Suppose you were preparing 1.0 L of a bleaching solution in a volumetric flask, and it calls for 0.24 mol of NaOCl If all you had available was a jug of bleach that contained 0.73 MNaOCl, what volume of bleach would you need to add to the volumetric flask before you added enough water to reach the 1.0 L line? Express the volume in milliliters to two significant figures.
Suppose you were preparing 1.0 L of a bleaching solution in a volumetric flask, and it calls for 0.24 mol of NaOCl If all you had available was a jug of bleach that contained 0.73 MNaOCl, what volume of bleach would you need to add to the volumetric flask before you added enough water to reach the 1.0 L line? Express the volume in milliliters to two significant figures.
Chemistry for Today: General, Organic, and Biochemistry
9th Edition
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Chapter7: Sollutions And Colloids
Section: Chapter Questions
Problem 7.2E
Related questions
Question
100%
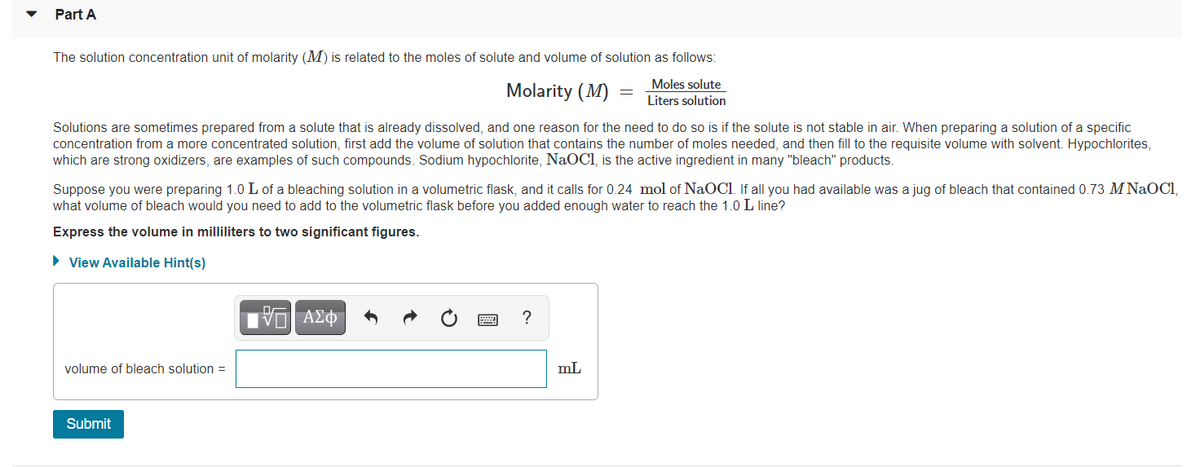
Transcribed Image Text:Part A
The solution concentration unit of molarity (M) is related to the moles of solute and volume of solution as follows:
Moles solute
Molarity (M)
Liters solution
Solutions are sometimes prepared from a solute that is already dissolved, and one reason for the need to do so is if the solute is not stable in air. When preparing a solution of a specific
concentration from a more concentrated solution, first add the volume of solution that contains the number of moles needed, and then fill to the requisite volume with solvent. Hypochlorites,
which are strong oxidizers, are examples of such compounds. Sodium hypochlorite, NaOCI, is the active ingredient in many "bleach" products.
Suppose you were preparing 1.0 L of a bleaching solution in a volumetric flask, and it calls for 0.24 mol of NaOCl. If all you had available was a jug of bleach that contained 0.73 M NAOCI,
what volume of bleach would you need to add to the volumetric flask before you added enough water to reach the 1.0 L line?
Express the volume in milliliters to two significant figures.
• View Available Hint(s)
?
volume of bleach solution =
mL
Submit
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning


Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning


Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning