Macroscale and Microscale Organic Experiments
7th Edition
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Kenneth L. Williamson, Katherine M. Masters
Chapter37: Dibenzalacetone By The Aldol Condensation
Section: Chapter Questions
Problem 10Q
Related questions
Question
Transcribed Image Text:b (a) Interpretatic X
B Homepage-C X
M Inbox - frances X
101 Chem101
M Inbox - fmehl1 X
Ô https://app.101edu.co
Question 4 of 4
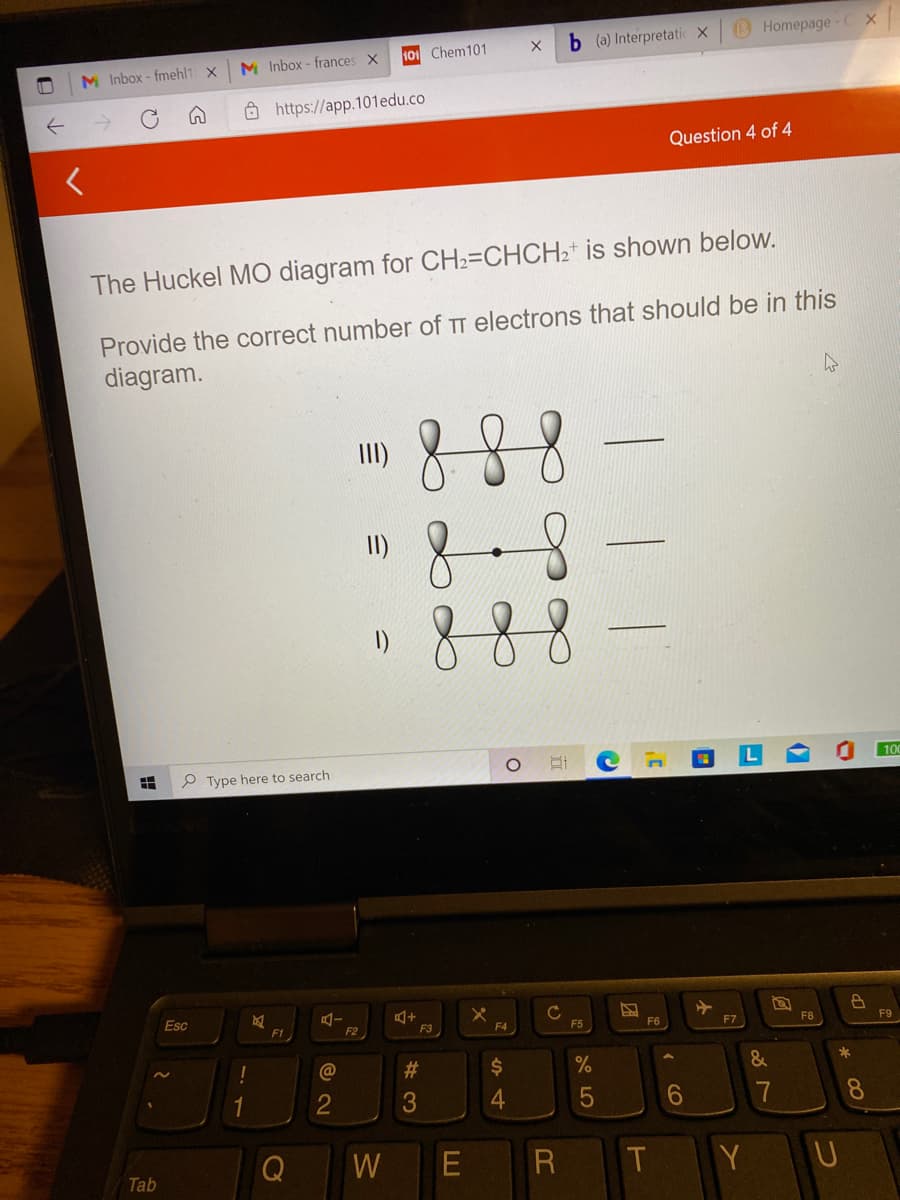
The Huckel MO diagram for CH2=CHCH2* is shown below.
Provide the correct number of TT electrons that should be in this
diagram.
88
8
888
III)
of
I)
100
P Type here to search
Esc
F1
F2
F3
F4
F5
F6
F7
F8
F9
%23
%24
&
*
1
2
4.
6
8.
Q
W
Y U
Tab
寺
出
Expert Solution
Step 1
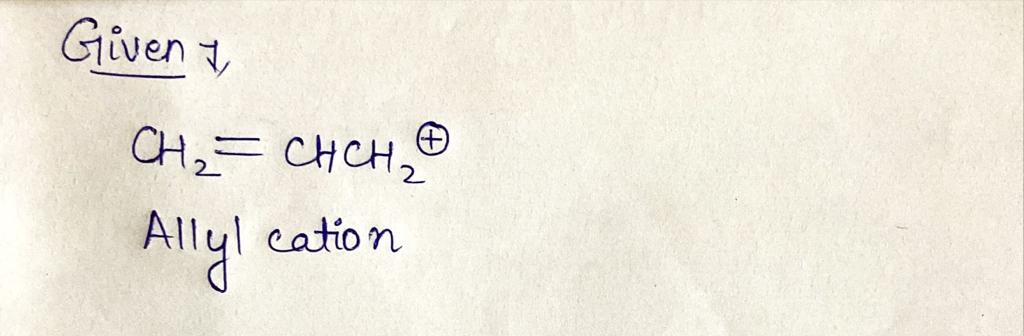
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,