The density of liquid benzene, C6H6, is 0.879 g/cm^3 at 15°C. How many moles are in 73.0 mL of benzene at 15°C? A. 0.365 mol B. 0.532 mol C. 2.43 mol D. 0.821 mol E. 0.467 mol
Q: CH3 1.ВН,THF a) 2. H2O2/-OH Br KOC(CH3)/isi b) OC2H5 1. 2 mol CeHgMgBr ? c) 2. H30* 1. NaOH (aq.) d)…
A:
Q: Balance the following equations:
A: balancing of reaction is done to follow the conservation of mass.
Q: Крand Kc the sanme?* А. 2 СО(g) + 0-(g) B. PCI5(g) → PC13(g) + Cl2(g) С. N2(g) + 3 H-(g) —> 2 NH3(g)…
A: Kp = Kc(RT)∆n ∆n = number of moles of gaseous product - number of moles of gaseous reactants
Q: A app.101edu.co E Apps M Gmail O YouTube A Maps 1 VitalSource Books. Question 1 of 24 Submit Balance…
A:
Q: How many moles of KBr are in 61.3 grams of KBr?
A: We are given with 61.3 grams of KBr and we have to calculate the moles of KBr present.
Q: 1) Determine the number of moles in each sample: a) 38.2g sodium chloride b) 36.5g nitrogen monoxide…
A:
Q: 56.172g of pure (elemental) Si is equivalent o how many moles of Si? A): 6.02 x 10^23 moles of…
A: SOLUTION Mass of elemental Si = 56.172 g Atomic mass of Si = 28.086 g/mol Moles of Si =…
Q: a) Determine the number of atoms in 1.75 mol Al(i) Calculate the number of formula units in 25.0 mol…
A: MOLES: In chemistry, a mole is the scientific unit which represents the quantity of the substance…
Q: Caproic acid, which is responsible for the foul odor of dirty socks, is composed of C, H, and O…
A: Since you have posted questions with multiple sub-parts, we are entitled to answer the first 3 only.…
Q: Phogene (COCl2) can be manufactured by mixing carbon monoxide with chlorine gas. CO + Cl2 → COCl2…
A: CO + Cl2 .....> COCl2 Given that : Moles of carbon monoxide(CO) = 7.25 moles
Q: How many moles of methane (CHA) are in 20.0 grams of methane? 20.0 moles 16.0 moles 1.25 moles
A: Moles is defined as the amount of substance that contains same number of particles as contain in 12…
Q: The molar mass for F e (C O 3 ) is: A. 251.4 grams/mole B.115.8 grams/mole C.139.8 grams/mole D.83.8…
A: We have given that The molar mass for Fe(CO3) is: A. 251.4 grams/mole B.115.8 grams/mole C.139.8…
Q: Ascorbic acid ( vitamin C) molar mass 176g/mol A healthy adult's daily requirement is 70-90 mg a…
A: The daily requirement of vitamin C for a healthy adult's is = 70-90 mg The molar mass of vitamin C…
Q: Assume that the molar mass of A is 82.93 grams per mole and the molar mass of E is 24.61 grams per…
A: Atoms are the smallest unit of the matter, anything which occupies space and has mass is known as…
Q: How many grams O in 221.8 g Fe(OH)g? Molar mass of Fe(OH)g is 106.867 Hint: 1 mole Fe(OH)3 has 3 mol…
A:
Q: How many moles are in 220.0g of NaCl? Round to 2 decimal places.
A: Mole concept: The number of molecules or atoms present in the one mole of the substance is equal to…
Q: Convert all of the following to mol a)25.1 kg FeFe (NO3)2 b)0.5500 g C8H18 c)151 kg CaO…
A: From molecular weight we can calculate the no of moles and the no of molecules .
Q: 6.) 2 H2O(1) + Al(s) + MnO4 (aq) → Al(OH)4 -(aq) + MnO2(s) 7.) Mg (s) + 2 HC1 (g) →MgCl2 (s) + H2…
A: We have to tell which species is oxidised and reduced
Q: 12) Na + H2O > NaOH + H2 13) Ag20 → Ag + _O2 14) Se + _O2 →_SO3 15) CO2 + H2O → C6H1206 + O2 16) K+…
A: Balance of reaction. In order to balance a reaction the number of atoms present at the left hand…
Q: (d) you (e) HCI - 80°C (f) HBr 40 °C
A: The complete chemical reaction with proper reactants and products are given in a attached image.
Q: Copyright © Savvas Learning Company T04.2 9 T04.2 9 CCC Energy and Matter Interpret the equation for…
A:
Q: Number of molecules in 9×10−6 molmol CH3CH2OHCH3CH2OH. Express your answer using four significant…
A: No. of moles of CH3CH2OH = 9×10-6 No. of molecules in 9×10-6 moles of CH3CH2OH can be calculated as…
Q: CIF 3(9) AH = ? 2.) CIF9) + F2(g) AH = - 49.4 kJ 20F2(9) Ozlg) + 2F2(9) AH= 205.6 kJ Cl,0(9) +…
A:
Q: 4. KFE3AISI3010(OH)2 + Cu + 02 + H2S → KAISİ3O8 + CuFeS2 + H2O
A: A balanced chemical equation is the one that contains equal number of atoms on both sides of the…
Q: When someone in my class asked the professor I'm not sure if she misunderstood and just said yes so…
A: Number of moles of He is determined,
Q: Part 2 O See Hir Consider a sample of gas that contains 625 moles of smokestack gas. How many…
A:
Q: C3H8 + O2 > CO2+ H2O C3H8 O2 CO2 H2O mol 5 mol mol 4 mol 3.33 mol mol 9 mol 13.32 mol mol 26.25 mol…
A: C3H8 + 5O2 → 3CO2 + 4H2O C3H8 O2 CO2 H2O 1 mol 5 mol 3 mol 4 mol 3.33 mol 16.65…
Q: a 5.000g sample that contained NaCl and BaCl2 *2H2O was heated. After heating the sample had a…
A: Given : Mass of sample of NaCl and BaCl2*2H2O = 5.000 g And mass of sample after heating = 4.688 g
Q: Laughing gas, N20, can be synthesized from H2 and NO: H2(g) + 2 NO(g)= N20(g) + H2O (g), Kc = 2.3 x…
A: If Kc >Qc then equilibrium will shift in forward direction ( right side ) If Kc< Qc then…
Q: How many grams O in 115.6 g Fe(OH)g? Molar mass of Fe(OH)g is 106.867 Hint: 1 mole Fe(OH)g has 3 mol…
A: Given: Molar mass of Fe(OH)3 = 106.867 Mass of Fe(OH)3 = 115.6 g. And each mole of Fe(OH)3 has 3…
Q: 50.0 mL 2.00M HCI and 50.0 mL 2.00 M NaOH mixed: HCI(aq) + NaOH(aq) → H,0() + NaCI(aq) - Csoln =…
A: In just above step of circled part, we have calculated the total heat released during the reaction…
Q: many
A:
Q: Zn(s) +. ZnCl (aq) + H2(g) 3. HCl(aq) Cu(s) + HC(aq) CuCly(aq) +. H2g) - Mg(s) + HCI(aq) MgCl2(aq) +…
A:
Q: Hi. I am trying to calculate the percent of water in an uknown hydrate. The given data is: mass of…
A: Law of conservation of Mass :- Law of conservation of mass states that mass can neither be created,…
Q: Find the percentage by mass of Cl in CaCl2 if it is 36.1% Ca by mass. (Round your answer to one…
A: (1) The percentage by mass of Cl is 63.8% in CaCl2 if it is 36.1% Ca by mass. (2) the…
Q: Cgrafito + 2H2 CH4(9) CH4(g) + 202(g) CO2(9) + 2H2Ou) ACH°= -802.37 kJ/mol Cgrafito + O2(g) CO2(9)…
A: Applying Hess's law and with help of data given it is determined that formation∆HCH4.
Q: Calculate the equilibrium constant for each reaction using the balanced chemical equations and the…
A: The ratio of products and reactants raised to the power of their respective coefficients in a…
Q: Hudrobromic acid t Potossium hydrovide7 Potassium bromide t HeO 70.50 M) (150m) (0.35M)( 295ml)…
A: Since you have posted multiple questions, we are entitled to answer the first only. Please repost…
Q: 5. How many molecules are in 142.69 L of CH4? A V-M. a. 8.59 x1025 molecules b. 6.37 molecules C.…
A: Mole concept: The number of molecules or atoms present in the one mole of the substance is equal to…
Q: How many moles are in 2.5 g of N2? 0.089 moles 0.18 moles 1.3 moles 11 moles
A: We have give that Mass of N2 = 2.5 g And we have to find number of moles in 2.5 g of N2
Q: Which of the following contains the greatest number of molecules? A. 1 g of CH3CI В. 1 g of CH,Cl2…
A:
Q: 39.) number of moleeules? a. 4.75 mmol of phosphire, PHs a 4.75g of phophney PHs e. 1.25 x10 g eof…
A: Since we only answer up to 3 sub-parts, we’ll answer the first 3. If you need help with other sub…
Q: (1.47 x 10-4 mol/L)2(2.3 x 10-3 mol/L) / (8.23 x 10-5 mol/L)(2.99 x 10-2 mol/L)
A: The equation given is, => (1.47 × 10-4 mol/L)2 × (2.3 × 10-3 mol/L)(8.23 × 10-5 mol/L) (2.99 ×…
Q: 4 Na O2 2 Naz0 12 mol 0.1 mol 2 AI 3 Cl2 2 AICI, 5 mol 0.1 mol 6% O2 4 CO2 5 H20 C4H10 0.5 mol 20…
A:
Q: CH3 1.BH3/THF a) 2. H2О2/-ОН Br KOC(CH3)3/isi b) ? OC2H5 1. 2 mol CoHsМgBr ? c) 2. H30*
A:
Q: Identify each of the following reactions as a precipitation, neutralization, or redox reaction:
A: (a) Redox reaction. Oxidation number of Mg is changed from 0 to +2. (b) Neutralization reaction.…
Q: 4. For each of the following chemical ........a. CH4(g) + O2(g) → CO2(g) + H2O(g)b. Al(s) + HCl(aq)…
A: (a)In a balanced equation number of atoms for every element and total change in the reaction are…
Q: 1. What is wrong with each of the following setups? NOTE: There may be nothing wrong or multiple…
A:
The density of liquid benzene, C6H6, is 0.879 g/cm^3 at 15°C. How many moles are in 73.0 mL of benzene at 15°C?
A. 0.365 mol
B. 0.532 mol
C. 2.43 mol
D. 0.821 mol
E. 0.467 mol
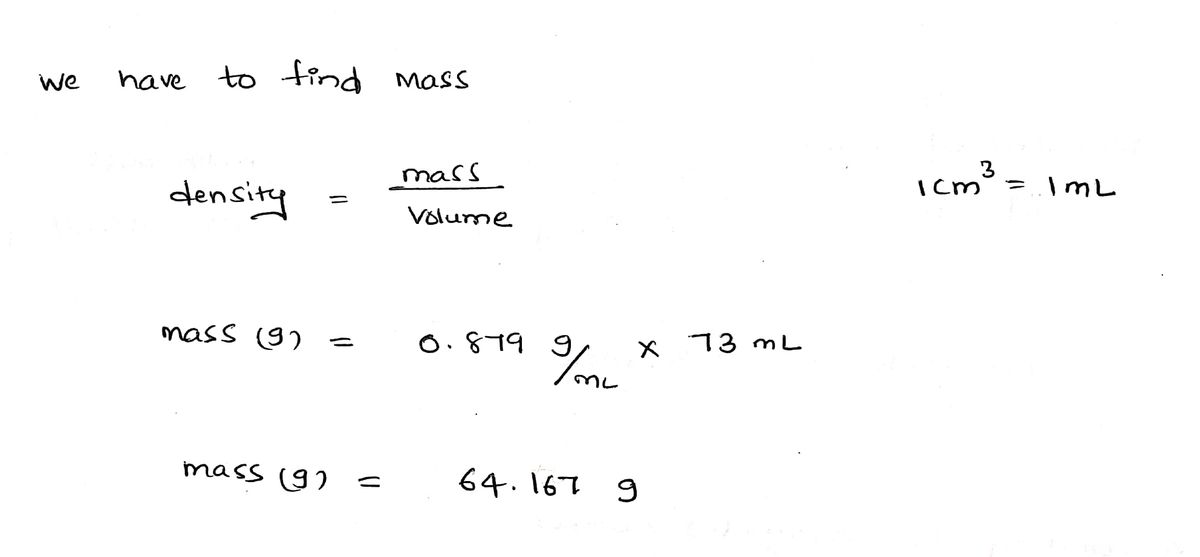
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Chemistry A group of students was tasked with synthesizing dibenzalacetone (DBA) in the lab. They used acetone as limiting reagent, calculated their TY to be 8.0 g DBA, obtained 14.0 g crude product (wet) and their % Yield was 65%. What was the actual yield for their final product (recrystallized, dry DBA)? What was the minimum volume (ml) of ethanol they needed to recrystallize their wet crude? What was the amount of impurities (side products, moisture,...) contained in their wet crude?Chemical conversions 1. What is the molecular weight of nitrobenzene, often known as C_6*H_5*O_2*N? 2. How many moles of nitrobenzen are contained in 50.0 g? 3. What is a substance's density in grammes per cubic centimetre if its specific gravity is 1.203? 4. How much space is filled by 50.0 g of nitrobenzene in cubic metres, feet, and inches? 5. What is the pressure at the base of a cylindrical container holding nitrobenzene that has a base that is 1 inch in diameter? What does gauge pressure mean? 6. How many molecules of nitrobenzene are in 50.0 g?A core sample is saturated with an oil (?o = 35oAPI), gas and water. The initial weight of the sample is 224.14 g. After the gas is displaced by water (?w = 1 g/cm3 ), the weight is increased to 225.90 g. The sample is the placed in a Soxhlet distillation apparatus, and 4.4 cm3 water is extracted. After drying the core sample, the weight is now 209.75 g. The sample bulk volume, 95 cm3 is measured in a mercury pycnometer. Find the porosity, water saturation, oil saturation, gas saturation and lithology of the core sample.
- The alcohol content of hard liquor is normally given in terms of the “proof,” which is defined as twice the percentage by volume of ethanol (C2H5OH) present. Calculate the number of grams of alcohol present in 1.00 L of 75-proof gin. The density of ethanol is 0.798 g/mL. Round off to 4 sig. figures for all calculations made prior to final answer and the final answer should be in 3 sig. figuresArterial blood pressure is traditionally measured in mm Hg. And the style pressure of 85 mm Hg it’s considered borderline between normal and high blood pressure convert this pressure to kPa units. ( 1atm =760 mm Hg= 101.33 kPa )The molar volumes of pure acetone and pure chloroform at room temperature are 72.0 cm^3 and 81.0 cm^3, respectively. The volume of a liquid mixture containing 0.6 moles of acetone and 0.5 moles of chloroform is
- When concentrated nitric is sold, the label contains no mention of the molarity of the acid. Instead, the label normally lists the concentration of nitric acid as a wt/wt percent and gives the specific gravity of the solution. If the solution is 68.0 (wt/wt %) and nitric acid and has a specific gravity of 1.41 kg/L, calculate the molarity of 68.0 % concentrated nitric acid. FW HNO3=63.01g/molWhat is the Molecular weight of a certain volatile liquid which is placed in 225 g flask which has total capacity volume of 251.07 ml where after the liquid was heated to 94.5 oC, the gas volatilized and the weight of the flask measured 227.08g.? Round you answer in one decimal placeA mixture of methanol and water contains 60.0% water by mass. Assuming volume additivity: If the actual specific gravity of the mixture is 0.9345: How many liters is required to provide 150 mol of methanol?
- Calculate the volume of 0.152 M C6H12O6 (aq) that should be used to prepare 25.0 mL of 1.59×10–5 M C6H12O6 (aq). A) 0.00262 L B) 2. 62 × 10–3 L C) 2.62 mL D) 2.62 μL E) 2.615 μLThe minimum level of mercury levels in water considered safe is 1 part per billion (ppb) by weight. Ifyou did an analysis and found that the concentration was 0.59 parts per billion. What quantity of mercuryin grams was present in 8.52 L of the water, the density of which is 0.998 g/ml? (1 ppb Hg = 1 ng Hg per1 g water)What is the molarity of a concentrated solution of sulfuric acid with a specific gravity of 1.84 and 95.0% by weight sulfuric acid? specific gravity = density of substance at certain temp/density of water at 4 degrees C the density of water is 0.999973 g/mL at 4 degrees celsius. Describe how to prepare 3.0 L of 0.100M. I will pipet ______ and add this to 200mL water. The solution will be stirred and diluted to 3L with water.

