Solubility (g/100g water) Question 17 What is the concentration (in molality, m, mol/kg water) in a saturated amonium bromide solution at 40 °C? Use the solubility graph below for your calculation. -091 8'HN ON)9d 120- 08 -09 NaCl CdSeO, 08 00L 09 Temperature (°C) O A. 9.2 m O B. 90. m OC. 0.92 m OD.0.0092 m
Solubility (g/100g water) Question 17 What is the concentration (in molality, m, mol/kg water) in a saturated amonium bromide solution at 40 °C? Use the solubility graph below for your calculation. -091 8'HN ON)9d 120- 08 -09 NaCl CdSeO, 08 00L 09 Temperature (°C) O A. 9.2 m O B. 90. m OC. 0.92 m OD.0.0092 m
Chemistry: An Atoms First Approach
2nd Edition
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Steven S. Zumdahl, Susan A. Zumdahl
Chapter10: Properties Of Solutions
Section: Chapter Questions
Problem 91AE: The solubility of benzoic acid (HC7H5O2), is 0.34 g/100 mL in water at 25C and is 10.0 g/100 mL in...
Related questions
Question
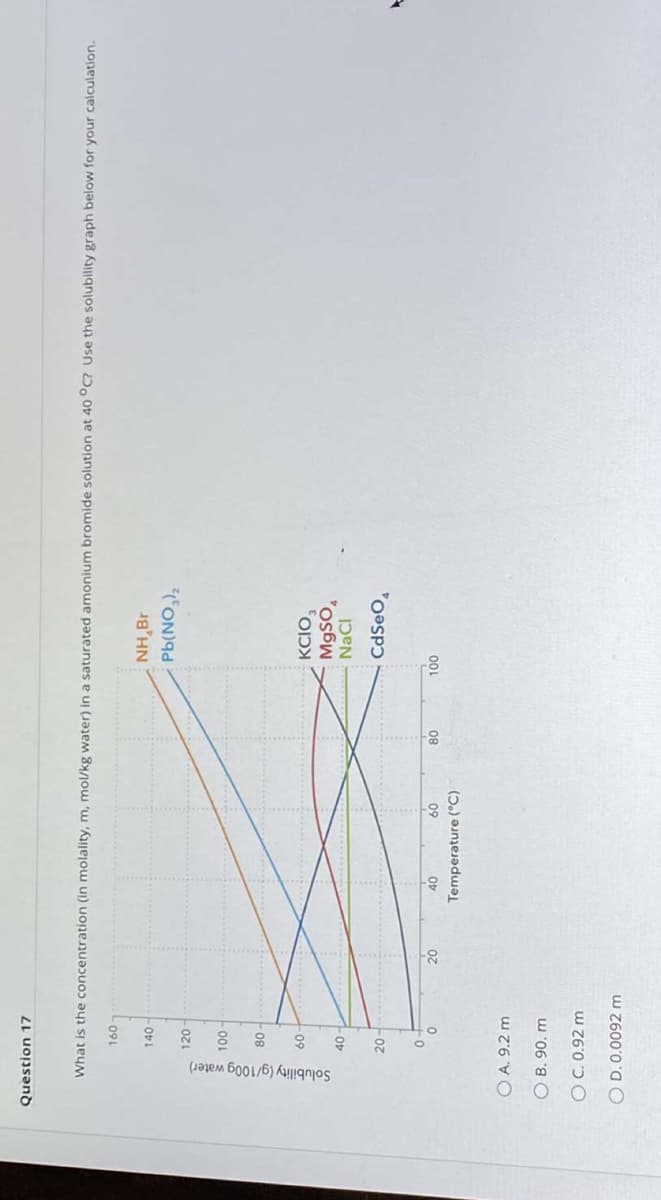
Transcribed Image Text:Solubility (g/100g water)
Question 17
What is the concentration (in molality, m, mol/kg water) in a saturated amonium bromide solution at 40 °C? Use the solubility graph below for your calculation.
091
18'HN
ON)9d
140
120
08
KCIO,
M9SO,
-09
NaCI
CdSeO,
-20-
08
00L
09
Temperature (°C)
O A. 9.2 m
O B. 90. m
OC. 0.92 m
O D. 0.0092 m
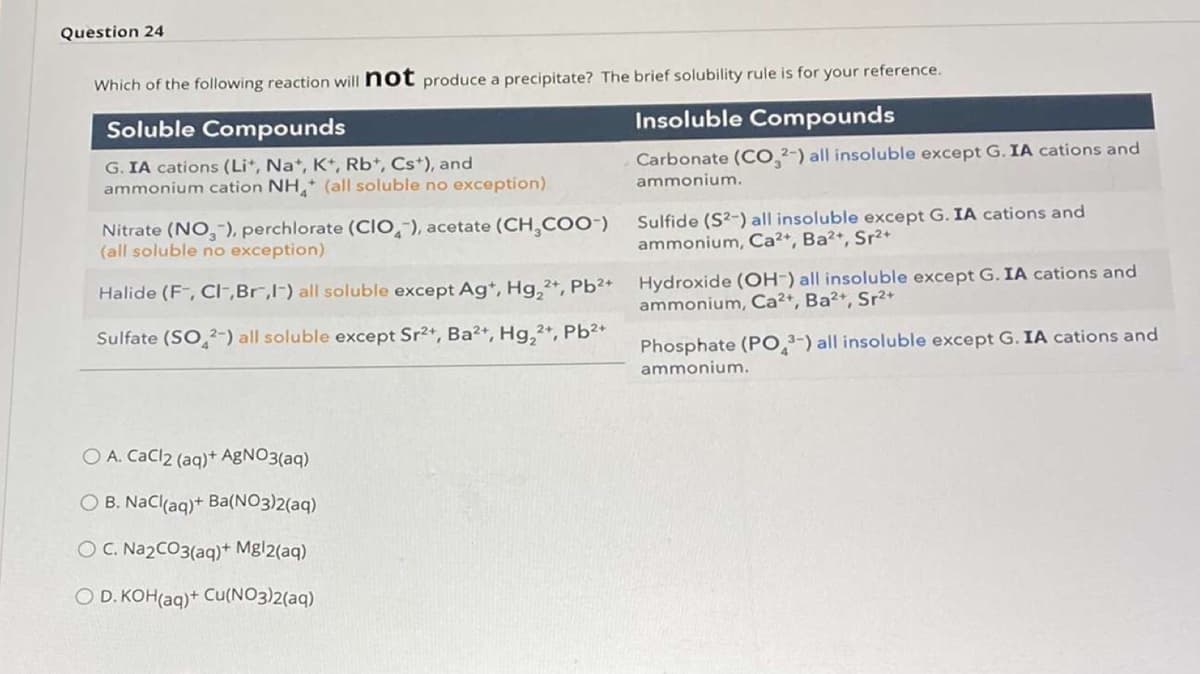
Transcribed Image Text:Question 24
Which of the following reaction will not produce a precipitate? The brief solubility rule is for your reference.
Soluble Compounds
Insoluble Compounds
Carbonate (CO.2-) all insoluble except G. IA cations and
G. IA cations (Li*, Na, K, Rb*, Cs+), and
ammonium cation NH (all soluble no exception)
ammonium.
Nitrate (NO-), perchlorate (CIO,-), acetate (CH COO-)
(all soluble no exception)
Sulfide (S2-) all insoluble except G. IA cations and
ammonium, Ca2+, Ba2+, Sr2+
Hydroxide (OH-) all insoluble except G. IA cations and
ammonium, Ca²+, Ba2+, Sr²+
Halide (F, CI-,Br,I-) all soluble except Ag*, Hg,2*, Pb²+
Sulfate (SO 2-) all soluble except Sr2+, Ba²+, Hg,2+, Pb²*
Phosphate (PO,3-) all insoluble except G. IA cations and
ammonium.
O A. CaCl2 (aq)+ AGNO3(aq)
O B. Nacl(aq)+ Ba(NO3)2(aq)
O C. Na2CO3(aq)+ Mgl2(aq)
O D. KOH(aq)+ Cu(NO3)2(aq)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co