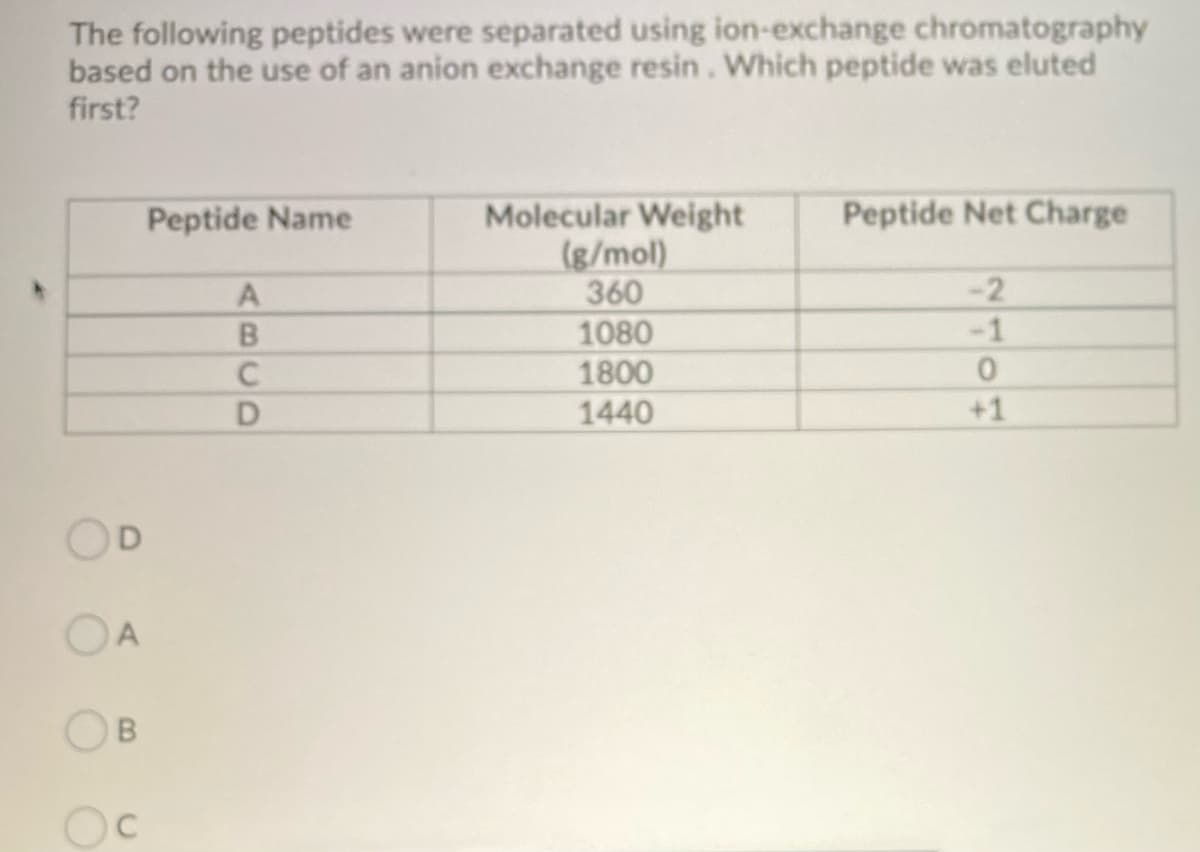
The following peptides were separated using ion-exchange chromatography based on the use of an anion exchange resin. Which peptide was eluted first? Peptide Name A B C D Molecular Weight (g/mol) 360 1080 1800 1440 Peptide Net Charge -2 0 +1
Q: Which of the following is zwitterion structure of the amino acid alanine? O NH3 NH3 O NH₂ O OH O no…
A: Amino acids are biomolecules that have an amino group (-NH2), a carboxyl group (-COOH) and a side…
Q: A patient weighing 38.4 pounds presents with a bacterial infection and is prescribed a course of…
A: A multitude of bacterial infections can be treated with the antibiotic amoxicillin. These include…
Q: Submit a drawing of the following phospholipid: -x-group is phosphoserine (use google if you need…
A: Glycerophospholipids or phospholipids are lipids found in the biological membrane. In phospholipids,…
Q: Suppose that the pH of distilled water is 6.2, what will be its resulting color using phenol red.
A: The pH of a solution refers to the H+ ion concentration in the solution. A solution with a pH lower…
Q: a) Draw the A-T and G-C base pairs. - Label the bases…
A: DNA are polymers of nucleotides. A nucleotide consists of a nitrogenous base(A, T, G, C) attached to…
Q: Analyze each item carefully and write your complete solution. Cysteine is an important amino acid…
A: Note: As per the honor code we are allowed to answer the first two questions. Kindly resubmit the…
Q: What is epistasis, and what is pleiotropy? Describe with examples.
A: “Since you have asked multiple question, we will solve the first question for you. If youwant any…
Q: Please help with 2a) 2a) There are two different DNA polymerase enzymes, DNA Polymerase I and DNA…
A: Replication is the process of duplication of two strands of a double stranded DNA. In bacteria, the…
Q: For each of the structures listed below identify the class of lipids to which it belongs (fatty acid…
A: Lipids are biomolecules that do not have a fixed chemical structure like carbohydrates or amino…
Q: Discuss how to determine the elements of proteins and its reactions.
A: Proteins are the most abundant biomacromolecule of life. Proteins are primarily polypeptide…
Q: In a healthy young adult, what may explain high levels of creatinine in serum and urine samples?
A: INTRODUCTION : Creatinine - It is a breakdown or by- product of creatine phosphate from muscles and…
Q: 1. Deduce the primary structure of this polypeptide. 2. Why would cyanogen bromide not be a good…
A: a) Given the molecular weight of the peptide is 4000Average MW of an amino acid is 110 Da. So, the…
Q: Concerning the citric acid cycle (TCA cycle), which of the following statements is false? The TCA…
A: Cellular respiration is a collection of three metabolic pathways that generate ATP by the oxidation…
Q: What effect is seen on a Lineweaver-Burke graph when a non-competitive inhibitor is added to an…
A: The enzymes can be regulated in presence of competitive, uncompetitive and noncompetitive…
Q: As the leading scientist in a biomedical science laboratory, it is up to you to give advice to your…
A: Polymerase chain reaction or PCR s a method to obtained large number of copies of a target DNA…
Q: Provide 3 most applications/purposes of DNA analysis.
A: Recall that: DNA is the genetic material passed down from one generation to another, ie, the…
Q: The activity of an enzyme can be regulated by a: A) competitive inhibitor binding to the active…
A: The enzymes increase the rate of biochemical reactions and can be regulated by binding to…
Q: 1) What is the full name of the following molecule? 2) Briefly explain the functions of this…
A: The biochemistry of cells is greatly influenced by a variety of biological components. These…
Q: The proton-motive force is a measure of the potential energy generated across the mitochondrial…
A: The catabolism of carbohydrates, fatty acids, and amino acids generates the reducing equivalents…
Q: The ΔG°' for hydrolysis of ATP to ADP + Pi is -30.5 kJ/mol. -Calculate the equilibrium constant for…
A: Standard Gibbs free energy change (∆G0') is the change in Gibbs free energy at standard conditions.
Q: Mutual intermediates of catabolism of proteins, carbohydrates and lipids; their role in the…
A: Catabolism is the process of breaking down of complex compounds into simple molecules. The catabolic…
Q: QUESTION 5 Oxaloacetate Acetyl-CoA AG" = -7.7 kcal/mol COA Citrate Fatty acids AG"=-1.5 kcal/mol…
A: Free energy change (∆G) of a reaction helps to find the spontaneity of a reaction. Negative ∆G of a…
Q: 384 Hemoglobin: Allostery and Evolution Q5.1 - 2,3-BPG is a negative allosteric regulator of…
A: Hemoglobin (Hb) is a protein that is found in red blood cells. A specific protein called haemoglobin…
Q: How to calculate percentages of arginine have positively charge at PH=7
A: pH is the measure of H+ ion concentration ( denoted by [H+]) in the solution. High pH means low H+…
Q: Canonical forces in protein folding. Describe how these forces come into play when a protein folds.
A: INTRODUCTION : Canonical forces in protein folding - Canonical forces are those forces which…
Q: Question 15 of 25 Which of the following is true for the acid-base properties of amino acids? Select…
A: Amino acids are the building blocks of proteins . they have two functional groups amine group and…
Q: 5. State whether the following statements are True or False, If False, explain why 5.1 Oxaloacetate…
A: 1. Oxaloacetate is formed by the oxidation of L-malate - Oxaloacetate is formed via oxidation of…
Q: Make use of the table below in answering the questions asked: Amino acid pK₁ PK₂ pK3 2.32 9.76 2.32…
A: Isoelectric point is the ph at which the peptide carries a net charge of 0. The proteins are least…
Q: In ATP synthase, the ____ subunit is the site of ATP synthesis while the ____ subunit forms the…
A: Oxidation of glucose in the glycolysis and TCA cycle generates electrons carriers NADH and FADH2, As…
Q: Relate the molecular properties to physicochemical properties of the following Linoleic acid…
A: Lipids are one of the 4 major biomacromolecules. Lipids are insoluble in water and soluble in…
Q: Calculate 1/[S] and 1/V to complete the table. Use this data to draw a Lineweaver-Burke plot, with…
A: A Lineweaver-Burk plot represents the relationship between the reciprocal values of substrate…
Q: Describe two different ways in which glucose oxidase is regulated. These mechanisms of regulation do…
A: Glucose oxidase: It is an oxidoreductase that catalyzes the conversion of glucose to hydrogen…
Q: Imagine a marine nitrogen fixing bacterium for the following questions. a. Calculate the cell’s…
A: INTRODUCTION : Marine nitrogen fixing bacterium - Marine N2 fixing bacterium(diazotrophs) convert…
Q: Q6.1: Three critical features of enzyme structure and function contribute to the overall efficiency…
A: The enzymes are biological catalysts that increases the rate of biochemical reactions. The enzymes…
Q: In the peptide, SWEETLY, bond via its side chain. can still form a standard peptide bond while can…
A: A standard peptide bond is formed as a result of a dehydration ( also called condensation ) reaction…
Q: Why carbon C-2 and C-5 are mark? why not just ona carbon is marked since just one carbon was marked…
A: Gluconeogenesis is the synthesis of glucose molecules from non-carbohydrate sources such as…
Q: QUESTION: For each enzyme identify the transition-state inhibitor analog by drawing a circle around…
A: Whenever a reaction takes place there are reactants and they form products but in between, during…
Q: The main stages of catabolism of biomolecules: proteins, carbohydrates and lipids.
A: Catabolism is the degradative pathway. It deals with the breakdown of complex molecules into simpler…
Q: In a Myoglobin and azide ligand-receptor binding experiment, instead of using 3.5 µM myoglobin you…
A: Myoglobin acts as oxygen reserve in the muscle cells. Myoglobin (Mb) has higher affinity towards…
Q: In the peptide, SWEETLY, bond via its side chain. (Give the full name of the amino acids) can still…
A: Recall that: for a given peptide amino acid sequences are written with N-terminal amino acid on the…
Q: The first reaction in glycolysis is the phosphorylation of glucose to form glucose 6-phosphate: P: +…
A: Given Values: ∆G°=13.8 kJ/mol or 13800 J/mol[Pi] = 5 mM[Glucose] = 5 mM
Q: What would be the standard free energy change if FADH 2 is used to reduce O 2 to water? Standard…
A: Biological oxidation-reduction reactions involve the transfer of electrons from one biomolecule,…
Q: Draw his trp arg pro glu met ile | PH = 2 1b) How many possible structures are there for this? 1c)…
A: Peptides are composed of amino acids. Amino acids are biomolecules with an alpha carbon bonded to an…
Q: 1. Write a short note on transcription. What is TFBS? What is the role of RNA polymerase?
A: Since you have asked multiple questions with multiple subparts, we will solve the first question for…
Q: In the corn snake Pantherophis guttatus, there are several different color variants, including…
A: Introns are non-coding sequences and exons are coding sequences in the gene. Splicing is the process…
Q: 12. The graph shows values of arterial plasma bicarbonate concentration and pH for different persons…
A: Metabolic alkalosis is caused by acid base imbalance in the blood. It is characterised by increase…
Q: 1. The second high energy intermediate metabolite of glycolysis that can be used for substrate level…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: What is one technique or property of a protein that you could use to monitor the fractions so you…
A: In column chromatography, there is a stationary phase and the mobile phase. The stationary phase…
Q: Which of the following statements is FALSE regarding oxidative phosphorylation? The pH is higher in…
A: Cellular respiration is a collection of three metabolic pathways that generate ATP by oxidation of…
Q: What are the components of a nucleotide? Provide 1- or 2-sentence description of each component.
A: Nucleic acids are biomolecules that are essential for all life forms. They are polymers of…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- Which method of chromatography would be the most appropriate for separating mixtures of thefollowing peptides at a neutral pH? (a) ARDDEKLLML, LMYWWILLDETSARALIV, DQNNA(b) GATSKKIVLM, ASCILLVMEN, MFDDAAYVNE(c) KLMDAATSPFLMAGHHHHHH, MFPSATEKRQQNNESTVLAG In what order would the following peptides be eluted from an anion exchange column at pH 7.5?Peptide 1: Ala–Asp–Glu–Gln–Trp–Leu–His–ValPeptide 3: Phe–Gln–Glu–Ala–Gln–His–Leu–Arg-IlePeptide 2: Arg–Ile–Pro–Lys–Cys–Met–LysPlease draw the given peptide and calculate the net charge at pH 1, 4, 8 and 12. GRNVGHEWAA biochemist wants to separate two peptides by ion-exchange chromatography. At the pH of the mobile phase to be used on the column,one peptide (A) has a net charge of −3 due to the presence of more Glu andAsp residues than Arg, Lys, and His residues. Peptide B has a net charge of+1. Which peptide would elute first from a cation-exchange resin? Whichwould elute first from an anion-exchange resin?
- Please calculate and plot the charge on a peptide with the sequence of NEYK over the pH range from 1 to 13. Please use the pKa values from the table. You may calculate the charge at 1 pH unit intervals.Draw the peptide PVLED and determine the following: Isoelectric point, pI The net charge at pH = 3.2 ; pH = 7.6 and pH = 9.3 Complete name of the peptide Name using the 3-letter codeUsing mass spec, the primary structure of 4 peptides is obtained from a tissue sample: VMDEL, GHKRT, CNPQI, and SAWFY. The pI of each peptide is calculated and ion exchange chromatography is used to purify each peptide. VMDEL, GHKRT, CNPQI, and SAWFY were separated into 4 pure samples. The labels on all the samples are erased by mistake, and the mass spec is no longer working. You can run a few simple tests to identify the samples. What are 4 tests to identify each peptide and why?
- Above or below what pH would the polypeptide become fully mobile on a cation exchange column? Explain reasoning please. Per my understanding, I believe I need to find pI and then select a pHExamine the following peptide and the inserted table, and answer the 2 questions below: Thr-Glu-Pro-Ile-Val-Ala-Pro-Met-Glu-Tyr-Gly-Lys 1. Estimate the net charge of the peptide at pH 2.0. Explain. 2. Estimate the net charge of the peptide at pH 7.0. Explain.Before high performance liquid chromatography (HPLC) methods were devised for the separation and analysis of small peptides, electrophoresis on a paper support was frequently used. Separation was affected on the basis of the charge on a peptide at different pH values. a. Calculate the pI (isoelectric point) of the following primary structures of the following protein i.) Lys- Gly- Ala- Glyii.) Lys- Gly-Ala-Gluiii.) His- Gly- Ala- Gluiv.) Glu- Gly- Ala -Gluv.) Gln-Gly-Ala-Lys b.) the migration toward the cathode, the negative pole; A for the migration toward the anode, the positive pole; and O if the peptide remains stationary.
- Five amino acids were subjected to normal phase chromatography, and the following Rf values were computed: A - 0.59 B - 0.12 C - 0.71 D - 0.33 E - 0.20 Which is the most polar amino acid?An amino acid mixture consisting of lysine,leucine, and glutamic acid is to be separated by ion-exchangechromatography, using a cation-exchange resin at pH 3.5, with theeluting buffer at the same pH. Which of these amino acids will beeluted from the column first? Will any other treatment be neededto elute one of these amino acids from the column?When performing a separation by ion-exchange chromatography, the rate of elution of an amino acid is influenced by three main factors: a. Temperature b. Solution (buffer) pH c. Ionic strength (buffer concentration) of the eluent Briefly, explain how each of these parameters influences elution rate. Please provide only typed answer solution no handwritten solution needed allowed

