STARTING AMOUNT X A shop-vac is capable of pulling in air at a rate of 210 ft3/min. What is the rate of the vacuum's air flow in L/s ? 2.54 1000 ADD FACTOR x( ) 6400 (12)³ (1)³ (0.01)³ 12 5900 99000 (210)³ ANSWER II 10 (2.54)³ (10)³ 210 60 1 RESET 99 (1000)³ 0.01 6.4
STARTING AMOUNT X A shop-vac is capable of pulling in air at a rate of 210 ft3/min. What is the rate of the vacuum's air flow in L/s ? 2.54 1000 ADD FACTOR x( ) 6400 (12)³ (1)³ (0.01)³ 12 5900 99000 (210)³ ANSWER II 10 (2.54)³ (10)³ 210 60 1 RESET 99 (1000)³ 0.01 6.4
Chapter6: Random Errors In Chemical Analysis
Section: Chapter Questions
Problem 6.8QAP
Related questions
Question
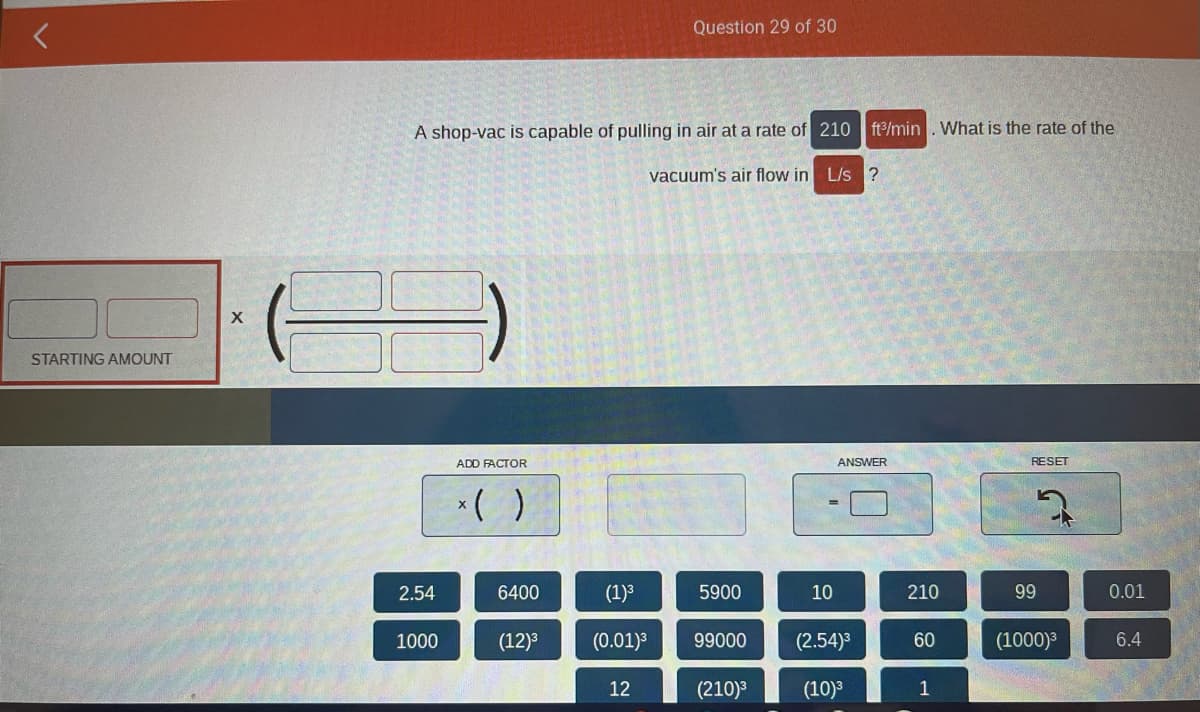
Transcribed Image Text:STARTING AMOUNT
X
A shop-vac is capable of pulling in air at a rate of 210 ft3/min. What is the rate of the
vacuum's air flow in L/s ?
2.54
1000
ADD FACTOR
x()
6400
(12)³
(1)³
(0.01)³
Question 29 of 30
12
5900
99000
(210)³
10
ANSWER
(2.54)³
(10)³
210
60
1
RESET
99
(1000)³
0.01
6.4
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

