. theory and...
Related questions
Question
I need help solving this chemistry equation on solving for a reactant in solution.
Transcribed Image Text:+II
P
08
CO
团
R
%#3
A ALEKS - Cameron Hite - Learn
Strong acid solutions (video) | Kh x
M Chem - merprockz@gmail.com - X
d: One way the U.S. Envi X
www-awu.aleks.com/alekscgi/x/Isl.exe/1o_u-IgNslkr7j8P3jH-IVDWKW_BBZZI6tTytly4Fcfu6zOtOf8oMM9svvguKhbGCwZ9KTTwF0JXL6qVOLHEU1dB2W5prXD9BvdYc1k1GK32UsCNOHq?1oB.. 2 ☆
O CHEMICAL REACTIONS
1/3
Cameron
三
Solving for a reactant in solution
Sodium hydrogen carbonate (NaHCO,), also known as sodium bicarbonate or "baking soda", can be used to relieve acid indigestion. Acid indigestion is the
burning sensation you get in your stomach when it contains too much hydrochloric acid (HCI) , which the stomach secretes to help digest food. Drinking a glass
of water containing dissolved NaHCO, neutralizes excess HCl through this reaction:
HCl(aq) + NaHCO,(aq) → NaCl(aq) + H,O(1) + CO,(g)
画
The CO, gas produced is what makes you burp after drinking the solution.
Suppose the fluid in the stomach of a woman suffering from indigestion can be considered to be 200. mL of a 0.035 M HCl solution. What mass of NaHCO,
would she need to ingest to neutralize this much HCl? Be sure your answer has the correct number of significant digits.
Explanation
Check
O 2022 McGraw Hill LLC. All Rights Reserved. Terms of Use Privacy Center Accessibility
Esc
PrtSc
Insert
Delete
-Do
+DD
F8
F10
F11
F12
F4
63
&
%23
Backspac
%24
6
Tab
Cansls
K.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 5 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781259911156
Author:
Raymond Chang Dr., Jason Overby Professor
Publisher:
McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781259911156
Author:
Raymond Chang Dr., Jason Overby Professor
Publisher:
McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9780078021558
Author:
Janice Gorzynski Smith Dr.
Publisher:
McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Elementary Principles of Chemical Processes, Bind…
Chemistry
ISBN:
9781118431221
Author:
Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:
WILEY