Suppose object A has triple the specific heat and double the mass of object B. If the same amount of heat is applied to both objects, how will the temperature change of A be related to the temperature change of B? Enter your answer to three significant figures. Temperature change of A = i x (temperature change of B) eTextbook and Media Save for Later Attempts: 0 of 15 used Submit Answer
Suppose object A has triple the specific heat and double the mass of object B. If the same amount of heat is applied to both objects, how will the temperature change of A be related to the temperature change of B? Enter your answer to three significant figures. Temperature change of A = i x (temperature change of B) eTextbook and Media Save for Later Attempts: 0 of 15 used Submit Answer
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter12: Thermodynamic Processes And Thermochemistry
Section: Chapter Questions
Problem 11P
Related questions
Question
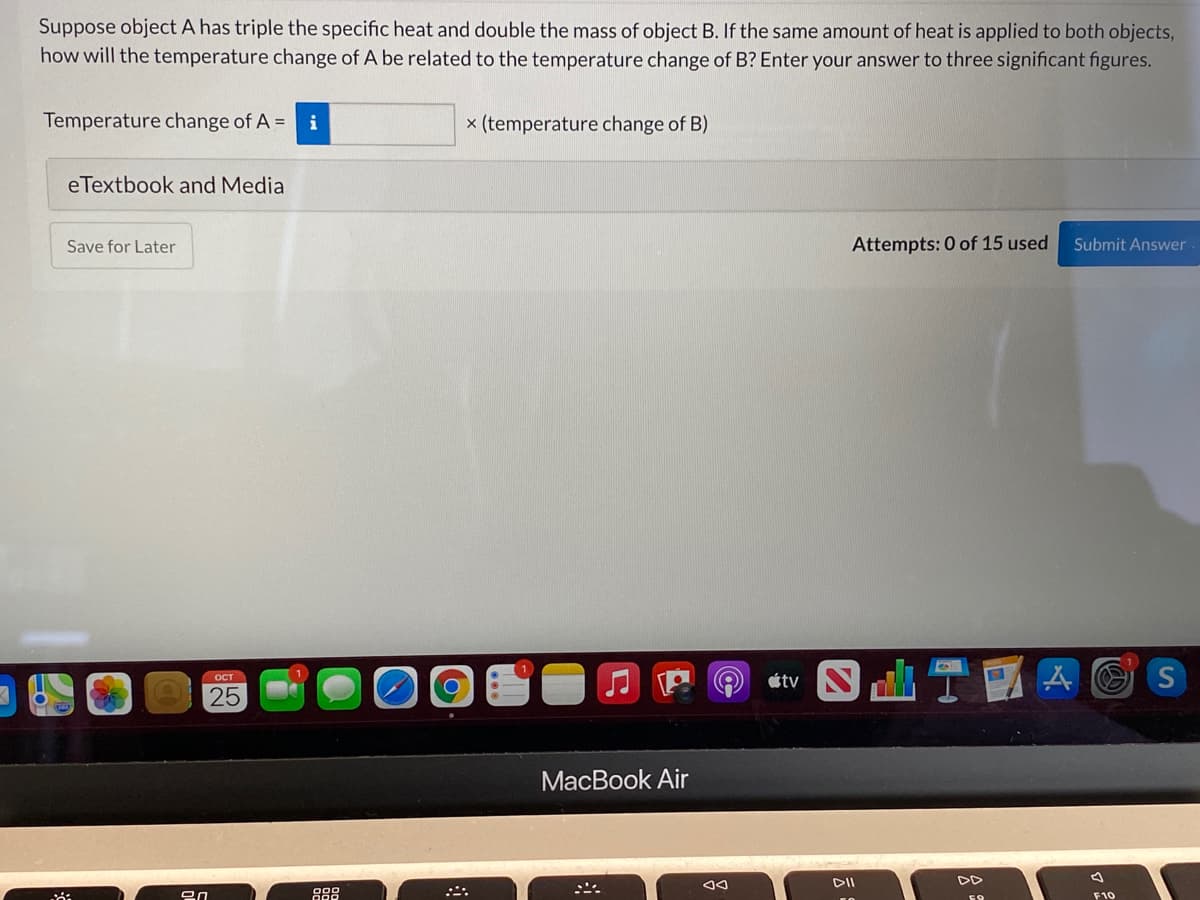
Transcribed Image Text:Suppose object A has triple the specific heat and double the mass of object B. If the same amount of heat is applied to both objects,
how will the temperature change of A be related to the temperature change of B? Enter your answer to three significant figures.
Temperature change of A = i
x (temperature change of B)
eTextbook and Media
Save for Later
Attempts: 0 of 15 used
Submit Answer
S TA O
OCT
étv
25
МacBook Air
DII
88
F10
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning
