Physical Chemistry
2nd Edition
ISBN:9781133958437
Author:Ball, David W. (david Warren), BAER, Tomas
Publisher:Ball, David W. (david Warren), BAER, Tomas
Chapter16: Introduction To Magnetic Spectroscopy
Section: Chapter Questions
Problem 16.49E
Related questions
Question
can someone provide me with the peaks and data from the IR spectrums in a table
1) table
2) table
and also explain both of them to me?
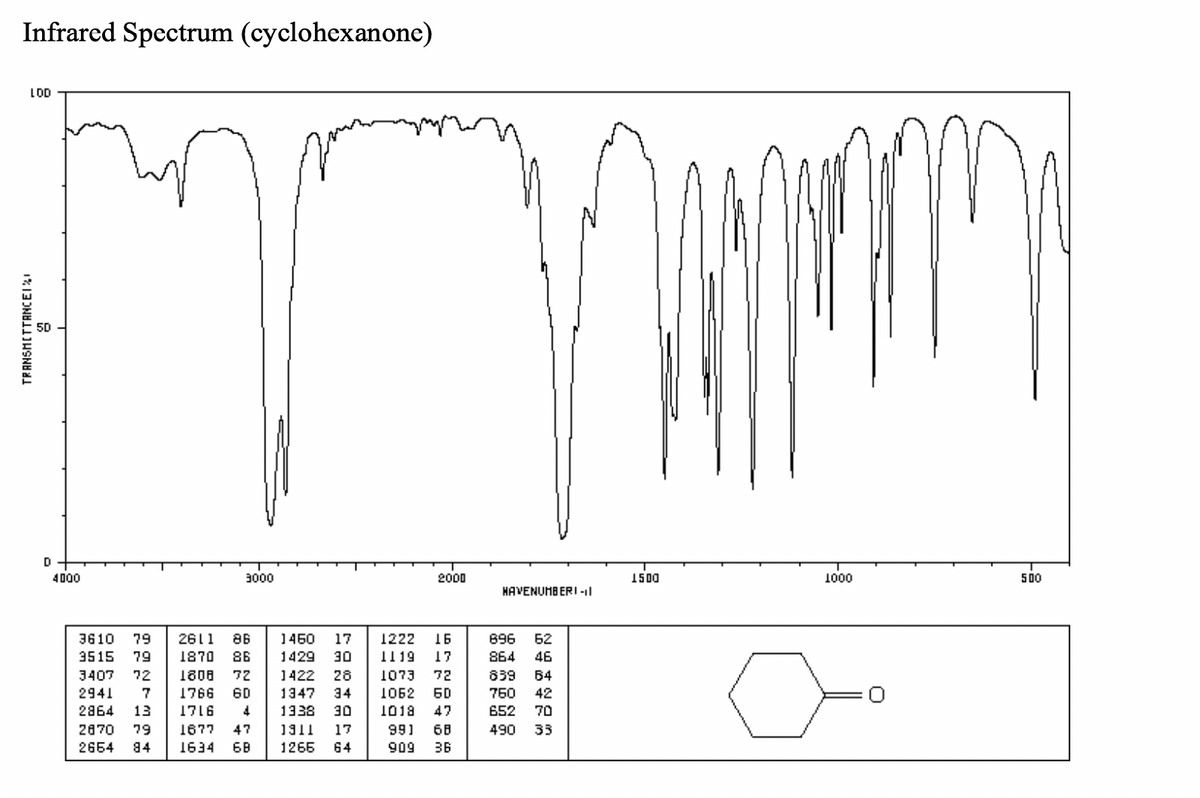
Transcribed Image Text:Infrared Spectrum (cyclohexanone)
LOD
4000
3000
2000
1500
1000
500
HAVENUHBERI -|
3610
79
2611
86
1460
17
1222
16
896
Б2
3515
79
1870
86
1429
30
1119
17
864
3407
72
72
1422
28
1073
72
639
В4
2941
7
1766
60
1347
34
1062
60
760
42
2864
13
1716
1338
30
1018
47
652
70
2070
79
1677
47
1911
17
99)
68
490
33
2664
84
1634
68
1266
64
909
36
TRANSMITTANCEIX
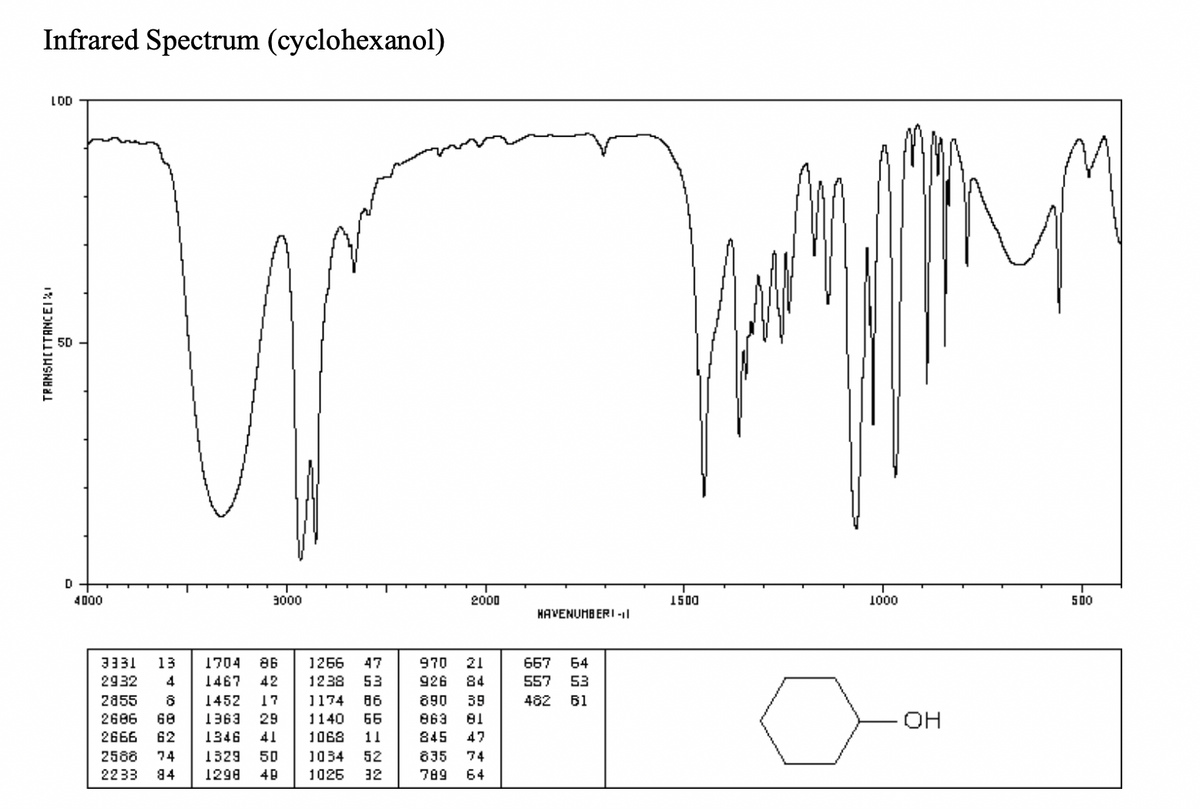
Transcribed Image Text:Infrared Spectrum (cyclohexanol)
LOD
4000
3000
2000
1500
1000
500
HAVENUMBERI -||
3331
13
1704
86
1266
47
970
21
667
Б4
2932
4
1467
42
1238
53
926
84
557
53
2855
1452
17
1174
86
690
39
482
61
2606
68
1963
29
1140
66
863
81
OH
2666
62
1346
41
1068
11
845
47
2588
74
1329
50
1034
52
835
74
2233
84
1298
40
1026
32
789
64
TRANSMITTANCEIX
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning