TABLE 13.3 Molal Boiling-Point-Elevation and Freezing-Point-Depression Constants Normal Boiling Point (°C) 100.0 K, (°C/m) Normal Freezing Point (°C) K; (°C/m) Solvent Water, H2O 1.86 0.51 0.0 Benzene, C,H, 2.53 80.1 5.5 5.12 Ethanol, C,H¿OH Carbon tetrachloride, CCI4 78.4 1.22 -114.6 1.99 76.8 -22.3 29.8 5.02 Chloroform, CHCI3 4.68 61.2 3.63 -63.5
TABLE 13.3 Molal Boiling-Point-Elevation and Freezing-Point-Depression Constants Normal Boiling Point (°C) 100.0 K, (°C/m) Normal Freezing Point (°C) K; (°C/m) Solvent Water, H2O 1.86 0.51 0.0 Benzene, C,H, 2.53 80.1 5.5 5.12 Ethanol, C,H¿OH Carbon tetrachloride, CCI4 78.4 1.22 -114.6 1.99 76.8 -22.3 29.8 5.02 Chloroform, CHCI3 4.68 61.2 3.63 -63.5
Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter17: Spontaneity, Entropy, And Free Energy
Section: Chapter Questions
Problem 128IP: Some nonelectrolyte solute (molar mass = 142 g/mol) was dissolved in 150. mL of a solvent (density =...
Related questions
Question
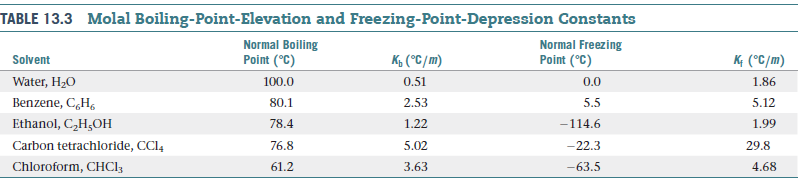
Using data from Table 13.3, calculate the freezing and boiling
points of each of the following solutions: (a) 0.22 m
glycerol (C3H8O3) in ethanol, (b) 0.240 mol of naphthalene
(C10H8) in 2.45 mol of chloroform, (c) 1.50 g NaCl
in 0.250 kg of water, (d) 2.04 g KBr and 4.82 g glucose
(C6H12O6) in 188 g of water.
Transcribed Image Text:TABLE 13.3 Molal Boiling-Point-Elevation and Freezing-Point-Depression Constants
Normal Boiling
Point (°C)
100.0
K, (°C/m)
Normal Freezing
Point (°C)
K; (°C/m)
Solvent
Water, H2O
1.86
0.51
0.0
Benzene, C,H,
2.53
80.1
5.5
5.12
Ethanol, C,H¿OH
Carbon tetrachloride, CCI4
78.4
1.22
-114.6
1.99
76.8
-22.3
29.8
5.02
Chloroform, CHCI3
4.68
61.2
3.63
-63.5
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 6 steps with 6 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning