The equilibrium constant for the “water gas" reaction C(s) + H2O(g) 2 C0(g) + H2(g) is K = 2.6 at a temperature of 1000 K. Calculate the reaction quotient Q for each of the following conditions, and state which direction the reaction shifts in coming to equilibrium. (a) PH;0 = 0.600 atm; Pco = 1.525 atm; PH, = 0.805 atm (b) P1,0 = 0.724 atm; Pco = 1.714 atm; PH, = 1.383 atm
The equilibrium constant for the “water gas" reaction C(s) + H2O(g) 2 C0(g) + H2(g) is K = 2.6 at a temperature of 1000 K. Calculate the reaction quotient Q for each of the following conditions, and state which direction the reaction shifts in coming to equilibrium. (a) PH;0 = 0.600 atm; Pco = 1.525 atm; PH, = 0.805 atm (b) P1,0 = 0.724 atm; Pco = 1.714 atm; PH, = 1.383 atm
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter12: Chemical Equilibrium
Section: Chapter Questions
Problem 12.3PAE
Related questions
Question
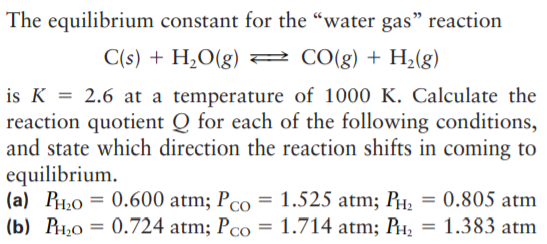
Transcribed Image Text:The equilibrium constant for the “water gas" reaction
C(s) + H2O(g) 2 C0(g) + H2(g)
is K = 2.6 at a temperature of 1000 K. Calculate the
reaction quotient Q for each of the following conditions,
and state which direction the reaction shifts in coming to
equilibrium.
(a) PH;0 = 0.600 atm; Pco = 1.525 atm; PH, = 0.805 atm
(b) P1,0 = 0.724 atm; Pco = 1.714 atm; PH, = 1.383 atm
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 4 images

Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning