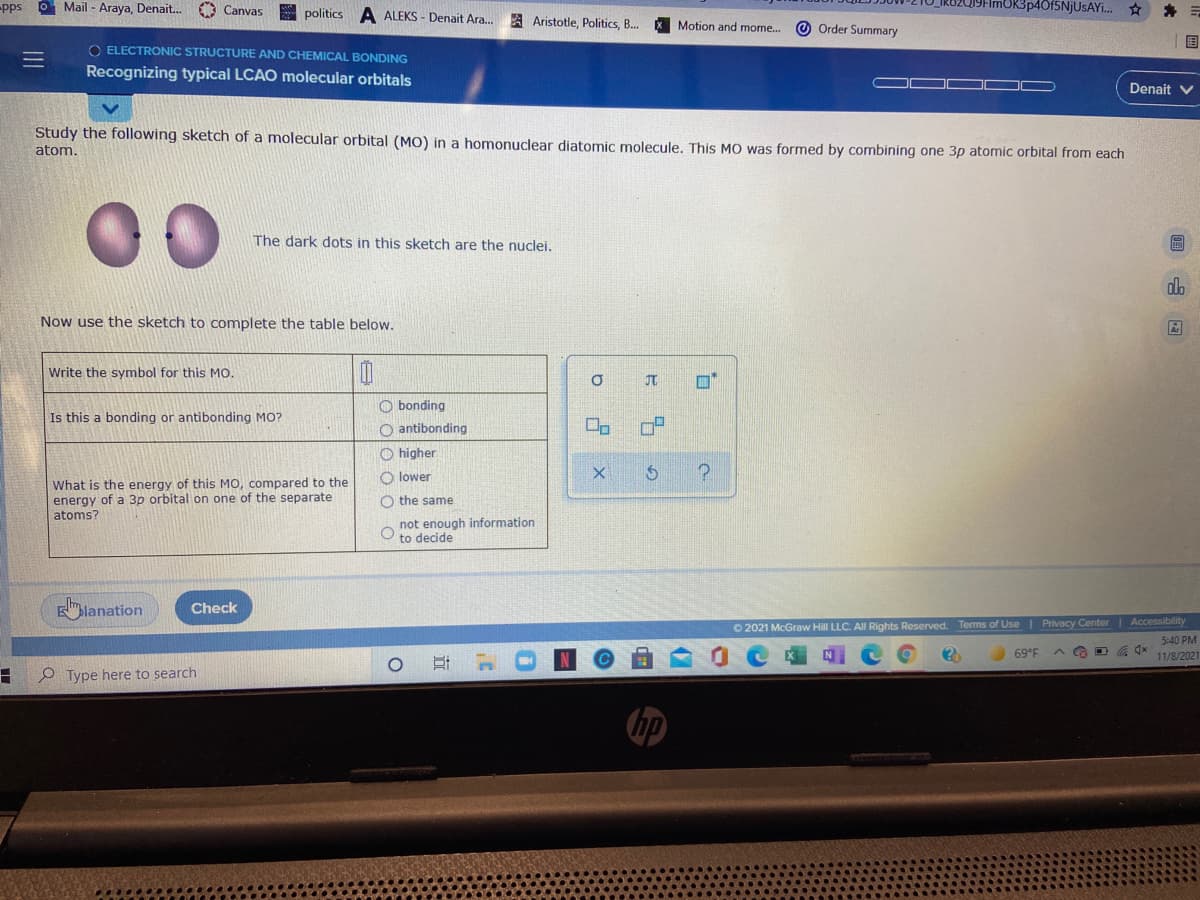
Study the following sketch of a molecular orbital (MO) in a homonuclear diatomic molecule. This MO was formed by combining one 3p atomic orbital from each atom. The dark dots in this sketch are the nuclei. Now use the sketch to complete the table below. Write the symbol for this MO. JT O bonding Is this a bonding or antibonding MO? O antibonding O higher O lower What is the energy of this MO, compared to the energy of a 3p orbital on one of the separate atoms? O the same not enough information to decide
Study the following sketch of a molecular orbital (MO) in a homonuclear diatomic molecule. This MO was formed by combining one 3p atomic orbital from each atom. The dark dots in this sketch are the nuclei. Now use the sketch to complete the table below. Write the symbol for this MO. JT O bonding Is this a bonding or antibonding MO? O antibonding O higher O lower What is the energy of this MO, compared to the energy of a 3p orbital on one of the separate atoms? O the same not enough information to decide
Chemistry: Principles and Practice
3rd Edition
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Chapter10: Molecular Structure And Bonding Theories
Section: Chapter Questions
Problem 10.101QE: The molecular orbital diagram of NO shown in Figure 10.47 also applies to OF. Draw the complete...
Related questions
Question
Transcribed Image Text:pps
O Mail - Araya, Denait.
Canvas
politics
3P40F5NJUSAY.. ☆
ALEKS - Denait Ara.
A Aristotle, Politics, B.
Motion and mome.
O Order Summary
国
O ELECTRONIC STRUCTURE AND CHEMICAL BONDING
Recognizing typical LCAO molecular orbitals
Denait v
Study the following sketch of a molecular orbital (MO) in a homonuclear diatomic molecule. This MO was formed by combining one 3p atomic orbital from each
atom.
The dark dots in this sketch are the nuclei.
dlo
Now use the sketch to complete the table below.
Write the symbol for this MO.
JT
O bonding
Is this a bonding or antibonding MO?
O antibonding
O higher
O lower
What is the energy of this MO, compared to the
energy of a 3p orbital on one of the separate
atoms?
O the same
not enough information
to decide
E"blanation
Check
Accessibility
O 2021 McGraw Hill LLC. All Rights Reserved. Terms of Use Privacy Center
5:40 PM
69°F
11/8/2021
O Type here to search
hp
石の
II
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning