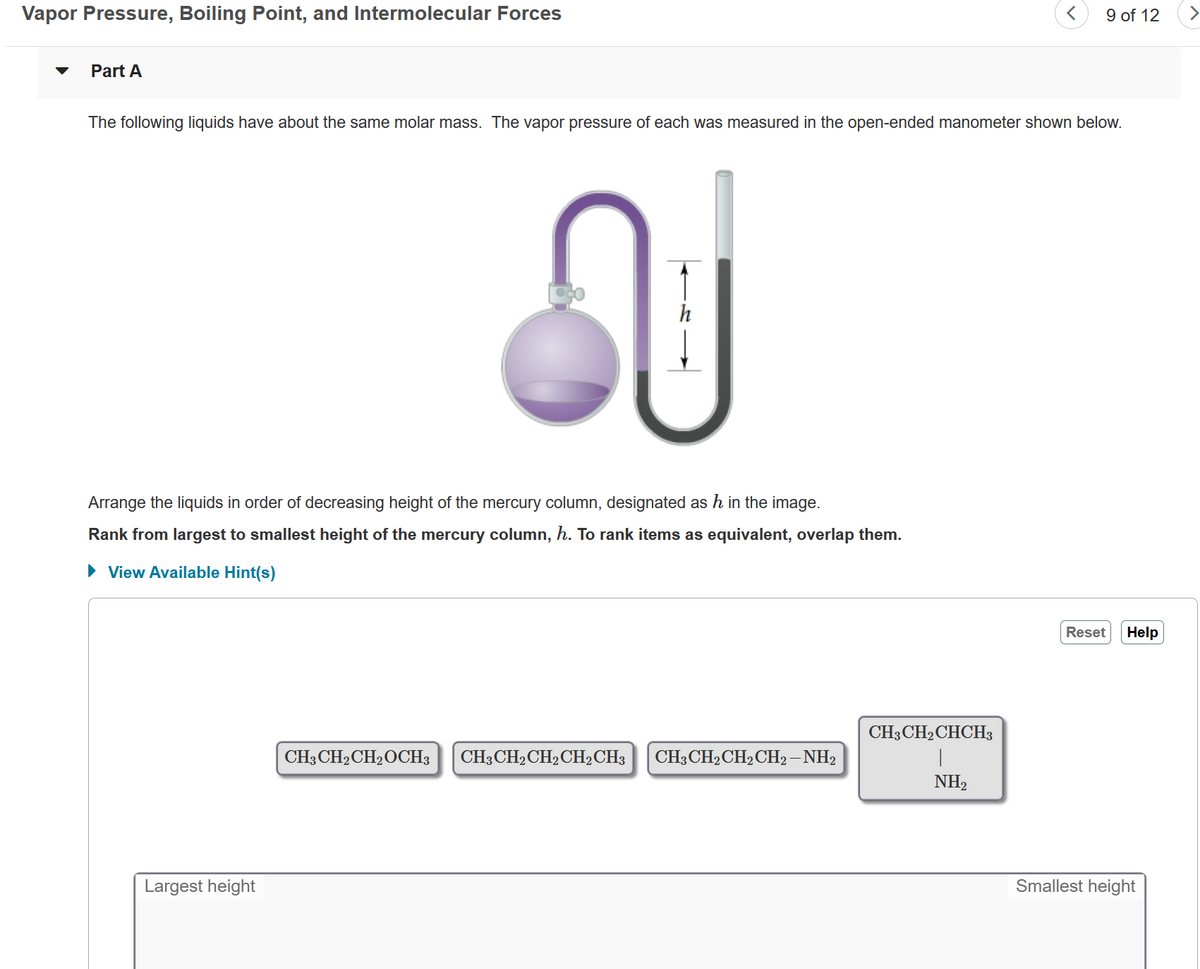
The following liquids have about the same molar mass. The vapor pressure of each was measured in the open-ended manometer shown below. Arrange the liquids in order of decreasing height of the mercury column, designated as h in the image. Rank from largest to smallest height of the mercury column, h. To rank items as equivalent, overlap them. • View Available Hint(s) Reset Hel CH-CH-CHCHз CH3CH2CH2OCH3 CH3CH2CH2CH2CH3 CH3CH2CH2CH2– NH2 NH2
The following liquids have about the same molar mass. The vapor pressure of each was measured in the open-ended manometer shown below. Arrange the liquids in order of decreasing height of the mercury column, designated as h in the image. Rank from largest to smallest height of the mercury column, h. To rank items as equivalent, overlap them. • View Available Hint(s) Reset Hel CH-CH-CHCHз CH3CH2CH2OCH3 CH3CH2CH2CH2CH3 CH3CH2CH2CH2– NH2 NH2
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter9: Liquids And Solids
Section: Chapter Questions
Problem 14QAP: Consider the following data for the vapor pressure of diethyl ether, a widely used anesthetic in the...
Related questions
Question
100%
I've attached a picture of the problem that I'm stuck on. Thanks!
Transcribed Image Text:Vapor Pressure, Boiling Point, and Intermolecular Forces
9 of 12
Part A
The following liquids have about the same molar mass. The vapor pressure of each was measured in the open-ended manometer shown below.
h
Arrange the liquids in order of decreasing height of the mercury column, designated as h in the image.
Rank from largest to smallest height of the mercury column, h. To rank items as equivalent, overlap them.
• View Available Hint(s)
Reset
Help
CH3CH2 CHCH3
CH3 CH2CH2OCH3
CH3 CH2CH2CH2CH3
CH3CH2CH2CH2–NH2
NH2
Largest height
Smallest height
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning